CTL Amedica09.02.20
CTL Amedica Corporation has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its MONDRIAN Lumbar Interbody Fusion (LIF) Cage System featuring TiCro surface technology, a unique approach to titanium interbody fusion devices.
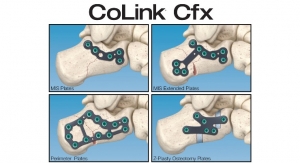
“Using our CEZANNE LIF System and other CTL Amedica products as a foundation, our team created the MONDRIAN LIF Cage System, a revolutionary new product that utilizes our proprietary TiCro technology, which has an aggressive macro surface and unique geometry that enhances interlocking properties and increases bone ingrowth surface area,” said Daniel Chon, CTL Amedica Corporation president and CEO. “With multiple material offerings, 15 different cage configurations, 34 footprints, seven lordotic options and 7,160 part combinations, the sheer breadth of this clearance is a tremendous accomplishment and a testament to our team’s dedication to ‘moving the needle’ in product innovation and design. We’re grateful for the clearance and look forward to presenting the MONDRIAN LIF Cage System to the market soon.”
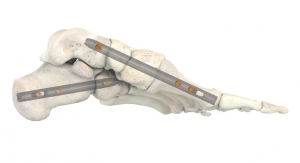
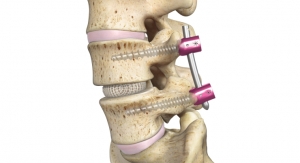
The MONDRIAN LIF Cage System is indicated for intervertebral body fusion procedures in skeletally mature patients with degenerative disc disease at one or two adjoining levels from L2-S1. The system is designed to be used with supplemental fixation cleared for use in the lower back. The robust fusion system features a large graft window, distinct radiographic markers, a self-distracting bullet tip, a bi-convex profile and lateral aperture.
“The MONDRIAN LIF Cage System is available in a wide variety of sizes and configurations to better accommodate anatomical variation,” added Chon. “This enables greater intraoperative flexibility, which can result in shorter, more efficient procedures, possibly improving success rates and recovery time for patients.”
Of the MONDRIAN LIF offerings, the titanium bullet TLIF and PLIF with TiCro technology are scheduled for limited launch in the fourth quarter, with other implant formats soon to follow.
“Using our CEZANNE LIF System and other CTL Amedica products as a foundation, our team created the MONDRIAN LIF Cage System, a revolutionary new product that utilizes our proprietary TiCro technology, which has an aggressive macro surface and unique geometry that enhances interlocking properties and increases bone ingrowth surface area,” said Daniel Chon, CTL Amedica Corporation president and CEO. “With multiple material offerings, 15 different cage configurations, 34 footprints, seven lordotic options and 7,160 part combinations, the sheer breadth of this clearance is a tremendous accomplishment and a testament to our team’s dedication to ‘moving the needle’ in product innovation and design. We’re grateful for the clearance and look forward to presenting the MONDRIAN LIF Cage System to the market soon.”
The MONDRIAN LIF Cage System is indicated for intervertebral body fusion procedures in skeletally mature patients with degenerative disc disease at one or two adjoining levels from L2-S1. The system is designed to be used with supplemental fixation cleared for use in the lower back. The robust fusion system features a large graft window, distinct radiographic markers, a self-distracting bullet tip, a bi-convex profile and lateral aperture.
“The MONDRIAN LIF Cage System is available in a wide variety of sizes and configurations to better accommodate anatomical variation,” added Chon. “This enables greater intraoperative flexibility, which can result in shorter, more efficient procedures, possibly improving success rates and recovery time for patients.”
Of the MONDRIAN LIF offerings, the titanium bullet TLIF and PLIF with TiCro technology are scheduled for limited launch in the fourth quarter, with other implant formats soon to follow.