Business Wire01.08.19
SpinalCyte LLC, a regenerative medicine company focused on regrowth of the spinal disc nucleus using its universal donor product, CybroCell, announced that a single injection of modified human dermal fibroblasts (HDFs), resulted in significant improvements of disc height and pain reduction 12 months after injection of the cell therapy for patients with degenerative disc disease (DDD).
The trial assessed patients using the Oswestry Disability Index (ODI), Visual Analogue Scale (VAS) and disc height via MRI scans. Using composite scoring, 54 percent of the treatment arm patients met all three endpoints as compared to only 17 percent with the placebo (p=0.0003). Over 84 percent of patients in the treatment group of CybroCell or CybroCell with platelet rich plasma (PRP) injections had an increase or no change in disc height compared to 25 percent for the placebo saline injection.
Analyzed individually, 83 percent of spinal discs that were injected with CybroCell had an increase or no change in disc height compared to 25 percent of saline injected discs. The 12-month MRI data from the landmark Phase 1/Phase 2 trial were analyzed for 17 patients by an independent radiological expert.
“Verification of structural improvements via MRI at six and 12 months strongly suggests that we are inducing a biological cascade of events associated with the induction of a regenerative process in the patient,” said Jesus Perez, M.D., principal investigator of the clinical trial. “Based on the clinical observations, imaging studies and patient testimonies, I believe that CybroCell is a breakthrough in the treatment of DDD.”
As previously reported, after six and 12 months, more than 90 percent of the treatment group had over a 10-point reduction in Oswestry Disability Index (ODI) and 100 percent had improvement in Visual Analogue Scale (VAS). On average, treatment group patients showed a 59 percent improvement in ODI scores, compared to the 22 percent improvement in the placebo group.
“The MRI image study demonstrates that a single injection of CybroCell leads to persistent structural and functional improvement in patients with DDD over a year,” said SpinalCyte Chief Scientific Officer Thomas Ichim, Ph.D. “To my knowledge, the pain and MRI data are clearly superior to all other reported cell therapy trial data for DDD. Developing solutions which target and treat the source of chronic pain for patients with DDD, a population which today accounts for about 50 percent of prescription opioid usage, has implications in combating the opioid epidemic.”
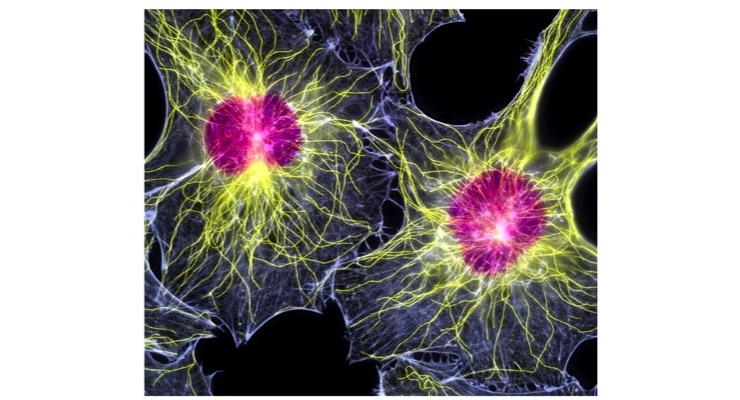
SpinalCyte’s Phase 1/Phase 2 clinical trial is the first allogeneic use of fibroblasts outside of skin conditions. CybroCell is based on research showing that skin derived cells are capable of healing the disc as previously reported in The Spine Journal.
“There are limited options to treat the pain in patients with DDD and no treatments that can regrow the spinal disc,” said SpinalCyte CEO Pete O’Heeron. “With the positive 12-month data from our trial, SpinalCyte is positioned at the forefront of this emerging sector of fibroblast-based cell therapies. In contrast to stem cells, which are difficult and costly to procure and develop into a drug, CybroCell uses fibroblasts which are found in large numbers and are already approved by the FDA for treatment of skin conditions. Our proprietary technology also holds promise for additional degenerative ailments including disc degeneration, cancer, diabetes, liver failure and heart failure.”
Degenerative disc disease (DDD) is a condition in which a patient’s spinal disc breaks down and can begin to collapse. It is estimated that 85 percent of people over the age of 50 have evidence of disc degeneration and over 1.3 million procedures a year are performed to treat the disease. The most common treatments for patients with DDD are either discectomy or spinal fusion. Discectomy is the partial or full removal of the degenerated disc to decompress and relieve the nervous system but can cause long term spinal pain. In a spinal fusion procedure, the entire disc is removed and the two adjacent vertebrae are fused together. It often increases strain on the adjacent discs and surrounding tissues leading to further degeneration.
