Sam Brusco, Associate Editor04.11.23
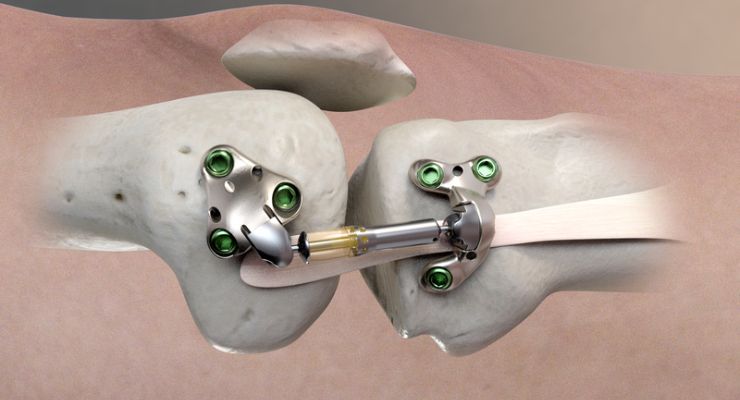
Moximed, a company working to improve the standard of care for patients with knee osteoarthritis (OA), has gained U.S. Food and Drug Administration (FDA) marketing authorization for its MISHA knee system, and implantable shock absorber (ISA) for the knee.
MISHA is indicated to treat those with knee OA who were unable to find relief from non-surgical or surgical treatment, continue to have pain that interferes with daily activities, and are ineligible for (or unwilling to undergo) joint replacement because of age or absence of advanced OA.
“I've been lucky enough to have been exposed to many major orthopedic innovations, most notably total knee arthroplasty (TKA). TKA is a phenomenal operation, but it has its limitations. I still see a clinical void between arthroscopic knee preservation and arthroplasty,” Anil Ranawat, MD, Chief, Hip and Knee Division of Sports Medicine Institute, Hospital for Special Surgery in New York, told the press. “With the FDA’s clearance of the MISHA Knee System, millions of people with symptomatic knee OA will now have an opportunity to achieve high levels of pain relief, enjoy the lifestyle and activities that are important to them, and preserve the option for a primary knee arthroplasty in the future. This system fills that void for both surgeons and patients.”
“Knee replacement is undoubtedly the gold standard for well-selected patients with severe OA, but for every patient I see that is ready for joint replacement, I see as many, if not more, who are not yet good arthroplasty candidates,” added William A. Jiranek, M.D., former president of the American Association of Hip and Knee Surgeons (AAHKS). “In my current role as Vice Chair for Practice Innovation at Duke University, I consider treatments for OA across the entire disease continuum, rather than focusing only on the end stage. As such, I was invited to provide an independent, non-investigator assessment of Moximed’s clinical study protocol, and I provided an independent, periodic review of the data during the study. I’m excited that the study met its superiority composite endpoint, which included effectiveness and safety components. This new joint-preserving treatment provides arthritis surgeons the ability to intervene earlier in the disease process to help patients not ready for a joint replacement.”

In its recent Calypso pivotal study, Moximed was able to demonstrate superiority of MISHA over high tibial osteotomy (HTO), a well-established surgery. The company used the established benefits of load reduction on knee joints to design the system, which is implanted in an outpatient-compatible procedure.
“This is a milestone event for knee OA sufferers, and it’s the result of unwavering clinical research and development that spans more than 10 years. We offer special thanks to our study patients and surgeon investigators who helped advance the understanding of this new treatment for OA. Also, we recognize the dedicated reviewers at FDA for completing their thorough benefit-risk assessment of our breakthrough technology. We’re thrilled to now be in a position to make the surgery available to patients,” said Anton Clifford, Ph.D., founder and CEO of Moximed. “We are committed to providing excellent medical education and customer service, supporting selection and treatment of indicated patients, and demonstrating scalability of our business as we introduce the MISHA Knee System to the U.S.”
MISHA is indicated to treat those with knee OA who were unable to find relief from non-surgical or surgical treatment, continue to have pain that interferes with daily activities, and are ineligible for (or unwilling to undergo) joint replacement because of age or absence of advanced OA.
“I've been lucky enough to have been exposed to many major orthopedic innovations, most notably total knee arthroplasty (TKA). TKA is a phenomenal operation, but it has its limitations. I still see a clinical void between arthroscopic knee preservation and arthroplasty,” Anil Ranawat, MD, Chief, Hip and Knee Division of Sports Medicine Institute, Hospital for Special Surgery in New York, told the press. “With the FDA’s clearance of the MISHA Knee System, millions of people with symptomatic knee OA will now have an opportunity to achieve high levels of pain relief, enjoy the lifestyle and activities that are important to them, and preserve the option for a primary knee arthroplasty in the future. This system fills that void for both surgeons and patients.”
“Knee replacement is undoubtedly the gold standard for well-selected patients with severe OA, but for every patient I see that is ready for joint replacement, I see as many, if not more, who are not yet good arthroplasty candidates,” added William A. Jiranek, M.D., former president of the American Association of Hip and Knee Surgeons (AAHKS). “In my current role as Vice Chair for Practice Innovation at Duke University, I consider treatments for OA across the entire disease continuum, rather than focusing only on the end stage. As such, I was invited to provide an independent, non-investigator assessment of Moximed’s clinical study protocol, and I provided an independent, periodic review of the data during the study. I’m excited that the study met its superiority composite endpoint, which included effectiveness and safety components. This new joint-preserving treatment provides arthritis surgeons the ability to intervene earlier in the disease process to help patients not ready for a joint replacement.”

In its recent Calypso pivotal study, Moximed was able to demonstrate superiority of MISHA over high tibial osteotomy (HTO), a well-established surgery. The company used the established benefits of load reduction on knee joints to design the system, which is implanted in an outpatient-compatible procedure.
“This is a milestone event for knee OA sufferers, and it’s the result of unwavering clinical research and development that spans more than 10 years. We offer special thanks to our study patients and surgeon investigators who helped advance the understanding of this new treatment for OA. Also, we recognize the dedicated reviewers at FDA for completing their thorough benefit-risk assessment of our breakthrough technology. We’re thrilled to now be in a position to make the surgery available to patients,” said Anton Clifford, Ph.D., founder and CEO of Moximed. “We are committed to providing excellent medical education and customer service, supporting selection and treatment of indicated patients, and demonstrating scalability of our business as we introduce the MISHA Knee System to the U.S.”