Business Wire05.17.19
BrainScope, a medical neuro-technology company focused on concussion and mild traumatic brain injury (mTBI) assessment, has concluded both algorithm development and clinical validation studies in adolescent and young adult populations using its objective, EEG-based technology. The purpose of the studies was to create and validate an objective, personalized capability for assessment of concussion and readiness to return to activities in males and females between the ages of 13 and 25. The studies conducted from 2015 through 2019 at 52 clinical sites included nearly 1,700 patients and healthy controls, with over 4,000 evaluations. Funding sources for the studies included a $9.9 million research contract from the U.S. Department of Defense, award funding from the NFL-GE Head Health Challenge (in which BrainScope was a two-time awardee), and BrainScope private funding.
“We are extremely pleased to have concluded these substantial clinical studies over four years, the results of which are extremely exciting as we work in the near-term through appropriate regulatory processes to bring important new concussion assessment capabilities to the market,” stated Michael Singer, CEO of BrainScope. “In particular I want to thank the U.S. Department of Defense, the NFL, GE, and BrainScope investors for their financial support of this major initiative which has yielded such excellent results.”
In January, BrainScope announced that researchers had published preliminary results related to a concussion assessment capability for potential severity and likelihood of prolonged recovery from concussions in the peer-reviewed journal Computers in Biology and Medicine. The paper entitled, “A multimodal biomarker for concussion identification, prognosis and management” describes a study which enrolled 568 concussed and matched control patients between the ages of 13 and 25. Results from this study demonstrated highly significant differences in the BrainScope EEG-based biomarker at the time of injury between the 177 concussed patients and controls, and no significant differences when return-to-play was clinically determined.
Progressive recovery over time was also objectively demonstrated, especially in the prolonged recovery group. Results also supported the hypothesis that some of the more severely injured subjects may have been allowed to return to play too soon.
“These large studies add important information to the understanding of the pathophysiology of concussion, leading to better assessment capabilities which can improve short and long-term outcomes of head-injured patients,” added Leslie Prichep, Ph.D., chief scientific officer of BrainScope.
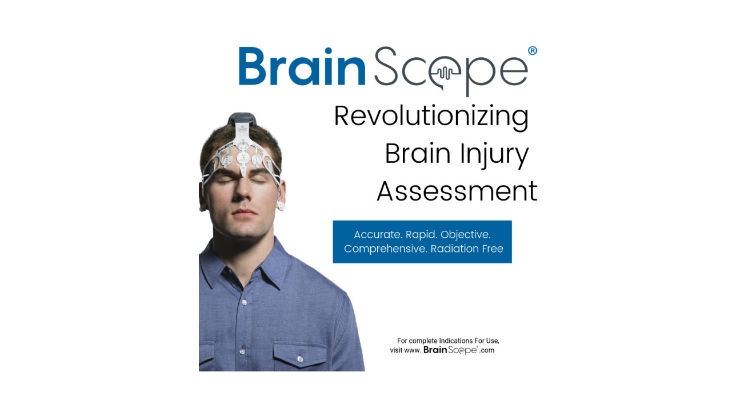
BrainScope’s flagship BrainScope One product, which is commercially available, is a multi-modal, comprehensive, handheld capability to help clinicians objectively and rapidly assess and triage head injured patients directly at the point of care. BrainScope One is the first U.S. Food and Drug Administration (FDA)-cleared medical device to offer capabilities to assess the full spectrum of brain injury—from concussions (functional abnormality) to brain bleeds (structural injury). BrainScope One is currently being fielded by the U.S. military, both stateside and internationally. BrainScope One is also being used in a cross-section of market segments including: urgent care and occupational health clinics; concussion clinics; hospital emergency rooms; university sports and student health centers; professional sports; and pharmaceutical clinical trials.
BrainScope’s mission is to revolutionize the rapid and objective assessment of brain-related conditions, starting with concussion and mild traumatic brain injury, utilizing multiple integrated assessment capabilities, artificial intelligence (AI) and digitization within a culture of quality, excellence and entrepreneurialism. The company’s first product, BrainScope One, incorporates a multi-modal, multi-parameter panel of assessment capabilities including EEG-based technology that is non-invasive for mildly presenting head-injured patients, 18-85 years old, within three days after injury and is not a replacement to CT scan. BrainScope’s technology platform integrates databases of thousands of brainwave recordings with AI technology and miniaturized hardware and disposable headset sensors, all of which are covered by an extensive intellectual property portfolio of over 100 issued and pending patents globally. BrainScope has received seven FDA clearances and ISO 13485 Certification. It has 26 peer-reviewed publications on its technology. Recent white papers authored by third parties have shown the potential for BrainScope One to decrease unnecessary head CT scans by one-third, to reduce head injury referrals to hospital emergency departments by up to 75 percent, and to reduce healthcare costs for insurers and patients by over 30 percent.
