Business Wire03.14.19
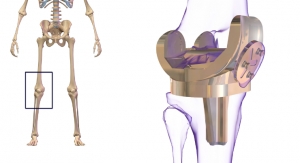
Exactech, a developer and producer of bone and joint restoration products and biologic solutions for extremities, knee, and hip announced today, that it has acquired XpandOrtho, a designer of soft tissue balancing instruments for knee replacement surgery. Exactech plans to incorporate XpandOrtho’s technology to enhance its ExactechGPS computer-assisted surgery system, the only navigation system that supports both shoulder and knee replacement.
Managing a patient’s soft tissue is an important aspect of knee replacement surgery. XpandOrtho received clearance from the U.S. Food and Drug Administration (FDA) in 2017 for an electronic, soft tissue balancing instrument for total knee arthroplasty. The intra-articular device uses multiple sensors within the knee joint to guide soft tissue balancing. The device uses a novel pneumatic-based bellows system, which wirelessly communicates with a display to provide the surgeon gap balancing feedback throughout the knee’s range of motion.
According to Exactech CEO David Petty, “The acquisition of XpandOrtho supports Exactech’s growth strategy and commitment to improving surgeon experience and patient outcomes. Their innovative ligament balancing technology provides a strong framework for integration into our ExactechGPS system to further advance our award-winning Truliant Total Knee System. We look forward to collaborating with XpandOrtho founders Cliff Colwell and Darryl D’Lima on continued development of advanced knee replacement technology.”
Darryl D’Lima M.D., Ph.D., and Clifford W. Colwell, Jr., M.D., report that they feel “privileged to have been purchased by an outstanding orthopedic device company such as Exactech.” Colwell added, “This allows our technology, which has been in development for more than seven years, to be brought to market for the benefit of surgeons and their patients.”
Managing a patient’s soft tissue is an important aspect of knee replacement surgery. XpandOrtho received clearance from the U.S. Food and Drug Administration (FDA) in 2017 for an electronic, soft tissue balancing instrument for total knee arthroplasty. The intra-articular device uses multiple sensors within the knee joint to guide soft tissue balancing. The device uses a novel pneumatic-based bellows system, which wirelessly communicates with a display to provide the surgeon gap balancing feedback throughout the knee’s range of motion.
According to Exactech CEO David Petty, “The acquisition of XpandOrtho supports Exactech’s growth strategy and commitment to improving surgeon experience and patient outcomes. Their innovative ligament balancing technology provides a strong framework for integration into our ExactechGPS system to further advance our award-winning Truliant Total Knee System. We look forward to collaborating with XpandOrtho founders Cliff Colwell and Darryl D’Lima on continued development of advanced knee replacement technology.”
Darryl D’Lima M.D., Ph.D., and Clifford W. Colwell, Jr., M.D., report that they feel “privileged to have been purchased by an outstanding orthopedic device company such as Exactech.” Colwell added, “This allows our technology, which has been in development for more than seven years, to be brought to market for the benefit of surgeons and their patients.”