PR Newswire09.25.18
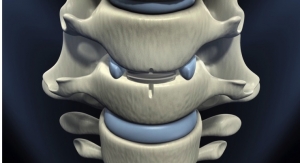
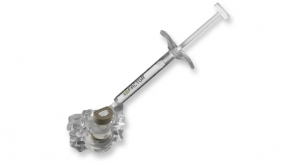
Paradigm Spine LLC, a provider of motion preservation solutions for the treatment of lumbar spinal stenosis, announced its company activities and scientific podium presentation on coflex Interlaminar Stabilization at this week's 2018 North American Spine Society (NASS) Annual Meeting, held in Los Angeles, Calif. coflex is the only posterior lumbar motion preservation solution with proven long-term outcomes for durable pain relief and stability for patients with moderate to severe spinal stenosis.
"The annual NASS meeting provides an optimal opportunity to directly engage with our U.S. spine surgeon audience and we look forward to demonstrating—on the podium and through our surgeon reception—the substantial results adding up from both our newly published ESCADA study and long-term findings from the U.S. Food and Drug Administration IDE trial supporting coflex against decompression alone and against fusion, respectively," said Marc Viscogliosi, chairman and CEO of Paradigm Spine.
Details of Paradigm Spine's activities follow:
Oral Presentation: Interlaminar Stabilization versus PLF in the ≤ 65 Year-Old Patient: Five-Year Follow-Up
Abstract: # 91
Session: Preserving Spinal Motion; Session moderated by Richard D. Guyer, M.D.
Date: Sept. 26 at 4:41 p.m.-4:47 p.m.
Location: Room 403A
Presenting Author: Antonio T. Brecevich, M.D.
Institutions: Hospital for Special Surgery (HSS), New York, N.Y.
Product: Coflex (approved for this indication)
Paradigm Spine Sponsored Surgeon Reception
Date: Sept. 27 at 5:30 p.m.-8 p.m.
Location: GRAMMY Museum at L.A. Live
800 West Olympic Boulevard, Los Angeles, Calif.
Paradigm Spine is showcasing coflex Interlaminar Stabilization and sharing the newly-published ESCADA results at booth No. 1429 in the Exhibit Hall.
Lumbar spinal stenosis (LSS), affecting 1.6 million patients annually in the United States, is a debilitating and degenerative disease often associated with significant leg and back pain, leg numbness and weakness, and significant reduction in an active lifestyle. Historically, the two traditional surgical treatment options for LSS included decompression alone or decompression with lumbar fusion. Decompression alone has proven effective at relieving pain symptoms caused by lumbar spinal stenosis, however, patients may not experience long term symptomatic relief, resulting in subsequent epidural injections for pain management, or additional surgeries for conversion to a fusion. Decompression with fusion has proven to provide pain relief and stabilize the diseased segment, but may lead to adjacent level disease requiring subsequent surgeries.
Paradigm Spine LLC, founded in 2004, is a privately held company that designs and develops solutions to manage spinal stenosis. The company's signature product is the coflex Interlaminar Stabilization device, which is currently used in over 60 countries worldwide. coflex is the only lumbar spinal device that has produced Level I evidence in two separate prospective, randomized, controlled studies against two different control groups, changing the standard of care for lumbar spinal stenosis treatment.
"The annual NASS meeting provides an optimal opportunity to directly engage with our U.S. spine surgeon audience and we look forward to demonstrating—on the podium and through our surgeon reception—the substantial results adding up from both our newly published ESCADA study and long-term findings from the U.S. Food and Drug Administration IDE trial supporting coflex against decompression alone and against fusion, respectively," said Marc Viscogliosi, chairman and CEO of Paradigm Spine.
Details of Paradigm Spine's activities follow:
Oral Presentation: Interlaminar Stabilization versus PLF in the ≤ 65 Year-Old Patient: Five-Year Follow-Up
Abstract: # 91
Session: Preserving Spinal Motion; Session moderated by Richard D. Guyer, M.D.
Date: Sept. 26 at 4:41 p.m.-4:47 p.m.
Location: Room 403A
Presenting Author: Antonio T. Brecevich, M.D.
Institutions: Hospital for Special Surgery (HSS), New York, N.Y.
Product: Coflex (approved for this indication)
Paradigm Spine Sponsored Surgeon Reception
Date: Sept. 27 at 5:30 p.m.-8 p.m.
Location: GRAMMY Museum at L.A. Live
800 West Olympic Boulevard, Los Angeles, Calif.
Paradigm Spine is showcasing coflex Interlaminar Stabilization and sharing the newly-published ESCADA results at booth No. 1429 in the Exhibit Hall.
Lumbar spinal stenosis (LSS), affecting 1.6 million patients annually in the United States, is a debilitating and degenerative disease often associated with significant leg and back pain, leg numbness and weakness, and significant reduction in an active lifestyle. Historically, the two traditional surgical treatment options for LSS included decompression alone or decompression with lumbar fusion. Decompression alone has proven effective at relieving pain symptoms caused by lumbar spinal stenosis, however, patients may not experience long term symptomatic relief, resulting in subsequent epidural injections for pain management, or additional surgeries for conversion to a fusion. Decompression with fusion has proven to provide pain relief and stabilize the diseased segment, but may lead to adjacent level disease requiring subsequent surgeries.
Paradigm Spine LLC, founded in 2004, is a privately held company that designs and develops solutions to manage spinal stenosis. The company's signature product is the coflex Interlaminar Stabilization device, which is currently used in over 60 countries worldwide. coflex is the only lumbar spinal device that has produced Level I evidence in two separate prospective, randomized, controlled studies against two different control groups, changing the standard of care for lumbar spinal stenosis treatment.