Business Wire07.20.20
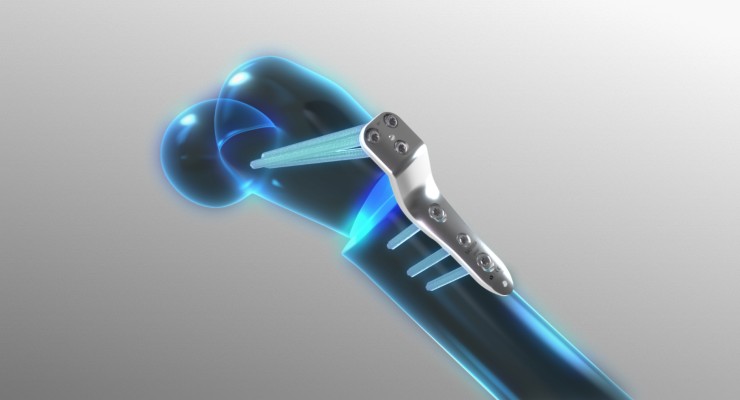
Orthofix Medical Inc., a global medical device company focused on musculoskeletal healing products, has received the U.S. Food and Drug Administration (FDA) 510(k) clearance and European CE mark approval for the JuniOrtho Plating System. Created specifically for pediatric patients, this innovative fixation system is designed to address the demands of advanced deformity and trauma reconstruction of the lower extremities.
“The field of deformity correction is rapidly evolving with multiple technical and technological advances every year,” said Dr. Philip McClure, an orthopedic surgeon at the International Center for Limb Lengthening in Baltimore. “External fixators are an incredibly powerful tool in caring for deformities in children around the world. Improved designs of internal fixation devices have allowed us to provide complimentary options to external fixators to benefit our patients and their families. Our goal is to obtain the best possible outcome so children can enjoy their lives after deformity correction surgery.”
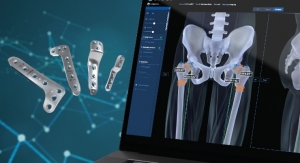
The JuniOrtho Plating System is complemented by a pre-operative planning software option that streamlines the implant selection prior to the surgical procedure. This unique platform enables the surgeon to accurately plan the osteotomy position to visualize the implant in relation to the anatomy. This aids in the selection of the precise size of device to ensure the best fit and optimal positioning for the patient’s body. Specifically developed to be used in combination with the JuniOrtho Plating System, the software is currently available in Europe and planned for release in the U.S. later this year.
“The JuniOrtho Plating System represents our continued commitment to advancing pediatric orthopedics by providing surgeons the devices they need to treat even the smallest of patients,” said Jon Serbousek, president and CEO of Orthofix. “We are excited to now be able to offer surgeons both an internal and external fixation systems to expand our portfolio of pediatric deformity care solutions.”
Offered in a wide range of plate sizes with a variety of lengths, the JuniOrtho Plating System accommodates both locking and non-locking screws corresponding to the plate size. The system is comprised of sterile implants and single-use tools to reduce the risk of contamination and optimize efficiency during the procedure.
The JuniOrtho Plating System is part of the JuniOrtho line of pediatric solutions that includes the TL-HEX system, TrueLok Ring Fixation System, eight-Plate Guided Growth System+, and many others. JuniOrtho brings products and resources together to give medical professionals and families alike the best in pediatric orthopedic solutions.
“The field of deformity correction is rapidly evolving with multiple technical and technological advances every year,” said Dr. Philip McClure, an orthopedic surgeon at the International Center for Limb Lengthening in Baltimore. “External fixators are an incredibly powerful tool in caring for deformities in children around the world. Improved designs of internal fixation devices have allowed us to provide complimentary options to external fixators to benefit our patients and their families. Our goal is to obtain the best possible outcome so children can enjoy their lives after deformity correction surgery.”
The JuniOrtho Plating System is complemented by a pre-operative planning software option that streamlines the implant selection prior to the surgical procedure. This unique platform enables the surgeon to accurately plan the osteotomy position to visualize the implant in relation to the anatomy. This aids in the selection of the precise size of device to ensure the best fit and optimal positioning for the patient’s body. Specifically developed to be used in combination with the JuniOrtho Plating System, the software is currently available in Europe and planned for release in the U.S. later this year.
“The JuniOrtho Plating System represents our continued commitment to advancing pediatric orthopedics by providing surgeons the devices they need to treat even the smallest of patients,” said Jon Serbousek, president and CEO of Orthofix. “We are excited to now be able to offer surgeons both an internal and external fixation systems to expand our portfolio of pediatric deformity care solutions.”
Offered in a wide range of plate sizes with a variety of lengths, the JuniOrtho Plating System accommodates both locking and non-locking screws corresponding to the plate size. The system is comprised of sterile implants and single-use tools to reduce the risk of contamination and optimize efficiency during the procedure.
The JuniOrtho Plating System is part of the JuniOrtho line of pediatric solutions that includes the TL-HEX system, TrueLok Ring Fixation System, eight-Plate Guided Growth System+, and many others. JuniOrtho brings products and resources together to give medical professionals and families alike the best in pediatric orthopedic solutions.