Bioventus01.31.18
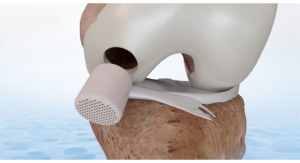
CartiHeal, developer of the proprietary Agili-C implant for the treatment of joint surface lesions in traumatic and osteoarthritic joints, has secured a $2.5 million investment from Bioventus, a global orthobiologics developer—reflecting the growing interest in CartiHeal’s technology.
Bioventus’s $2.5 million boost complements CartiHeal’s latest financing round, led by aMoon, together with Johnson & Johnson Innovation (JJDC Inc.), Peregrine Ventures, and Elron, bringing the total round to $21 million.
The funding will focus on CartiHeal’s ongoing Agili-C IDE clinical study. Further to the trial's initiation three months ago, over 30 patients were already enrolled, according to enrollment rate forecasts.
“This investment is a vote of confidence by an important orthobiologic strategic player. Bioventus joins our team of strategic investors who perceived the potential of the Agili-C implant. We are confident that Bioventus will bring substantial added value to CartiHeal,” said CartiHeal CEO Nir Altschuler.
The Agili-C IDE study is set to include a minimum of 250 patients in U.S. and non-U.S. centers, aiming for a PMA submission. The trial's objective is to demonstrate the superiority of the Agili-C implant over the surgical standard of care (microfracture and debridement) for the treatment of cartilage or osteochondral defects, in both osteoarthritic knees and knees without degenerative changes.
“Agili-C shows great promise to relieve osteoarthritis pain for a large patient population and we look forward following the pivotal trial to a successful culmination,” said Tony Bihl, CEO of Bioventus.
CartiHeal, a privately-held medical device company with headquarters in Israel, develops proprietary implants for the treatment of cartilage and osteochondral defects in traumatic and osteoarthritic joints.
Bioventus is an orthobiologics company that delivers clinically proven, cost-effective products that help people heal quickly and safely. Its mission is to make a difference by helping patients resume and enjoy active lives. Orthobiologic products from Bioventus include offerings for bone healing, bone graft and knee osteoarthritis. The company is based in Durham, N.C.
Bioventus’s $2.5 million boost complements CartiHeal’s latest financing round, led by aMoon, together with Johnson & Johnson Innovation (JJDC Inc.), Peregrine Ventures, and Elron, bringing the total round to $21 million.
The funding will focus on CartiHeal’s ongoing Agili-C IDE clinical study. Further to the trial's initiation three months ago, over 30 patients were already enrolled, according to enrollment rate forecasts.
“This investment is a vote of confidence by an important orthobiologic strategic player. Bioventus joins our team of strategic investors who perceived the potential of the Agili-C implant. We are confident that Bioventus will bring substantial added value to CartiHeal,” said CartiHeal CEO Nir Altschuler.
The Agili-C IDE study is set to include a minimum of 250 patients in U.S. and non-U.S. centers, aiming for a PMA submission. The trial's objective is to demonstrate the superiority of the Agili-C implant over the surgical standard of care (microfracture and debridement) for the treatment of cartilage or osteochondral defects, in both osteoarthritic knees and knees without degenerative changes.
“Agili-C shows great promise to relieve osteoarthritis pain for a large patient population and we look forward following the pivotal trial to a successful culmination,” said Tony Bihl, CEO of Bioventus.
CartiHeal, a privately-held medical device company with headquarters in Israel, develops proprietary implants for the treatment of cartilage and osteochondral defects in traumatic and osteoarthritic joints.
Bioventus is an orthobiologics company that delivers clinically proven, cost-effective products that help people heal quickly and safely. Its mission is to make a difference by helping patients resume and enjoy active lives. Orthobiologic products from Bioventus include offerings for bone healing, bone graft and knee osteoarthritis. The company is based in Durham, N.C.