MTF Biologics and the University of Missouri04.11.18
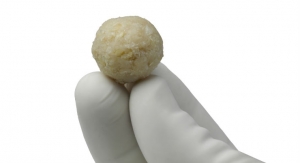
A unique system that has potential to improve outcomes from joint repair procedures continues to show positive clinical results. The Missouri Osteochondral Preservation System (MOPSSM) is a technology that more than doubles the storage life of allografts used for joint repairs. Created by the University of Missouri and available exclusively with tissue processed by MTF Biologics, MOPSSM allografts have been shown to maintain more than twice the viable chondrocyte density for twice as long as standard of care (SOC) allografts.
“Not only does doubling the viable cell density and extending the life of an osteochondral allograft increase the time for a surgeon to schedule and perform the implantation, it has the potential to have a positive impact on clinical outcomes,” said Marc Long, Ph.D., vice president of Research & Development at MTF Biologics. “MTF Biologics works hand in hand with clinicians and researchers like those at the University of Missouri to stay on the forefront of science and clinical research. We are honored to provide tissue for a remarkable innovation like MOPSSM as we continue to provide new clinical solutions for surgeons and their patients.”
A study, “Chondrocyte Viability at Time of Transplantation for Osteochondral Allografts Preserved by the Missouri Osteochondral Preservation System versus Standard Tissue Bank Protocol,” published in The Journal of Knee Surgery in October 2017 showed that MOPSSM allografts maintained 98.8 percent viable cell density at 40 to 55 days, compared to SOC allografts, which maintained only 22.9 percent viable cell density at 21 to 28 days after procurement. Importantly, all MOPSSM allografts evaluated in the study had higher than the desired minimum essential viability level of 70 percent, while only 27 percent of SOC grafts evaluated exceeded the desired level at the time of implantation. MOPSSM allografts also had a revision rate of only 2 percent, while the SOC allograft revision rate was 11.5 percent. Additionally, MOPSSM allografts had no early failures (six-24 months, mean = 12 months postop), compared to SOC allografts, which demonstrated an early failure rate of 3.8 percent in this study.
“We are very pleased with the overwhelmingly positive early results with the use of MOPSSM tissues and believe the data support the benefits of using grafts with high chondrocyte viability to optimize outcomes in patients,” said James L. Cook, DVM, Ph.D. “The partnership with MTF Biologics has allowed us to safely bring this technology to clinical use to help patients, surgeons, and donor families.”
The quality of tissue preserved with MOPSSM is important to its viability. MTF Biologics uses a unique aseptic processing method. This method ensures optimal tissue quality and helps to maintain the biomechanical integrity and biochemistry of every graft.
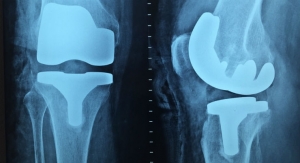
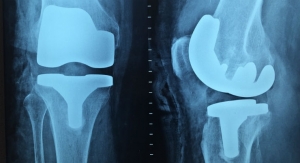
“Although I can reconstruct most of a knee joint, the biggest challenge is replacing the protective lining on the ends of the femur and tibia,” Stannard said. “This lining, called articular cartilage, is a thousand times slicker than wet ice, and it protects the joint during everyday activities.”
When articular cartilage is damaged from disease or injury, the bones it protects eventually become damaged. This condition, and the pain associated with it, is known as arthritis.
“We can use osteochondral allografts from a donor to fill in the damaged areas, rather than remove bone, and implant metal and plastic components,” said James Stannard, M.D. “However, current preservation methods for the allografts themselves limit the number we have available to us.”
With the current preservation method, only 20 percent of donated allografts are used. To overcome this challenge, MU’s Orthopaedic Research Team developed their preservation technology. With the new system developed at MU, the grafts’ shelf life can be extended to at least 60 days, with 90 percent or more of cells remaining viable.
“So, by increasing quality of the grafts for a much longer period of time, we also increase the quantity of grafts that will be available to help patients,” Cook said.
He is hopeful that they will be able to begin using the technology for Missouri Orthopaedic Institute patients this year. Research studies about the preservation system have been published in academic journals, including the Journal of Knee Surgery in 2012 and Clinical Orthopaedics and Related Research, a publication of the Association of Bone and Joint Surgeons, in 2014. Cook said the technology is approved by the Food and Drug Administration and has provisional patents in the United States and several other countries. It is in the final stage of pre-clinical evaluation at the Comparative Orthopaedic Laboratory in preparation for clinical use at the Missouri Orthopaedic Institute.
