Business Wire03.29.18
Zimmer Biomet Holdings Inc. announced full seven-year follow-up data from a study evaluating the company’s Mobi-C Cervical Disc for total disc replacement, an option for cervical total disc replacement (cTDR) that is designed to maintain patient range of motion post-surgery.
The prospective, randomized clinical trial of cTDR with Mobi-C compared study subjects to the previous standard of care, anterior cervical discectomy and fusion (ACDF), and follows nearly 600 patients through seven years. Researchers found that cTDR with Mobi-C was associated with lower pain scores, maintained range of motion, less adjacent level degeneration and adjacent level subsequent surgery, as well as a lower rate of secondary surgery compared to ACDF.
“This study further validates the strengths of Mobi-C as a solution for cervical total disc replacement. The positive clinical outcomes remain consistent with previous findings, mainly that Mobi-C continues to deliver positive patient outcomes including efficacy, safety, patient satisfaction and fewer follow-up surgeries,” said Joe Ross, general manager of Zimmer Biomet’s Spine division. “It is particularly important that the clinical effectiveness of cTDR versus ACDF becomes more apparent when treatment increases from one to two levels, indicating a significant benefit for Mobi-C in two-level procedures, which affects an estimated one in three cervical disc patients.”
The Investigational Device Exemption/Post-Approval study evaluated 599 patients, including 164 treated with one-level cTDR, 225 treated with two-level cTDR, 81 treated with one-level ACDF and 105 treated with two-level ACDF. At seven years, follow-up rates ranged from 73.5 percent to 84.4 percent (overall 80.2 percent). Overall success rates for two-level patients were 60.8 percent for cTDR and 34.6 percent for ACDF. Overall success rates for one-level patients were 55.2 percent for cTDR and 50.0 percent for ACDF. Researchers concluded that the composite success analysis demonstrated clinical superiority of two-level cTDR over ACDF and non-inferiority of one-level cTDR versus ACDF. For two-level procedures, the Neck Disability Index (NDI) success rate was significantly greater in the cTDR group (79.0 percent) compared to ACDF (58.0 percent), marking significant patient-reported improvements with cTDR. In addition, overall, more than 86.0 percent of patients who received cTDR (two-level) and 73.9 percent of patients who underwent ACDF (two-level) reported they were “very satisfied” after seven years. Finally, the rate of subsequent surgery at the index level was significantly lower in the two-level cTDR group compared to the ACDF group (cTDR: 4.4 percent vs. ACDF:16.2 percent; p=0.001); and similarly, the rate of adjacent level secondary surgery was significantly lower in the two-level cTDR (4.4 percent) patients compared to the ACDF (11.4 percent; p=0.03) patients. The clinical data for the seven-year results and associated product labeling updates are currently under review with the U.S. Food and Drug Administration (FDA).
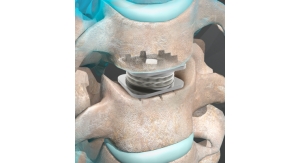
Mobi-C is the first cervical disc prosthesis approved by the FDA for reconstruction of a cervical disc at both one and two levels (C3-C7). Mobi-C is a cobalt chromium alloy and polyethylene mobile-bearing prosthesis that is inserted in a single step, without requiring bone chiseling to accommodate vertebral anchorage such as screws or keels. The Mobi-C Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc from C3-C7 following discectomy at one level or two contiguous levels for intractable radiculopathy (arm pain and/or neurological deficit) with or without neck pain or myelopathy due to abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (CT, MRI or X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes) and/or visible loss of disc height compared to adjacent levels. The Mobi-C Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least six weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C Cervical Disc Prosthesis.
Founded in 1927 and headquartered in Warsaw, Ind., Zimmer Biomet designs, manufactures and markets orthopedic reconstructive products; sports medicine, biologics, extremities and trauma products; office based technologies; spine, craniomaxillofacial and thoracic products; dental implants; and related surgical products. Its products and solutions help treat patients suffering from disorders of, or injuries to, bones, joints or supporting soft tissues. The company has operations in more than 25 countries around the world and sell products in more than 100 countries.
The prospective, randomized clinical trial of cTDR with Mobi-C compared study subjects to the previous standard of care, anterior cervical discectomy and fusion (ACDF), and follows nearly 600 patients through seven years. Researchers found that cTDR with Mobi-C was associated with lower pain scores, maintained range of motion, less adjacent level degeneration and adjacent level subsequent surgery, as well as a lower rate of secondary surgery compared to ACDF.
