Business Wire05.07.18
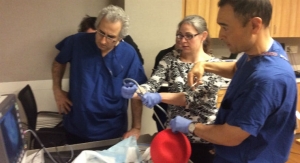
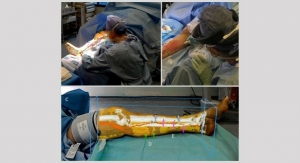
SpineGuard, a company that develops and markets instruments designed to secure the placement of surgical implants by bringing real-time digital technology into the operating room, announced it has been granted a second U.S. patent for the application of its Dynamic Surgical Guidance technology for a new application: bone quality measurement, in this particular patent with a focus on monitoring the consolidation of the bony fusion bed.
Stéphane Bette, CEO and co-founder of SpineGuard, said, “The success of skeletal fixation procedures is based on obtaining a solid bony fusion providing long-term stability of the repair, thanks to the implants that are inserted internally to hold the bone structures in place while they fuse. We believe that the DSG technology can allow real-time in-situ monitoring of the progress of the bony fusion. This new patent granted in the U.S. is one more illustration that our core technology is a platform that has multiple applications; our company is constantly innovating and working at providing valuable digital information about the health status of patients.”
“The potential applications of in-situ monitoring of the bony fusion are multiple in the post-operative phase: from the adaptation of the physical therapy, the adjustment of the stiffness of braces, to the connection with the implants themselves to make them smart and adapting to the bone healing status.” concluded Maurice Bourlion, Ph.D., co-inventor of the DSGt technology and director of SpineGuard.
Founded in 2009 in France and the United States by Pierre Jérôme and Bette, SpineGuard’s mission is to make spine surgery safer by bringing real-time digital technology into the operating room. Its primary objective is to establish its proprietary DSG (Dynamic Surgical Guidance) technology as the global standard of surgical care, starting with safer screw placement in spine surgery and then in other surgeries. PediGuard, the first device designed using DSG, was co-invented by Bourlion; Ciaran Bolger, M.D., Ph.D.; and Alain Vanquaethem, biomedical engineer. It is the world’s first and only handheld device capable of alerting surgeons to potential pedicular or vertebral breaches. Over 60,000 surgical procedures have been performed worldwide with DSG enabled devices. Numerous studies published in peer-reviewed medical and scientific journals have demonstrated the multiple benefits that PediGuard delivers to patients, surgical staff and hospitals. SpineGuard is expanding the scope of its DSG platform through strategic partnerships with medical device companies and the development of smart instruments and implants. SpineGuard has offices in San Francisco, Calif.; and Paris, France.
Stéphane Bette, CEO and co-founder of SpineGuard, said, “The success of skeletal fixation procedures is based on obtaining a solid bony fusion providing long-term stability of the repair, thanks to the implants that are inserted internally to hold the bone structures in place while they fuse. We believe that the DSG technology can allow real-time in-situ monitoring of the progress of the bony fusion. This new patent granted in the U.S. is one more illustration that our core technology is a platform that has multiple applications; our company is constantly innovating and working at providing valuable digital information about the health status of patients.”
“The potential applications of in-situ monitoring of the bony fusion are multiple in the post-operative phase: from the adaptation of the physical therapy, the adjustment of the stiffness of braces, to the connection with the implants themselves to make them smart and adapting to the bone healing status.” concluded Maurice Bourlion, Ph.D., co-inventor of the DSGt technology and director of SpineGuard.
Founded in 2009 in France and the United States by Pierre Jérôme and Bette, SpineGuard’s mission is to make spine surgery safer by bringing real-time digital technology into the operating room. Its primary objective is to establish its proprietary DSG (Dynamic Surgical Guidance) technology as the global standard of surgical care, starting with safer screw placement in spine surgery and then in other surgeries. PediGuard, the first device designed using DSG, was co-invented by Bourlion; Ciaran Bolger, M.D., Ph.D.; and Alain Vanquaethem, biomedical engineer. It is the world’s first and only handheld device capable of alerting surgeons to potential pedicular or vertebral breaches. Over 60,000 surgical procedures have been performed worldwide with DSG enabled devices. Numerous studies published in peer-reviewed medical and scientific journals have demonstrated the multiple benefits that PediGuard delivers to patients, surgical staff and hospitals. SpineGuard is expanding the scope of its DSG platform through strategic partnerships with medical device companies and the development of smart instruments and implants. SpineGuard has offices in San Francisco, Calif.; and Paris, France.