Globe Newswire06.22.20
Conformis Inc. today announced 510(k) clearance by the U.S. Food and Drug Administration (FDA) of the company’s next-generation iTotal Identity PS Knee System.
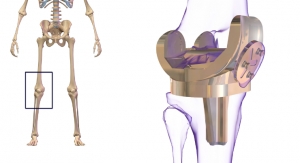
The iTotal Identity PS Knee System uses proprietary advanced imaging and design software, to deliver a patient-specific, pre-operative surgical plan with a comprehensive set of iJig instruments and patient-matched implants. Updates to this system include stem extensions available for patients with high Body Mass Index (BMI), titanium tibial baseplates with patient-specific cement rails, and metal cut guides and refined iJigs for a more traditional bone cutting experience.
“Our new iTotal Identity PS knee is an important addition to our total knee portfolios. Having a posterior stabilized (PS) option for our Identity system will allow surgeons to treat a wider range of patients more effectively and with greater confidence than ever before,” said Mark Augusti, president and CEO of Conformis Inc. “We remain the leader in bringing best-in-class PSI guides in a market-leading, efficient 'knee-in-a-box' model to the orthopedic surgeons.”
The global knee joint reconstruction market is projected at over $9 billion, with approximately 1 million knee replacements projected to be performed in the United States this year.
In January, Conformis achieved the milestone of 100,000 knee joint replacement implants.
“We are very pleased to announce this tremendous milestone for personalized joint replacement implant treatments,” said Augusti. “We are strong advocates for the utilization of patient-specific implants in total knee replacement because the size, shape and curvature of the knee are unique for each patient. Implants designed specifically for each patient enable efficient surgery and higher levels of patient satisfaction versus standard ‘off-the-shelf’ implants. Thank you to all of the surgeons and patients who have traveled this journey with us and helped achieve this significant milestone.”
More than 20 peer-reviewed clinical studies published in medical journals to date support the strong performance of Conformis technology. In 2018, Conformis engaged PrecisionEffect and SurveyHealthcareGlobus (SHG) to conduct a study of patient satisfaction with knee implants (Conformis vs. non-Conformis). The results of this third-party study show that 87 percent of the Conformis patients surveyed were highly likely to recommend their knee implant to a friend or colleague, versus only 70 percent of the non-Conformis patients surveyed.
The 100,000 knee joint replacement implant milestone followed the November 2019 FDA clearance of the 3D-designed Conformis Hip System and the system’s full commercial launch. Similar to the design process for Conformis knee technologies, the Conformis Hip System uses proprietary advanced imaging and design software, to deliver a patient-specific, pre-operative surgical plan with a comprehensive set of iJig instruments and patient-matched implants. The Conformis Hip System iJigs work in conjunction with anatomic landmarks to ensure the proper placement and orientation of the femoral stem, as well as the acetabular cup. In addition, Conformis’ proprietary reamer system provides a single-use, patient-matched reaming solution that delivers efficient and accurate cup orientation to a controlled depth for each surgical case.
The iTotal Identity PS Knee System uses proprietary advanced imaging and design software, to deliver a patient-specific, pre-operative surgical plan with a comprehensive set of iJig instruments and patient-matched implants. Updates to this system include stem extensions available for patients with high Body Mass Index (BMI), titanium tibial baseplates with patient-specific cement rails, and metal cut guides and refined iJigs for a more traditional bone cutting experience.
“Our new iTotal Identity PS knee is an important addition to our total knee portfolios. Having a posterior stabilized (PS) option for our Identity system will allow surgeons to treat a wider range of patients more effectively and with greater confidence than ever before,” said Mark Augusti, president and CEO of Conformis Inc. “We remain the leader in bringing best-in-class PSI guides in a market-leading, efficient 'knee-in-a-box' model to the orthopedic surgeons.”
The global knee joint reconstruction market is projected at over $9 billion, with approximately 1 million knee replacements projected to be performed in the United States this year.
In January, Conformis achieved the milestone of 100,000 knee joint replacement implants.
“We are very pleased to announce this tremendous milestone for personalized joint replacement implant treatments,” said Augusti. “We are strong advocates for the utilization of patient-specific implants in total knee replacement because the size, shape and curvature of the knee are unique for each patient. Implants designed specifically for each patient enable efficient surgery and higher levels of patient satisfaction versus standard ‘off-the-shelf’ implants. Thank you to all of the surgeons and patients who have traveled this journey with us and helped achieve this significant milestone.”
More than 20 peer-reviewed clinical studies published in medical journals to date support the strong performance of Conformis technology. In 2018, Conformis engaged PrecisionEffect and SurveyHealthcareGlobus (SHG) to conduct a study of patient satisfaction with knee implants (Conformis vs. non-Conformis). The results of this third-party study show that 87 percent of the Conformis patients surveyed were highly likely to recommend their knee implant to a friend or colleague, versus only 70 percent of the non-Conformis patients surveyed.
The 100,000 knee joint replacement implant milestone followed the November 2019 FDA clearance of the 3D-designed Conformis Hip System and the system’s full commercial launch. Similar to the design process for Conformis knee technologies, the Conformis Hip System uses proprietary advanced imaging and design software, to deliver a patient-specific, pre-operative surgical plan with a comprehensive set of iJig instruments and patient-matched implants. The Conformis Hip System iJigs work in conjunction with anatomic landmarks to ensure the proper placement and orientation of the femoral stem, as well as the acetabular cup. In addition, Conformis’ proprietary reamer system provides a single-use, patient-matched reaming solution that delivers efficient and accurate cup orientation to a controlled depth for each surgical case.