BrainScope has partnered with the U.S. Department of Defense (DoD) for the development of its mTBI and concussion assessment technology. BrainScope has been the recipient of several prestigious awards, including the Frost & Sullivan 2017 Best Practices Award for New Product Innovation in the Traumatic Brain Injury Assessment Solutions Market, was a two-time winner of the GE-NFL Head Health Challenge, and has received two nominations for the Prix Galien Best Medical Technology (2017 and 2018), regarded as the equivalent of the Nobel Prize for medical devices. BrainScope has received significant funding from private investors including DBL Partners, Revolution (created by AOL co-founder Steve Case), ZG Ventures and Maryland Venture Fund.
The clinical study was supported in part by the U.S. Army Contracting Command, Aberdeen Proving Ground, Natick Contracting Division.
“We are extremely pleased to have concluded these substantial clinical studies over four years, the results of which are extremely exciting as we work in the near-term through appropriate regulatory processes to bring important new concussion assessment capabilities to the market,” stated Michael Singer, CEO of BrainScope. “In particular I want to thank the U.S. Department of Defense, the NFL, GE, and BrainScope investors for their financial support of this major initiative which has yielded such excellent results.”
In January, BrainScope announced that researchers had published preliminary results related to a concussion assessment capability for potential severity and likelihood of prolonged recovery from concussions in the peer-reviewed journal Computers in Biology and Medicine. The paper entitled, “A multimodal biomarker for concussion identification, prognosis and management” describes a study which enrolled 568 concussed and matched control patients between the ages of 13 and 25. Results from this study demonstrated highly significant differences in the BrainScope EEG-based biomarker at the time of injury between the 177 concussed patients and controls, and no significant differences when return-to-play was clinically determined.
Progressive recovery over time was also objectively demonstrated, especially in the prolonged recovery group. Results also supported the hypothesis that some of the more severely injured subjects may have been allowed to return to play too soon.
“These large studies add important information to the understanding of the pathophysiology of concussion, leading to better assessment capabilities which can improve short and long-term outcomes of head-injured patients,” added Leslie Prichep, Ph.D., chief scientific officer of BrainScope.
BrainScope’s flagship BrainScope One product, which is commercially available, is a multi-modal, comprehensive, handheld capability to help clinicians objectively and rapidly assess and triage head injured patients directly at the point of care. BrainScope One is the first U.S. Food and Drug Administration (FDA)-cleared medical device to offer capabilities to assess the full spectrum of brain injury—from concussions (functional abnormality) to brain bleeds (structural injury). BrainScope One is currently being fielded by the U.S. military, both stateside and internationally. BrainScope One is also being used in a cross-section of market segments including: urgent care and occupational health clinics; concussion clinics; hospital emergency rooms; university sports and student health centers; professional sports; and pharmaceutical clinical trials.
BrainScope’s mission is to revolutionize the rapid and objective assessment of brain-related conditions, starting with concussion and mild traumatic brain injury, utilizing multiple integrated assessment capabilities, artificial intelligence (AI) and digitization within a culture of quality, excellence and entrepreneurialism. The company’s first product, BrainScope One, incorporates a multi-modal, multi-parameter panel of assessment capabilities including EEG-based technology that is non-invasive for mildly presenting head-injured patients, 18-85 years old, within three days after injury and is not a replacement to CT scan. BrainScope’s technology platform integrates databases of thousands of brainwave recordings with AI technology and miniaturized hardware and disposable headset sensors, all of which are covered by an extensive intellectual property portfolio of over 100 issued and pending patents globally. BrainScope has received seven FDA clearances and ISO 13485 Certification. It has 26 peer-reviewed publications on its technology. Recent white papers authored by third parties have shown the potential for BrainScope One to decrease unnecessary head CT scans by one-third, to reduce head injury referrals to hospital emergency departments by up to 75 percent, and to reduce healthcare costs for insurers and patients by over 30 percent.
BrainScope has partnered with the U.S. Department of Defense (DoD) for the development of its mTBI and concussion assessment technology. BrainScope has been the recipient of several prestigious awards, including the Frost & Sullivan 2017 Best Practices Award for New Product Innovation in the Traumatic Brain Injury Assessment Solutions Market, was a two-time winner of the GE-NFL Head Health Challenge, and has received two nominations for the Prix Galien Best Medical Technology (2017 and 2018), regarded as the equivalent of the Nobel Prize for medical devices. BrainScope has received significant funding from private investors including DBL Partners, Revolution (created by AOL co-founder Steve Case), ZG Ventures and Maryland Venture Fund.
The clinical study was supported in part by the U.S. Army Contracting Command, Aberdeen Proving Ground, Natick Contracting Division.