Global Market Insights09.03.19
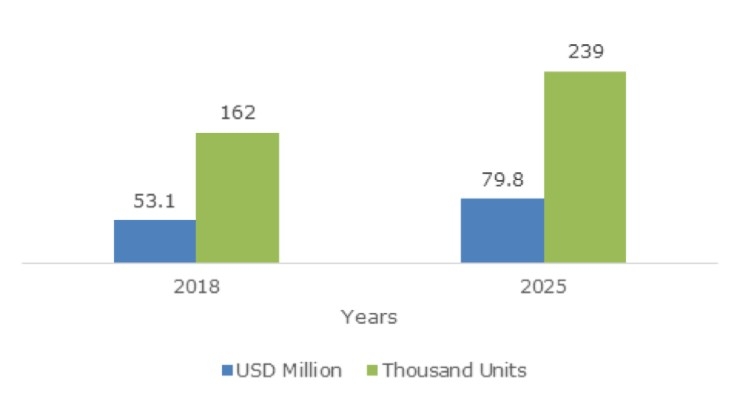
The Bone Growth Stimulators Market is set to grow from its current market value of more than $861.1 million to over 1.2 billion by 2024; according to a new research report by Global Market Insights.
A commendable increase in the number of clinical cases associated with bone injuries along with a growing elderly population worldwide will fuel the bone growth stimulators market size over 2019-2025. A bone growth stimulator is used to offer pain-free, electrical, or ultrasound stimulation that promotes faster bone healing and growth. An increase in cases of osteoarthritis across the globe has led to surging demand for bone growth stimulators.
According to the World Health Organization (WHO), rise in life expectancy and an aging population are expected to make osteoarthritis the fourth major cause of disability by 2020. The condition is increasingly witnessed among elderly people prone to bone injuries. The expanding geriatric population is also likely to boost bone growth stimulator demand in the coming years.
According to the report ‘An Aging World 2015,’ the number of aged people (over 65 years) is estimated to reach around 1.6 billion by 2050, representing 16.7 percent of the world’s total population. Femoral neck fractures have also intensified significantly over recent years, primarily due to rising elderly population and rapid urbanization.
Meanwhile, surge in number of cases associated with hip fractures worldwide are estimated to supplement the bone growth stimulators market’s outlook during the forecast time period. Credible reports suggest around 1.6 million hip fractures occur each year across the globe, and is likely to reach between 4.5 million and 6.3 million by 2050.
Based on product type, the external bone growth stimulators segment is anticipated to generate commendable revenues in the forecast period. External bone growth stimulators are portable, nonsurgical treatments offering devices designed specifically to promote healing of bone fractures that have failed to mend naturally.
With stringent regulations focusing on product quality and efficacy as well as rapid technological advancements, the bone growth stimulators industry players have developed innovative products. For instance, in 2018 Orthofix International N.V. announced U.S. Food and Drug Administration (FDA) and European CE mark approval for its next-gen PhysioStim bone growth stimulator, designed specifically for treating patients with nonunion fractures. More such advancements and enhanced product offerings are expected to lead the external bone growth stimulators market to register momentous growth over the coming years.
Growing prevalence of spinal fusion surgeries worldwide will expand the bone growth stimulators market size. According to WHO, each year around 250,000 and 500,000 people suffer from spinal cord injury (SCI) worldwide. Most of these injuries occur due to instances like road accidents, sports injuries, falls, assault, or due to diseases like arthritis, osteoporosis, and infections (TB). Growing incidence of herniated disc and spinal stenosis are also responsible for increased need for spinal fusion surgeries.
Bone growth stimulation (BGS) is increasingly prescribed to patients following spinal fusion surgeries, as the device can effectively activate the body’s natural bone healing process, which may be impaired in at-risk patients. BGS can also help fuse spinal bone after a fusion procedure or as a treatment from a failed fusion procedure.
Rapid technological advancements will lead to development of advanced spinal injury-specific stimulators, thereby supplementing trends in the bone growth stimulators industry. For instance, Orthofix has develop SpinalStim, a lightweight, flexible, and portable BGS that can be used after spinal fusion surgery or to treat a failed fusion from a prior surgery.
Technological advancements have also led to development of BGS products that offer advanced features like LCD display and an audible alarm to offer insights like operational status, treatment time remaining, and battery capacity. With growing demands and advancements, the spinal fusion surgeries segment of the bone growth stimulators market is estimated to grow with the highest CAGR of 6.0 percent over the forecast timeframe.
From a regional perspective, the Asia-Pacific bone growth stimulators market is estimated to garner substantial proceeds owing to rising cases of hip fractures and growing elderly population. According to the International Osteoporosis Foundation (IOF), over 50 percent of all osteoporotic hip fractures are likely to occur in Asia by 2050.
The condition is greatly underdiagnosed and undertreated in Asia and is at high risk among patients with fractures. The problem is particularly acute in rural areas across populous countries like China and India, where hip fractures are often treated at home instead of hospitals.