MTF Biologics, also known as the Musculoskeletal Transplant Foundation, is a nonprofit organization based in Edison, N.J. It is a consortium comprised of leading organ procurement organizations, tissue recovery organizations and academic medical institutions, and governed by a board of surgeons who are leading experts in tissue transplantation. As the world’s largest tissue bank, MTF Biologics saves and heals lives by honoring donated gifts, serving patients and advancing science. Since its inception in 1987, the organization has received tissue from more than 125,000 donors and distributed more than 8 million grafts for transplantation. Through its IIAM subsidiary, it has placed more than 55,000 non-transplantable organs for research. Through its Statline subsidiary, it has managed more than 10 million donor referrals.
“Not only does doubling the viable cell density and extending the life of an osteochondral allograft increase the time for a surgeon to schedule and perform the implantation, it has the potential to have a positive impact on clinical outcomes,” said Marc Long, Ph.D., vice president of Research & Development at MTF Biologics. “MTF Biologics works hand in hand with clinicians and researchers like those at the University of Missouri to stay on the forefront of science and clinical research. We are honored to provide tissue for a remarkable innovation like MOPSSM as we continue to provide new clinical solutions for surgeons and their patients.”
A study, “Chondrocyte Viability at Time of Transplantation for Osteochondral Allografts Preserved by the Missouri Osteochondral Preservation System versus Standard Tissue Bank Protocol,” published in The Journal of Knee Surgery in October 2017 showed that MOPSSM allografts maintained 98.8 percent viable cell density at 40 to 55 days, compared to SOC allografts, which maintained only 22.9 percent viable cell density at 21 to 28 days after procurement. Importantly, all MOPSSM allografts evaluated in the study had higher than the desired minimum essential viability level of 70 percent, while only 27 percent of SOC grafts evaluated exceeded the desired level at the time of implantation. MOPSSM allografts also had a revision rate of only 2 percent, while the SOC allograft revision rate was 11.5 percent. Additionally, MOPSSM allografts had no early failures (six-24 months, mean = 12 months postop), compared to SOC allografts, which demonstrated an early failure rate of 3.8 percent in this study.
“We are very pleased with the overwhelmingly positive early results with the use of MOPSSM tissues and believe the data support the benefits of using grafts with high chondrocyte viability to optimize outcomes in patients,” said James L. Cook, DVM, Ph.D. “The partnership with MTF Biologics has allowed us to safely bring this technology to clinical use to help patients, surgeons, and donor families.”
The quality of tissue preserved with MOPSSM is important to its viability. MTF Biologics uses a unique aseptic processing method. This method ensures optimal tissue quality and helps to maintain the biomechanical integrity and biochemistry of every graft.
“Although I can reconstruct most of a knee joint, the biggest challenge is replacing the protective lining on the ends of the femur and tibia,” Stannard said. “This lining, called articular cartilage, is a thousand times slicker than wet ice, and it protects the joint during everyday activities.”
When articular cartilage is damaged from disease or injury, the bones it protects eventually become damaged. This condition, and the pain associated with it, is known as arthritis.
“We can use osteochondral allografts from a donor to fill in the damaged areas, rather than remove bone, and implant metal and plastic components,” said James Stannard, M.D. “However, current preservation methods for the allografts themselves limit the number we have available to us.”
With the current preservation method, only 20 percent of donated allografts are used. To overcome this challenge, MU’s Orthopaedic Research Team developed their preservation technology. With the new system developed at MU, the grafts’ shelf life can be extended to at least 60 days, with 90 percent or more of cells remaining viable.
“So, by increasing quality of the grafts for a much longer period of time, we also increase the quantity of grafts that will be available to help patients,” Cook said.
He is hopeful that they will be able to begin using the technology for Missouri Orthopaedic Institute patients this year. Research studies about the preservation system have been published in academic journals, including the Journal of Knee Surgery in 2012 and Clinical Orthopaedics and Related Research, a publication of the Association of Bone and Joint Surgeons, in 2014. Cook said the technology is approved by the Food and Drug Administration and has provisional patents in the United States and several other countries. It is in the final stage of pre-clinical evaluation at the Comparative Orthopaedic Laboratory in preparation for clinical use at the Missouri Orthopaedic Institute.
MTF Biologics, also known as the Musculoskeletal Transplant Foundation, is a nonprofit organization based in Edison, N.J. It is a consortium comprised of leading organ procurement organizations, tissue recovery organizations and academic medical institutions, and governed by a board of surgeons who are leading experts in tissue transplantation. As the world’s largest tissue bank, MTF Biologics saves and heals lives by honoring donated gifts, serving patients and advancing science. Since its inception in 1987, the organization has received tissue from more than 125,000 donors and distributed more than 8 million grafts for transplantation. Through its IIAM subsidiary, it has placed more than 55,000 non-transplantable organs for research. Through its Statline subsidiary, it has managed more than 10 million donor referrals.