Business Wire09.26.19
RTI Surgical Holdings Inc., a global surgical implant company announced the full commercial launch of the CervAlign Anterior Cervical Plate (ACP) System to add to the growth of its Established Therapies spine portfolio.
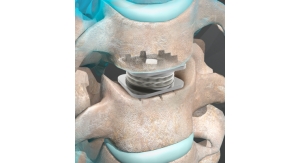
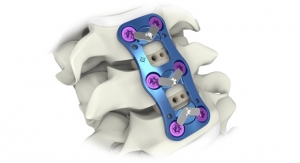
The CervAlign ACP System is designed to promote cervical fusion by providing temporary resistance to flexion, extension, lateral bending and axial rotation with strength and stiffness in the cervical spine (C2-C7). The system includes implants of various plate and screw sizes to accommodate varying patient anatomies. It also helps reduce complexity for the surgeon customer with an integrated cover-style locking mechanism that adds zero profile, streamlined instrumentation with the same driver for screws, pins, locking and revisions, and low-profile plates ranging from one to five levels, each with large graft windows for intraoperative visibility. Additionally, using the Fortilink-C Interbody Fusion (IBF) System with TETRAfuse 3D Technology and ViBone Viable Bone Matrix with the CervAlign ACP provides surgeons a complete anterior cervical discectomy and fusion (ACDF) solution.
“The locking mechanism on the CervAlign ACP System is easily visible and engaged, and there is no guesswork as to whether it is completely locked,” said Franco E. Vigna, M.D., MPH, FACOS of reVive Spine Center in Buffalo, New York. “Additionally, the amount of screw angulation allows for use of a shorter plate. Combined with the large graft windows and visibility of the plate, it allows surgeons to easily instrument the hard-to-access levels such as C3.”
The CervAlign ACP System is intended for anterior cervical fixation (C2-C7) for the following conditions: degenerative disc disease (DDD) defined as neck pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies, spondylolisthesis, trauma (e.g., fracture or dislocation), spinal stenosis, deformities or curvatures (e.g., scoliosis, kyphosis, and/or lordosis), tumor, pseudarthrosis and failed previous fusion.
“After a successful Limited Market Release, we are excited to commercially launch the CervAlign ACP system to offer a plate with a cover-style locking mechanism and streamlined instrumentation,” said Camille Farhat, president and CEO. “This is a great example of how we plan to use home-grown innovation to accelerate our spine business. CervAlign ACP adds another option for surgeons in our current Established Therapies portfolio, which we have recently reinvigorated through our R&D efforts across hardware, interbodies and biologics. As we further accelerate growth in spine, we will be able to meet the demands of more surgeons and treat more patients.”
The CervAlign ACP System is designed to promote cervical fusion by providing temporary resistance to flexion, extension, lateral bending and axial rotation with strength and stiffness in the cervical spine (C2-C7). The system includes implants of various plate and screw sizes to accommodate varying patient anatomies. It also helps reduce complexity for the surgeon customer with an integrated cover-style locking mechanism that adds zero profile, streamlined instrumentation with the same driver for screws, pins, locking and revisions, and low-profile plates ranging from one to five levels, each with large graft windows for intraoperative visibility. Additionally, using the Fortilink-C Interbody Fusion (IBF) System with TETRAfuse 3D Technology and ViBone Viable Bone Matrix with the CervAlign ACP provides surgeons a complete anterior cervical discectomy and fusion (ACDF) solution.
“The locking mechanism on the CervAlign ACP System is easily visible and engaged, and there is no guesswork as to whether it is completely locked,” said Franco E. Vigna, M.D., MPH, FACOS of reVive Spine Center in Buffalo, New York. “Additionally, the amount of screw angulation allows for use of a shorter plate. Combined with the large graft windows and visibility of the plate, it allows surgeons to easily instrument the hard-to-access levels such as C3.”
The CervAlign ACP System is intended for anterior cervical fixation (C2-C7) for the following conditions: degenerative disc disease (DDD) defined as neck pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies, spondylolisthesis, trauma (e.g., fracture or dislocation), spinal stenosis, deformities or curvatures (e.g., scoliosis, kyphosis, and/or lordosis), tumor, pseudarthrosis and failed previous fusion.
“After a successful Limited Market Release, we are excited to commercially launch the CervAlign ACP system to offer a plate with a cover-style locking mechanism and streamlined instrumentation,” said Camille Farhat, president and CEO. “This is a great example of how we plan to use home-grown innovation to accelerate our spine business. CervAlign ACP adds another option for surgeons in our current Established Therapies portfolio, which we have recently reinvigorated through our R&D efforts across hardware, interbodies and biologics. As we further accelerate growth in spine, we will be able to meet the demands of more surgeons and treat more patients.”