PR Newswire10.17.19
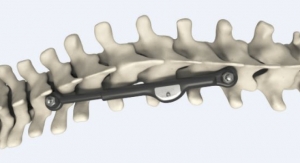
The U.S. Food and Drug Administration (FDA) has cleared CarboFix's CarboClear Carbon Fiber Vertebral Body Replacement (VBR) System to replace a collapsed, damaged, or unstable vertebral body due to tumor or trauma.
The CarboClear VBR System joins the FDA cleared CarboClear Pedicle Screw and CarboClear Fenestrated Pedicle Screw Systems.
The CarboClear VBR implant is made of carbon fibers, with integrated porous Ti-alloy endplates. Carbon fiber implants are proposing unique advantages to the oncological patients and their physicians. Among those advantages are: enhanced radiation therapy planning abilities, allowing radiation treatment to optimize the radiation dose to the tumor with minimal collateral tissue damage, and enhanced follow up abilities due to artifacts-free imaging. In addition, the implants provide unparalleled fatigue strength to support the impaired healing process in those patients, as well as compatibility with particle radiation (proton and carbon ion) and other stereotactic radio-surgery modalities.
Indications for Use: The CarboClear Vertebral Body Replacement (VBR) System is intended for use during open surgical procedures in the thoracolumbar spine (T1-L5) to replace a collapsed, damaged, or unstable vertebral body due to tumor or trauma (e.g., fracture).
The CarboClear Vertebral Body Replacement device is intended to be used with supplemental internal spinal fixation systems that have been labeled for use in the thoracic and lumbar spine (i.e., posterior pedicle screw and rod systems, anterior plate systems, and anterior screw and rod systems).
The use of allograft or autograft with the CarboClear Vertebral Body Replacement device is optional.
To learn more about the CarboClear Pedicle Screw System and the new CarboClear Vertebral Body Replacement (VBR) System, please stop by booth #249 during the CNS 2019 in San Francisco.
The CarboClear VBR System joins the FDA cleared CarboClear Pedicle Screw and CarboClear Fenestrated Pedicle Screw Systems.
The CarboClear VBR implant is made of carbon fibers, with integrated porous Ti-alloy endplates. Carbon fiber implants are proposing unique advantages to the oncological patients and their physicians. Among those advantages are: enhanced radiation therapy planning abilities, allowing radiation treatment to optimize the radiation dose to the tumor with minimal collateral tissue damage, and enhanced follow up abilities due to artifacts-free imaging. In addition, the implants provide unparalleled fatigue strength to support the impaired healing process in those patients, as well as compatibility with particle radiation (proton and carbon ion) and other stereotactic radio-surgery modalities.
Indications for Use: The CarboClear Vertebral Body Replacement (VBR) System is intended for use during open surgical procedures in the thoracolumbar spine (T1-L5) to replace a collapsed, damaged, or unstable vertebral body due to tumor or trauma (e.g., fracture).
The CarboClear Vertebral Body Replacement device is intended to be used with supplemental internal spinal fixation systems that have been labeled for use in the thoracic and lumbar spine (i.e., posterior pedicle screw and rod systems, anterior plate systems, and anterior screw and rod systems).
The use of allograft or autograft with the CarboClear Vertebral Body Replacement device is optional.
To learn more about the CarboClear Pedicle Screw System and the new CarboClear Vertebral Body Replacement (VBR) System, please stop by booth #249 during the CNS 2019 in San Francisco.