Globe Newswire07.26.19
SeaSpine Holdings Corporation, a global medical technology company focused on surgical solutions for the treatment of spinal disorders, announced today the implantation of the 20,000th NanoMetalene interbody device. This milestone exemplifies SeaSpine's mission to develop clinically relevant, cost-effective procedural solutions to treat spinal disorders by providing high-quality, differentiated, and complementary technologies that leverage the company’s core competencies in orthobiologics, interbody devices, and modular spinal implant systems.
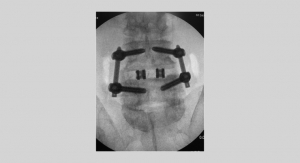
NanoMetalene is a proprietary surface technology for interbody implants that incorporates a sub-micron layer of commercially pure titanium molecularly bonded to a PEEK implant using a high-energy, low-temperature process referred to as atomic fusion deposition. It is designed to provide a bone-friendly titanium surface on endplates and throughout graft apertures, while retaining the benefits associated with traditional PEEK implants, such as biocompatibility, a modulus of elasticity similar to bone, and excellent radiographic visibility for post-operative imaging.
“This milestone reflects the success of our strategy to align NanoMetalene with our DBM portfolio and deliver synergistic solutions that maximize clinical value while reducing the need for high-cost orthobiologics,” stated Troy Woolley, vice president of Marketing, Spinal Implants. “Our ability to develop and deliver procedural solutions that address key clinical needs enables us to differentiate our products with both surgeons and our distributor partners and take share in this competitive market.”
SeaSpine has expanded NanoMetalene technology throughout its entire interbody portfolio. With the recent full launch of the Regatta Lateral System, NanoMetalene is now featured across seven different product families. They include Shoreline ACS (cervical), Cambria (cervical), Hollywood (posterior lumbar), Hollywood VI (posterior lumbar), Ventura (posterior lumbar), Vu aPOD Prime (anterior lumbar), and Regatta (lateral lumbar).
SeaSpine is a global medical technology company focused on the design, development, and commercialization of surgical solutions for treating spinal disorders. SeaSpine has a portfolio of orthobiologics and spinal implants solutions to meet the combinations of products that neurosurgeons and orthopedic spine surgeons need to perform fusion procedures on the lumbar, thoracic and cervical spine. SeaSpine’s orthobiologics products consist of a advanced and traditional bone graft substitutes that are designed to improve bone fusion rates following a wide range of orthopedic surgeries, including spine, hip, and extremities procedures. SeaSpine’s spinal implants portfolio consists of products to facilitate spinal fusion in degenerative, minimally invasive surgery (MIS), and complex spinal deformity procedures. Expertise in both orthobiologic sciences and spinal implants product development allows SeaSpine to offer its surgeon customers a differentiated portfolio and a complete solution to meet their fusion requirements. SeaSpine currently markets its products in the United States and in approximately 30 countries worldwide through a network of increasingly exclusive distribution partners.
NanoMetalene is a proprietary surface technology for interbody implants that incorporates a sub-micron layer of commercially pure titanium molecularly bonded to a PEEK implant using a high-energy, low-temperature process referred to as atomic fusion deposition. It is designed to provide a bone-friendly titanium surface on endplates and throughout graft apertures, while retaining the benefits associated with traditional PEEK implants, such as biocompatibility, a modulus of elasticity similar to bone, and excellent radiographic visibility for post-operative imaging.
“This milestone reflects the success of our strategy to align NanoMetalene with our DBM portfolio and deliver synergistic solutions that maximize clinical value while reducing the need for high-cost orthobiologics,” stated Troy Woolley, vice president of Marketing, Spinal Implants. “Our ability to develop and deliver procedural solutions that address key clinical needs enables us to differentiate our products with both surgeons and our distributor partners and take share in this competitive market.”
SeaSpine has expanded NanoMetalene technology throughout its entire interbody portfolio. With the recent full launch of the Regatta Lateral System, NanoMetalene is now featured across seven different product families. They include Shoreline ACS (cervical), Cambria (cervical), Hollywood (posterior lumbar), Hollywood VI (posterior lumbar), Ventura (posterior lumbar), Vu aPOD Prime (anterior lumbar), and Regatta (lateral lumbar).
SeaSpine is a global medical technology company focused on the design, development, and commercialization of surgical solutions for treating spinal disorders. SeaSpine has a portfolio of orthobiologics and spinal implants solutions to meet the combinations of products that neurosurgeons and orthopedic spine surgeons need to perform fusion procedures on the lumbar, thoracic and cervical spine. SeaSpine’s orthobiologics products consist of a advanced and traditional bone graft substitutes that are designed to improve bone fusion rates following a wide range of orthopedic surgeries, including spine, hip, and extremities procedures. SeaSpine’s spinal implants portfolio consists of products to facilitate spinal fusion in degenerative, minimally invasive surgery (MIS), and complex spinal deformity procedures. Expertise in both orthobiologic sciences and spinal implants product development allows SeaSpine to offer its surgeon customers a differentiated portfolio and a complete solution to meet their fusion requirements. SeaSpine currently markets its products in the United States and in approximately 30 countries worldwide through a network of increasingly exclusive distribution partners.