PR Newswire10.09.20
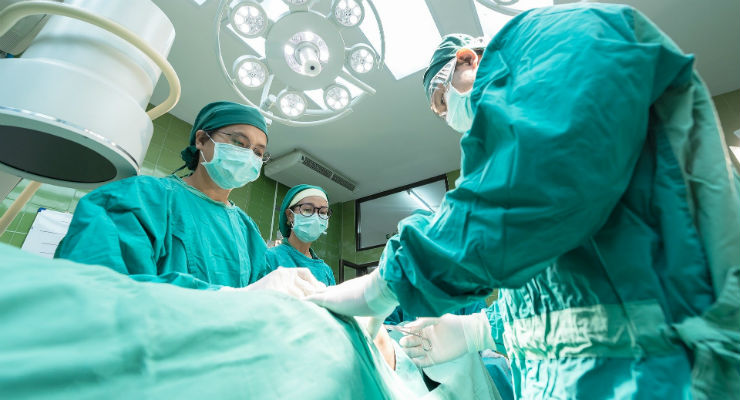
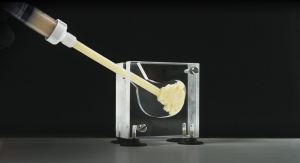
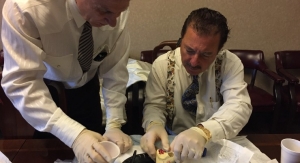
Biologica Technologies, developer and marketer of ProteiOS, announced the introduction and initial usage of their growth factor enriched cellular bone matrix (CBM), OsseoGEN. OsseoGEN CBM contains all three elements needed for bone regeneration; osteoconductivity, osteoinductivity and osteogenicity and is supplied via an open-bore syringe for easy dispensing.
Following in the footsteps of ProteiOS with its novel, proprietary processing technology, OsseoGEN contains significant osteoinductive, angiogenic and mitogenic growth factors setting it apart from other cellular bone matrices on the market today. OsseoGEN is DMSO-free, eliminating the need to rinse and decant the tissue prior to implantation, and possesses a thaw time approximately half of competitive CBMs being used today.
"We are excited to bring this bone graft to the market and demonstrate clinical efficacy in a segment of the market that lacks sufficient clinical proof," stated Amit Govil, president and founder of Biologica Technologies, "We feel that the growth factor enrichment processes that are utilized to produce OsseoGEN will provide surgeons with another option to improve patient outcomes."
Following in the footsteps of ProteiOS with its novel, proprietary processing technology, OsseoGEN contains significant osteoinductive, angiogenic and mitogenic growth factors setting it apart from other cellular bone matrices on the market today. OsseoGEN is DMSO-free, eliminating the need to rinse and decant the tissue prior to implantation, and possesses a thaw time approximately half of competitive CBMs being used today.
"We are excited to bring this bone graft to the market and demonstrate clinical efficacy in a segment of the market that lacks sufficient clinical proof," stated Amit Govil, president and founder of Biologica Technologies, "We feel that the growth factor enrichment processes that are utilized to produce OsseoGEN will provide surgeons with another option to improve patient outcomes."