Hip fractures in the elderly and adult population are also rising rapidly in the Indian subcontinent. For instance, in 2018, a study published in the Indian Journal of Medical Research showed over 69 percent of people aged between 38-68 in Delhi suffered from continuous bone loss and high risk of fractures. Meanwhile, reports from the George Institute estimates that by 2020 almost 20 percent of the Indian population will be older than 60 years, while annual incidence of hip fractures will reach around 600,000, augmenting the bone growth stimulators industry’s trends.
In China, spending on hip fracture treatment is estimated to rise from $12.5 billion in 2020 to over $264.7 billion by 2050. Trends such as these are expected to immensely boost the Asia Pacific bone growth stimulators industry’s share over the forecast period.
A commendable increase in the number of clinical cases associated with bone injuries along with a growing elderly population worldwide will fuel the bone growth stimulators market size over 2019-2025. A bone growth stimulator is used to offer pain-free, electrical, or ultrasound stimulation that promotes faster bone healing and growth. An increase in cases of osteoarthritis across the globe has led to surging demand for bone growth stimulators.
According to the World Health Organization (WHO), rise in life expectancy and an aging population are expected to make osteoarthritis the fourth major cause of disability by 2020. The condition is increasingly witnessed among elderly people prone to bone injuries. The expanding geriatric population is also likely to boost bone growth stimulator demand in the coming years.
According to the report ‘An Aging World 2015,’ the number of aged people (over 65 years) is estimated to reach around 1.6 billion by 2050, representing 16.7 percent of the world’s total population. Femoral neck fractures have also intensified significantly over recent years, primarily due to rising elderly population and rapid urbanization.
Meanwhile, surge in number of cases associated with hip fractures worldwide are estimated to supplement the bone growth stimulators market’s outlook during the forecast time period. Credible reports suggest around 1.6 million hip fractures occur each year across the globe, and is likely to reach between 4.5 million and 6.3 million by 2050.
Based on product type, the external bone growth stimulators segment is anticipated to generate commendable revenues in the forecast period. External bone growth stimulators are portable, nonsurgical treatments offering devices designed specifically to promote healing of bone fractures that have failed to mend naturally.
With stringent regulations focusing on product quality and efficacy as well as rapid technological advancements, the bone growth stimulators industry players have developed innovative products. For instance, in 2018 Orthofix International N.V. announced U.S. Food and Drug Administration (FDA) and European CE mark approval for its next-gen PhysioStim bone growth stimulator, designed specifically for treating patients with nonunion fractures. More such advancements and enhanced product offerings are expected to lead the external bone growth stimulators market to register momentous growth over the coming years.
Growing prevalence of spinal fusion surgeries worldwide will expand the bone growth stimulators market size. According to WHO, each year around 250,000 and 500,000 people suffer from spinal cord injury (SCI) worldwide. Most of these injuries occur due to instances like road accidents, sports injuries, falls, assault, or due to diseases like arthritis, osteoporosis, and infections (TB). Growing incidence of herniated disc and spinal stenosis are also responsible for increased need for spinal fusion surgeries.
Bone growth stimulation (BGS) is increasingly prescribed to patients following spinal fusion surgeries, as the device can effectively activate the body’s natural bone healing process, which may be impaired in at-risk patients. BGS can also help fuse spinal bone after a fusion procedure or as a treatment from a failed fusion procedure.
Rapid technological advancements will lead to development of advanced spinal injury-specific stimulators, thereby supplementing trends in the bone growth stimulators industry. For instance, Orthofix has develop SpinalStim, a lightweight, flexible, and portable BGS that can be used after spinal fusion surgery or to treat a failed fusion from a prior surgery.
Technological advancements have also led to development of BGS products that offer advanced features like LCD display and an audible alarm to offer insights like operational status, treatment time remaining, and battery capacity. With growing demands and advancements, the spinal fusion surgeries segment of the bone growth stimulators market is estimated to grow with the highest CAGR of 6.0 percent over the forecast timeframe.
From a regional perspective, the Asia-Pacific bone growth stimulators market is estimated to garner substantial proceeds owing to rising cases of hip fractures and growing elderly population. According to the International Osteoporosis Foundation (IOF), over 50 percent of all osteoporotic hip fractures are likely to occur in Asia by 2050.
The condition is greatly underdiagnosed and undertreated in Asia and is at high risk among patients with fractures. The problem is particularly acute in rural areas across populous countries like China and India, where hip fractures are often treated at home instead of hospitals.
Hip fractures in the elderly and adult population are also rising rapidly in the Indian subcontinent. For instance, in 2018, a study published in the Indian Journal of Medical Research showed over 69 percent of people aged between 38-68 in Delhi suffered from continuous bone loss and high risk of fractures. Meanwhile, reports from the George Institute estimates that by 2020 almost 20 percent of the Indian population will be older than 60 years, while annual incidence of hip fractures will reach around 600,000, augmenting the bone growth stimulators industry’s trends.
In China, spending on hip fracture treatment is estimated to rise from $12.5 billion in 2020 to over $264.7 billion by 2050. Trends such as these are expected to immensely boost the Asia Pacific bone growth stimulators industry’s share over the forecast period.