Sam Brusco, Associate Editor02.18.21
The U.S. Food and Drug Administration approved Additive Orthopaedics’ Patient Specific Talus Spacer 3D-printed talus implant for humanitarian use. According to the FDA, it’s the first in the world and first-of-its-kind implant to replace the talus to treat avascular necrosis (AVN) of the ankle joint, a serious and progressive condition that causes bone tissue death due to lack of blood supply to the area. The implant provides a joint-sparing alternative to surgeries commonly used for late-stage AVN that may disable ankle joint motion.
“Avascular necrosis of the ankle, while a rare condition, is a serious and potentially debilitating one that causes pain and can lead to inhibited motion of the ankle joint, and in some cases, removal of part of the leg,” Capt. Raquel Peat, Ph.D., M.P.H., USPHS, director of the FDA’s Center for Devices and Radiological Health’s Office of Orthopedic Devices told the press. “[This] action provides patients with a treatment option that could potentially reduce pain, retain range of motion of their joint and improve quality of life.”
AVN is often caused by a sudden injury or sustained damage to the tissue, and it occurs when there is a lack of blood supply to bone tissue, causing it to become necrotic. Late-stage AVN of the ankle may result in the talus bone partially or fully collapsing. Current available treatments include fusing the joints in the foot and ankle together—a procedure which helps to alleviate pain caused by AVN but eliminates joint motion—or below-the-knee amputation.
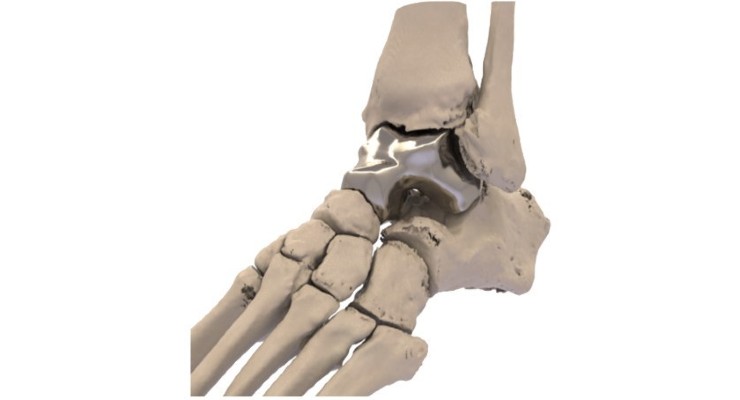
The Patient Specific Talus Spacer is a 3D printed implant that can be used in talus replacement surgery. The talus spacer is made for each patient individually, modeled from computed tomography (CT) imaging, and is fitted to a patient’s specific anatomy. During the replacement surgery, the patient’s talus bone is removed and replaced with the implant, which is made from cobalt chromium alloy.
While fusion may become necessary in the future should the condition worsen, talus replacement surgery with the Patient Specific Talus Spacer is intended to be a joint-sparing procedure, as it allows the patient to retain motion in the ankle joint.
The FDA reviewed data for the Patient Specific Talus Spacer through the humanitarian device exemption (HDE) process. A Humanitarian Use Device (HUD) is a device that is intended to benefit patients by treating or diagnosing a disease or condition that affects not more than 8,000 individuals in the U.S. per year.
“Avascular necrosis of the ankle, while a rare condition, is a serious and potentially debilitating one that causes pain and can lead to inhibited motion of the ankle joint, and in some cases, removal of part of the leg,” Capt. Raquel Peat, Ph.D., M.P.H., USPHS, director of the FDA’s Center for Devices and Radiological Health’s Office of Orthopedic Devices told the press. “[This] action provides patients with a treatment option that could potentially reduce pain, retain range of motion of their joint and improve quality of life.”
AVN is often caused by a sudden injury or sustained damage to the tissue, and it occurs when there is a lack of blood supply to bone tissue, causing it to become necrotic. Late-stage AVN of the ankle may result in the talus bone partially or fully collapsing. Current available treatments include fusing the joints in the foot and ankle together—a procedure which helps to alleviate pain caused by AVN but eliminates joint motion—or below-the-knee amputation.
The Patient Specific Talus Spacer is a 3D printed implant that can be used in talus replacement surgery. The talus spacer is made for each patient individually, modeled from computed tomography (CT) imaging, and is fitted to a patient’s specific anatomy. During the replacement surgery, the patient’s talus bone is removed and replaced with the implant, which is made from cobalt chromium alloy.
While fusion may become necessary in the future should the condition worsen, talus replacement surgery with the Patient Specific Talus Spacer is intended to be a joint-sparing procedure, as it allows the patient to retain motion in the ankle joint.
The FDA reviewed data for the Patient Specific Talus Spacer through the humanitarian device exemption (HDE) process. A Humanitarian Use Device (HUD) is a device that is intended to benefit patients by treating or diagnosing a disease or condition that affects not more than 8,000 individuals in the U.S. per year.