CybroCell is the first off-the-shelf allogenic human dermal fibroblast (HDF) product for the treatment of degenerative disc disease. SpinalCyte’s Phase 1/Phase 2 clinical trial for injected human dermal fibroblasts in the treatment of DDD demonstrated after 12 months, patients who were injected with CybroCell had sustained improvement in pain relief and increased back mobility.
Based in Houston, Texas, SpinalCyte LLC is a regenerative medicine company developing a solution for spinal disc regeneration using human dermal fibroblasts. Currently, SpinalCyte holds 35 U.S. and international issued patents and has filed for an additional 41 patents pending. SpinalCyte holds 110 U.S. and international patents pending and issued across a variety of clinical pathways, including disc degeneration, cancer, diabetes, liver failure and heart failure. Funded entirely by angel investors, SpinalCyte represents the next generation of medical advancement in cell therapy.
The trial assessed patients using the Oswestry Disability Index (ODI), Visual Analogue Scale (VAS) and disc height via MRI scans. Using composite scoring, 54 percent of the treatment arm patients met all three endpoints as compared to only 17 percent with the placebo (p=0.0003). Over 84 percent of patients in the treatment group of CybroCell or CybroCell with platelet rich plasma (PRP) injections had an increase or no change in disc height compared to 25 percent for the placebo saline injection.
Analyzed individually, 83 percent of spinal discs that were injected with CybroCell had an increase or no change in disc height compared to 25 percent of saline injected discs. The 12-month MRI data from the landmark Phase 1/Phase 2 trial were analyzed for 17 patients by an independent radiological expert.
“Verification of structural improvements via MRI at six and 12 months strongly suggests that we are inducing a biological cascade of events associated with the induction of a regenerative process in the patient,” said Jesus Perez, M.D., principal investigator of the clinical trial. “Based on the clinical observations, imaging studies and patient testimonies, I believe that CybroCell is a breakthrough in the treatment of DDD.”
As previously reported, after six and 12 months, more than 90 percent of the treatment group had over a 10-point reduction in Oswestry Disability Index (ODI) and 100 percent had improvement in Visual Analogue Scale (VAS). On average, treatment group patients showed a 59 percent improvement in ODI scores, compared to the 22 percent improvement in the placebo group.
“The MRI image study demonstrates that a single injection of CybroCell leads to persistent structural and functional improvement in patients with DDD over a year,” said SpinalCyte Chief Scientific Officer Thomas Ichim, Ph.D. “To my knowledge, the pain and MRI data are clearly superior to all other reported cell therapy trial data for DDD. Developing solutions which target and treat the source of chronic pain for patients with DDD, a population which today accounts for about 50 percent of prescription opioid usage, has implications in combating the opioid epidemic.”
SpinalCyte’s Phase 1/Phase 2 clinical trial is the first allogeneic use of fibroblasts outside of skin conditions. CybroCell is based on research showing that skin derived cells are capable of healing the disc as previously reported in The Spine Journal.
“There are limited options to treat the pain in patients with DDD and no treatments that can regrow the spinal disc,” said SpinalCyte CEO Pete O’Heeron. “With the positive 12-month data from our trial, SpinalCyte is positioned at the forefront of this emerging sector of fibroblast-based cell therapies. In contrast to stem cells, which are difficult and costly to procure and develop into a drug, CybroCell uses fibroblasts which are found in large numbers and are already approved by the FDA for treatment of skin conditions. Our proprietary technology also holds promise for additional degenerative ailments including disc degeneration, cancer, diabetes, liver failure and heart failure.”
Degenerative disc disease (DDD) is a condition in which a patient’s spinal disc breaks down and can begin to collapse. It is estimated that 85 percent of people over the age of 50 have evidence of disc degeneration and over 1.3 million procedures a year are performed to treat the disease. The most common treatments for patients with DDD are either discectomy or spinal fusion. Discectomy is the partial or full removal of the degenerated disc to decompress and relieve the nervous system but can cause long term spinal pain. In a spinal fusion procedure, the entire disc is removed and the two adjacent vertebrae are fused together. It often increases strain on the adjacent discs and surrounding tissues leading to further degeneration.
CybroCell is the first off-the-shelf allogenic human dermal fibroblast (HDF) product for the treatment of degenerative disc disease. SpinalCyte’s Phase 1/Phase 2 clinical trial for injected human dermal fibroblasts in the treatment of DDD demonstrated after 12 months, patients who were injected with CybroCell had sustained improvement in pain relief and increased back mobility.
Based in Houston, Texas, SpinalCyte LLC is a regenerative medicine company developing a solution for spinal disc regeneration using human dermal fibroblasts. Currently, SpinalCyte holds 35 U.S. and international issued patents and has filed for an additional 41 patents pending. SpinalCyte holds 110 U.S. and international patents pending and issued across a variety of clinical pathways, including disc degeneration, cancer, diabetes, liver failure and heart failure. Funded entirely by angel investors, SpinalCyte represents the next generation of medical advancement in cell therapy.