“This study further validates the strengths of Mobi-C as a solution for cervical total disc replacement. The positive clinical outcomes remain consistent with previous findings, mainly that Mobi-C continues to deliver positive patient outcomes including efficacy, safety, patient satisfaction and fewer follow-up surgeries,” said Joe Ross, general manager of Zimmer Biomet’s Spine division. “It is particularly important that the clinical effectiveness of cTDR versus ACDF becomes more apparent when treatment increases from one to two levels, indicating a significant benefit for Mobi-C in two-level procedures, which affects an estimated one in three cervical disc patients.”
The Investigational Device Exemption/Post-Approval study evaluated 599 patients, including 164 treated with one-level cTDR, 225 treated with two-level cTDR, 81 treated with one-level ACDF and 105 treated with two-level ACDF. At seven years, follow-up rates ranged from 73.5 percent to 84.4 percent (overall 80.2 percent). Overall success rates for two-level patients were 60.8 percent for cTDR and 34.6 percent for ACDF. Overall success rates for one-level patients were 55.2 percent for cTDR and 50.0 percent for ACDF. Researchers concluded that the composite success analysis demonstrated clinical superiority of two-level cTDR over ACDF and non-inferiority of one-level cTDR versus ACDF. For two-level procedures, the Neck Disability Index (NDI) success rate was significantly greater in the cTDR group (79.0 percent) compared to ACDF (58.0 percent), marking significant patient-reported improvements with cTDR. In addition, overall, more than 86.0 percent of patients who received cTDR (two-level) and 73.9 percent of patients who underwent ACDF (two-level) reported they were “very satisfied” after seven years. Finally, the rate of subsequent surgery at the index level was significantly lower in the two-level cTDR group compared to the ACDF group (cTDR: 4.4 percent vs. ACDF:16.2 percent; p=0.001); and similarly, the rate of adjacent level secondary surgery was significantly lower in the two-level cTDR (4.4 percent) patients compared to the ACDF (11.4 percent; p=0.03) patients. The clinical data for the seven-year results and associated product labeling updates are currently under review with the U.S. Food and Drug Administration (FDA).
| Mobi-C | ACDF | ||
| 575 patients included in the 7-year analysis: | |||
| Two-level | 225 | 105 | |
| One-level | 164 | 81 | |
| Overall Success Rate: | |||
| Two-level | 60.8% | 34.6% |
Clinical Superiority of Mobi-C versus ACDF (p<0.0001) |
| One-level | 55.2% | 50.0% |
Non-inferiority of Mobi-C versus ACDF (p>0.05) |
| Two-level Neck Disability Index (NDI) success rate | |||
| Two-level | 79.0% | 58.0% | p<0.05 |
| One-level | 76.5% | 77.8% | p=NS |
| Patients (from both groups) reporting “very satisfied” with the procedure | |||
| Two-level | 86.0% | 73.9% | p=0.039 |
| One-level | 90.9% | 77.8% | p=0.028 |
| Rate of secondary surgery at the index level | |||
| Two-level | 4.4% | 16.2% | p=0.001 |
| One-level | 3.0% | 12.3% | p=0.008 |
| Rate of secondary surgery at an adjacent level | |||
| Two-level | 4.4% | 11.4% | p=0.03 |
| One-level | 3.7% | 13.6% | p=0.007 |
Mobi-C is the first cervical disc prosthesis approved by the FDA for reconstruction of a cervical disc at both one and two levels (C3-C7). Mobi-C is a cobalt chromium alloy and polyethylene mobile-bearing prosthesis that is inserted in a single step, without requiring bone chiseling to accommodate vertebral anchorage such as screws or keels. The Mobi-C Cervical Disc Prosthesis is indicated in skeletally mature patients for reconstruction of the disc from C3-C7 following discectomy at one level or two contiguous levels for intractable radiculopathy (arm pain and/or neurological deficit) with or without neck pain or myelopathy due to abnormality localized to the level of the disc space and at least one of the following conditions confirmed by radiographic imaging (CT, MRI or X-rays): herniated nucleus pulposus, spondylosis (defined by the presence of osteophytes) and/or visible loss of disc height compared to adjacent levels. The Mobi-C Cervical Disc Prosthesis is implanted using an anterior approach. Patients should have failed at least six weeks of conservative treatment or demonstrated progressive signs or symptoms despite nonoperative treatment prior to implantation of the Mobi-C Cervical Disc Prosthesis.
Founded in 1927 and headquartered in Warsaw, Ind., Zimmer Biomet designs, manufactures and markets orthopedic reconstructive products; sports medicine, biologics, extremities and trauma products; office based technologies; spine, craniomaxillofacial and thoracic products; dental implants; and related surgical products. Its products and solutions help treat patients suffering from disorders of, or injuries to, bones, joints or supporting soft tissues. The company has operations in more than 25 countries around the world and sell products in more than 100 countries.