Axolotl Biologix Inc.06.27.19
A Texas private equity firm has announced the purchase of Axolotl Biologix Inc., a developer of regenerative medicine. Terms of the deal were not disclosed.
With the purchase, Axolotl board member Gary Lauterbach accepted the role of Interim CEO as Phil Larson exits. Other staff, including Chief Science Officer Robert Kellar, Ph.D., and Senior Director of Research & Development Robert B Diller, Ph.D., will remain as the company expects to initiate U.S. Food and Drug Administration (FDA) clinical trials of its current products and others under development.
“The investment by the private equity company allows Axolotl Biologix to execute a more long-term strategy which includes conducting FDA approved clinical trials to further grow acceptance and adoption of our regenerative products,” said Lauterbach. “We’re very excited about both the short and long-term opportunities awaiting Axolotl Biologix.”
Lauterbach’s goal is to expand distribution of Axolotl products beyond the 500 offices and clinics across the United States already utilizing the company’s products and secure new partners as a GSA contractor.
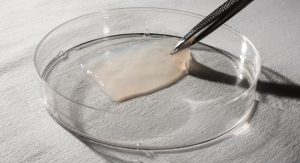
Axolotl Biologix Inc. develops regenerative medicine treatments through research, technology and clinical application. Axolotl Biologix is expanding the human body’s ability to regenerate by developing and manufacturing regenerative human cell and tissue medical technologies that are disrupting traditional, more invasive, painful and expensive treatment protocols. The company is based in Phoenix, Ariz.
With the purchase, Axolotl board member Gary Lauterbach accepted the role of Interim CEO as Phil Larson exits. Other staff, including Chief Science Officer Robert Kellar, Ph.D., and Senior Director of Research & Development Robert B Diller, Ph.D., will remain as the company expects to initiate U.S. Food and Drug Administration (FDA) clinical trials of its current products and others under development.
“The investment by the private equity company allows Axolotl Biologix to execute a more long-term strategy which includes conducting FDA approved clinical trials to further grow acceptance and adoption of our regenerative products,” said Lauterbach. “We’re very excited about both the short and long-term opportunities awaiting Axolotl Biologix.”
Lauterbach’s goal is to expand distribution of Axolotl products beyond the 500 offices and clinics across the United States already utilizing the company’s products and secure new partners as a GSA contractor.
Axolotl Biologix Inc. develops regenerative medicine treatments through research, technology and clinical application. Axolotl Biologix is expanding the human body’s ability to regenerate by developing and manufacturing regenerative human cell and tissue medical technologies that are disrupting traditional, more invasive, painful and expensive treatment protocols. The company is based in Phoenix, Ariz.