Sam Brusco, Associate Editor03.31.21
Premia Spine, a company aiming to change how debilitating chronic leg and back pain is treated, won Breakthrough Device Designation from the U.S. Food & Drug Administration for its TOPS facet arthroplasty system.
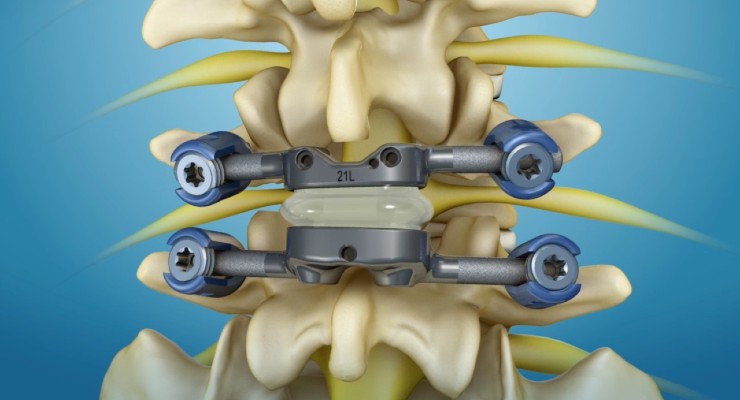
According to Premia, TOPS is the first and only lumbar spine facet joint replacement system. Developed to offer a new treatment option for spinal stenosis or spondylolisthesis, the system provides mobility, stability, and durability following decompression. TOPS has been proven in early clinical studies to deliver immediate and sustained pain relief and improvement in patient quality of life, and is currently the subject of a pivotal clinical trial.
“The TOPS System is designed to relieve debilitating lower back and leg pain while enabling long-term mobility, stability, and durability for patients,” Premia Spine CEO Ron Sacher told the press. “We are gratified that the FDA recognizes TOPS’s potential to advance the standard of care for patients with spinal stenosis and spondylolisthesis.”
Breakthrough designation expedites the development and FDA review of new devices with the potential to more effectively treat or diagnose life-threatening or irreversibly debilitating conditions. FDA recognition of the TOPS System as a Breakthrough Device means Premia Spine will receive feedback throughout the regulatory process, as well as prioritized review.
“TOPS has the potential to fill a void in our treatment armamentarium for spinal stenosis and spondylolisthesis. If the results of the pivotal study mirror those found in early clinical trials, TOPS could be a game changer for surgeons and their patients,” said Dr. Dom Coric, chief of neurosurgery at Carolinas Medical Center in Charlotte and lead investigator in the U.S. pivotal trial. “There's no other posterior arthroplasty device for the lumbar spine. Like most surgeons, I welcome the opportunity to advance spinal therapy with pioneering solutions like TOPS.”
According to Premia, TOPS is the first and only lumbar spine facet joint replacement system. Developed to offer a new treatment option for spinal stenosis or spondylolisthesis, the system provides mobility, stability, and durability following decompression. TOPS has been proven in early clinical studies to deliver immediate and sustained pain relief and improvement in patient quality of life, and is currently the subject of a pivotal clinical trial.
“The TOPS System is designed to relieve debilitating lower back and leg pain while enabling long-term mobility, stability, and durability for patients,” Premia Spine CEO Ron Sacher told the press. “We are gratified that the FDA recognizes TOPS’s potential to advance the standard of care for patients with spinal stenosis and spondylolisthesis.”
Breakthrough designation expedites the development and FDA review of new devices with the potential to more effectively treat or diagnose life-threatening or irreversibly debilitating conditions. FDA recognition of the TOPS System as a Breakthrough Device means Premia Spine will receive feedback throughout the regulatory process, as well as prioritized review.
“TOPS has the potential to fill a void in our treatment armamentarium for spinal stenosis and spondylolisthesis. If the results of the pivotal study mirror those found in early clinical trials, TOPS could be a game changer for surgeons and their patients,” said Dr. Dom Coric, chief of neurosurgery at Carolinas Medical Center in Charlotte and lead investigator in the U.S. pivotal trial. “There's no other posterior arthroplasty device for the lumbar spine. Like most surgeons, I welcome the opportunity to advance spinal therapy with pioneering solutions like TOPS.”