PR Newswire04.02.18
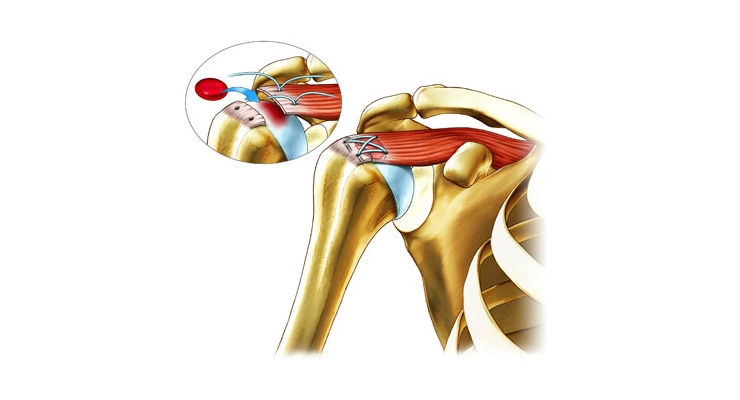
Ortho Regenerative Technologies Inc., an emerging orthopedic and sports medicine technology company, announced positive preliminary MR imaging outcomes data from a study examining its Ortho-R technology in the biologic repair of rotator cuff injuries.
Conducted by researchers from Montreal's prestigious École Polytechnique, the pilot dose escalation study used MR imaging outcomes, read by two blinded experts at 3 intervals, to compare the results of Ortho-R versus standard of care in a non-clinical rotator cuff injury model in sheep. In this study, treatment with Ortho-R was shown to significantly improve healing, as measured by gap reduction between the tendon and bone, versus standard of care, with the highest Ortho-R dose having the greatest effect. Detailed results from the study will be submitted to a future medical conference and for publication.
"We are very encouraged by this data, as Ortho-R demonstrated an ability to induce healing in as little as three months," said Ortho RTi's Executive Chairman and CEO, Dr. Brent Norton. "With a small study size, getting meaningful data is challenging, yet the relative gap reduction induced by Ortho-R was statistically significant (p=0.01). With more than 500,000 rotator cuff surgeries being performed in the United States each year, and with that number continuing to grow, we are looking forward to taking this technology into the clinic as soon as possible. To that end, we are beginning to make preparations to submit an Investigational New Drug Application to the FDA for this, our lead indication. As always, we look forward to updating our stakeholders as we progress."
Conducted by researchers from Montreal's prestigious École Polytechnique, the pilot dose escalation study used MR imaging outcomes, read by two blinded experts at 3 intervals, to compare the results of Ortho-R versus standard of care in a non-clinical rotator cuff injury model in sheep. In this study, treatment with Ortho-R was shown to significantly improve healing, as measured by gap reduction between the tendon and bone, versus standard of care, with the highest Ortho-R dose having the greatest effect. Detailed results from the study will be submitted to a future medical conference and for publication.
"We are very encouraged by this data, as Ortho-R demonstrated an ability to induce healing in as little as three months," said Ortho RTi's Executive Chairman and CEO, Dr. Brent Norton. "With a small study size, getting meaningful data is challenging, yet the relative gap reduction induced by Ortho-R was statistically significant (p=0.01). With more than 500,000 rotator cuff surgeries being performed in the United States each year, and with that number continuing to grow, we are looking forward to taking this technology into the clinic as soon as possible. To that end, we are beginning to make preparations to submit an Investigational New Drug Application to the FDA for this, our lead indication. As always, we look forward to updating our stakeholders as we progress."