PR Newswire08.09.18
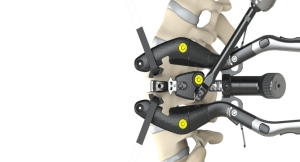
NuVasive Inc. and Siemens Healthineers have announced a strategic partnership focused on technology development, marketing, and commercial activities to advance clinical outcomes in minimally invasive spine surgery. NuVasive is an innovation leader in spine health technology, focused on transforming spine surgery with minimally disruptive, predictable and clinically reproducible procedurally-integrated solutions, while Siemens Healthineers offers surgeons a broad portfolio of imaging systems including 3D imaging for complex spine cases.
In their Spine Precision Partnership, the companies will join forces in advancing their proprietary technologies with the objective of creating solutions which improve operating room (OR) workflow efficiency and provide increased precision in the delivery of minimally disruptive spine surgery technologies. The first step in the Spine Precision Partnership is to integrate NuVasive's Pulse surgical automation platform with Siemens Healthineers' Cios Spin* cutting-edge mobile 3D imaging for intra-operative quality assurance.
"Currently, a number of hospitals and healthcare systems treat patients undergoing spine surgery through often cost-intensive, intra-operative CT scans with a general navigation system with limited utilization in spine surgery cases. The combination of NuVasive's Pulse system and the Siemens Healthineers' next-generation advanced imaging technology provides a compelling offering for hospitals and surgeons who require a scalable, cost-efficient technology that maximizes OR workflow efficiency and significantly improves visualization for spine surgery," said Gregory T. Lucier, chairman and chief executive officer of NuVasive. "We firmly believe that this combined offering will enable advanced clinical benefits driving superior patient outcomes, delivered through greatly improved visualization of the anatomy promoting successful anatomical access and spinal implant placement."
"We at Siemens Healthineers are excited to work with NuVasive to develop intra-operative 3D-imaging and navigation tools for our advanced imaging systems that empower spine surgeons and neurosurgeons to be more precise, faster and cost efficient in the operating room. Increased workflow efficiency, better image quality, as well as predictable and reproducible results, will transform care delivery and set a new standard in spine surgery," said Peter Seitz, head of Surgery at Siemens Healthineers.
NuVasive and Siemens Healthineers will feature the Spine Precision Partnership at the North American Spine Society 2018 Annual Meeting held September 26-29, 2018 in Los Angeles, California.
* Cios Spin from Siemens Healthineers is pending 510(k) clearance, and is not yet commercially available in the United States.
In their Spine Precision Partnership, the companies will join forces in advancing their proprietary technologies with the objective of creating solutions which improve operating room (OR) workflow efficiency and provide increased precision in the delivery of minimally disruptive spine surgery technologies. The first step in the Spine Precision Partnership is to integrate NuVasive's Pulse surgical automation platform with Siemens Healthineers' Cios Spin* cutting-edge mobile 3D imaging for intra-operative quality assurance.
"Currently, a number of hospitals and healthcare systems treat patients undergoing spine surgery through often cost-intensive, intra-operative CT scans with a general navigation system with limited utilization in spine surgery cases. The combination of NuVasive's Pulse system and the Siemens Healthineers' next-generation advanced imaging technology provides a compelling offering for hospitals and surgeons who require a scalable, cost-efficient technology that maximizes OR workflow efficiency and significantly improves visualization for spine surgery," said Gregory T. Lucier, chairman and chief executive officer of NuVasive. "We firmly believe that this combined offering will enable advanced clinical benefits driving superior patient outcomes, delivered through greatly improved visualization of the anatomy promoting successful anatomical access and spinal implant placement."
"We at Siemens Healthineers are excited to work with NuVasive to develop intra-operative 3D-imaging and navigation tools for our advanced imaging systems that empower spine surgeons and neurosurgeons to be more precise, faster and cost efficient in the operating room. Increased workflow efficiency, better image quality, as well as predictable and reproducible results, will transform care delivery and set a new standard in spine surgery," said Peter Seitz, head of Surgery at Siemens Healthineers.
NuVasive and Siemens Healthineers will feature the Spine Precision Partnership at the North American Spine Society 2018 Annual Meeting held September 26-29, 2018 in Los Angeles, California.
* Cios Spin from Siemens Healthineers is pending 510(k) clearance, and is not yet commercially available in the United States.