Globe Newswire09.20.18
Simplify Medical Pty. Ltd., maker of the Simplify cervical artificial disc, announced that an abstract for preliminary data from its one-level Investigational Device Exemption (IDE) clinical trial has been selected as an Innovative Technology Presentation during the 2018 North American Spine Society (NASS) Annual Meeting, being held next week in Los Angeles, Calif. Richard Guyer, M.D., co-principal investigator for the study, will present the data at the session, and it will also be featured in a poster presentation.
David Hovda, CEO of Simplify Medical, commented, “We look forward to sharing initial results from a key site of our multi-center, prospective pivotal U.S. trial evaluating the Simplify Disc at one-level, for which we completed enrollment in February of this year. Our PEEK on ceramic Simplify Disc is engineered for durability, enhanced imaging with MRI, and with lower height options to better match patient anatomies. We are encouraged by our early data. If FDA-approved, we believe the Simplify Disc will offer patients and physicians in the U.S. a meaningful alternative to cervical fusion surgery and other currently available discs.”
The oral presentation will take place on Sept. 26 from 1:21 p.m. to 1:28 p.m. in Room 404AB. Presenting authors are Guyer and Donna D. Ohnmeiss, Ph.D.
In addition, during NASS, the Simplify Cervical Artificial Disc will be awarded as a winner in the 10th Annual 2018 Spine Technology Awards. The awards recognize the best new spine technologies, engineering teams and inventors for 2018 and, in the process, reward excellence in innovation.
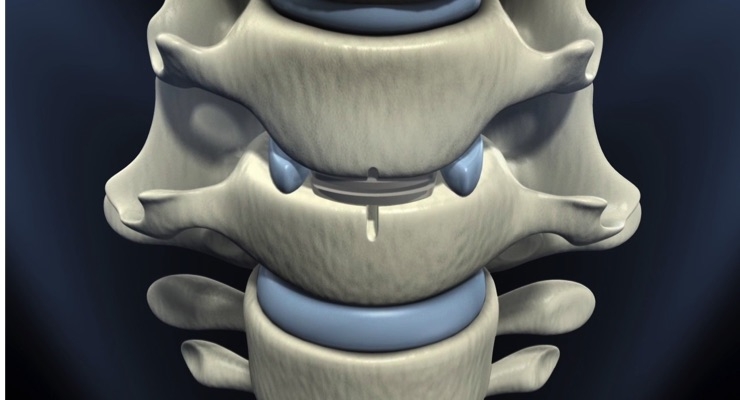
The Simplify Disc is a motion preserving cervical artificial disc designed to allow for advanced imaging capability of magnetic resonance imaging (MRI), and better match patients’ anatomies and kinematics. It is composed of advanced, primarily non-metal materials (PEEK-on-ceramic) to permit the full diagnostic imaging capability of MRI and may eliminate the need for computed tomography (CT)/Myelogram and CT imaging in order to minimize patient exposure to radiation. The Simplify Disc is anatomically designed, offering a broader range of disc heights including low height implant options to better fit patients’ anatomies. With no metal in its articulating components, the disc is also designed for low levels of wear to optimize long-term durability. Implantation of the Simplify Disc is accomplished in a straightforward, three-step procedure. The Simplify Disc is an investigational device in the United States and is limited by law to investigational use.
Simplify Medical is focused on cervical spinal disc arthroplasty, using MRI-compatible materials designed to decrease the need for ionizing radiation and enhance patient options. Simplify Medical is located in Sunnyvale, Calif.
David Hovda, CEO of Simplify Medical, commented, “We look forward to sharing initial results from a key site of our multi-center, prospective pivotal U.S. trial evaluating the Simplify Disc at one-level, for which we completed enrollment in February of this year. Our PEEK on ceramic Simplify Disc is engineered for durability, enhanced imaging with MRI, and with lower height options to better match patient anatomies. We are encouraged by our early data. If FDA-approved, we believe the Simplify Disc will offer patients and physicians in the U.S. a meaningful alternative to cervical fusion surgery and other currently available discs.”
The oral presentation will take place on Sept. 26 from 1:21 p.m. to 1:28 p.m. in Room 404AB. Presenting authors are Guyer and Donna D. Ohnmeiss, Ph.D.
In addition, during NASS, the Simplify Cervical Artificial Disc will be awarded as a winner in the 10th Annual 2018 Spine Technology Awards. The awards recognize the best new spine technologies, engineering teams and inventors for 2018 and, in the process, reward excellence in innovation.
The Simplify Disc is a motion preserving cervical artificial disc designed to allow for advanced imaging capability of magnetic resonance imaging (MRI), and better match patients’ anatomies and kinematics. It is composed of advanced, primarily non-metal materials (PEEK-on-ceramic) to permit the full diagnostic imaging capability of MRI and may eliminate the need for computed tomography (CT)/Myelogram and CT imaging in order to minimize patient exposure to radiation. The Simplify Disc is anatomically designed, offering a broader range of disc heights including low height implant options to better fit patients’ anatomies. With no metal in its articulating components, the disc is also designed for low levels of wear to optimize long-term durability. Implantation of the Simplify Disc is accomplished in a straightforward, three-step procedure. The Simplify Disc is an investigational device in the United States and is limited by law to investigational use.
Simplify Medical is focused on cervical spinal disc arthroplasty, using MRI-compatible materials designed to decrease the need for ionizing radiation and enhance patient options. Simplify Medical is located in Sunnyvale, Calif.