Michael Barbella, Managing Editor04.16.18
Kevin R. Stone, M.D., helped usher in the Age of Anabolics.
The San Francisco, Calif.-based orthopedic surgeon invented the world’s first orthopedic tissue regeneration template in 1986, giving hope to countless numbers of patients with torn knee cartilage. Debuting in 2000, Stone’s collagen meniscus implant (CMI) induces the body’s self- healing prowess to repair torn or missing segments of the knee’s main shock absorber. The implant is comprised of highly purified collagen, which acts as a trellis for new meniscus tissue growth.
Since gaining market admittance, Stone’s invention has helped repair more than 4,000 injured knees worldwide. Perhaps more importantly, though, it laid the groundwork for the cultivation of biological treatments for orthopedic injuries, a sector that was valued at $5 billion in 2015 and is expected to swell to $10.2 billion in 2025, according to Million Insights data. Driving this growth over the next seven years will be the planet’s aging population, technological advancements, changing (more active) lifestyles, and a steady increase in musculoskeletal-related conditions like arthritis and osteoporosis.
There will likely be no shortage of technologies available in 2025, as the orthobiologics field is already brimming with companies jockeying for market share. ODT’s January/February feature story “Biological Building Blocks” examines the trends and challenges shaping the orthobiologics market as well as some of the latest technologies available to patients. John Pracyk, M.D., Ph.D., franchise medical leader; Matthew D. Putnam, M.D., franchise medical director; and Tim Reed, director of biomaterials marketing; were among the DePuy Synthes Companies experts interviewed for the feature; their full input is provided in the following Q&A.
Michael Barbella: Please discuss the current trends in orthobiologics. What forces are driving these trends? Have they changed in recent years?
Tim Reed: As a company, we are consistently looking at the many ways we can help customers achieve their Triple Aim goals—improving patient care while reducing treatment costs and therefore improving patient satisfaction. Biomaterials play a role in helping to achieve these goals. In today’s environment biomaterials must be effective as customers are seeking the advantages of reducing OR time and getting patients ambulatory and out of the care facility sooner. Using a bone graft substitute instead of harvesting autograft from the patient’s iliac crest is a great example of this. ViviGen Cellular Bone Matrix, a differentiated cellular autograft for the repair or reconstruction of musculoskeletal defects that DePuy Synthes launched in collaboration with LifeNet Health, is a product that can be used in these types of cases. On a more micro-level, we’re seeing customers continue to demand less costly alternatives to BMP (bone morthogenetic protein) based products, driven by price sensitivity etc. This is why VivGen has been trending so well since its launch—it’s an innovative, natural solution that doesn’t involve recombinant proteins.
Barbella: What opportunities does this sector present to companies that operate in this space?
John Pracyk: Biomaterials are very frequently used in spine fusion procedures as well as in many other orthopedic surgeries, such as extremity cases and trauma procedures. In general, providers are often interested in working with manufacturers like DePuy Synthes and Johnson & Johnson Medical Devices Companies who can provide a full range of hardware fixation products and biomaterials solutions for all orthopedic and neurological spine surgeries across the continuum of care. Surgeons and providers rely on DePuy Synthes to deliver innovative and effective solutions, so bringing new bone graft substitutes like VivGen and ViviGen Formable Cellular Bone Matrix to market has enabled us to meet our existing customers’ evolving needs and also engage with new customers.
Barbella: What challenges/concerns are facing the orthobiologics industry? How can the industry and companies in this sector overcome these challenges?
Pracyk: Cost pressures and customer education are two of the biggest challenges we face. Continuing to research and innovate in this cost-sensitive environment is difficult, but we know there is a demand for solutions that meet the aspects of Triple Aim, and this is what keeps our teams motivated. Also, cost often needs to be considered for allograft-derived biomaterials due to the complexity and highly variable cost associated with donor recovery and processing. On the customer education side, the challenge is in helping our customers navigate the myriad of products available while ensuring our portfolio includes cost-effective solutions that can be used in less complex surgeries as well as the most challenging cases. For example, there must easily be over 100 different brands of bone void filler products on the market today, which are derived in the forms of simple allograft chips and ceramic granules to highly engineered cellular allografts, bioactive ceramics, and BMPs. It’s very challenging for customers to know the differences between products, why some are more expensive, and why certain products are better suited for particular procedures. As a leader in professional education, DePuy Synthes strives to reduce this complexity by making biomaterials an integral part of our curriculum across all orthopedic specialties and ensuring that all of our offerings have well-defined value propositions with accompanying explanations that can directly highlight the clinical benefits of each product.
Barbella: Why is it so difficult/challenging to mimic the body’s natural biological healing process for bone?
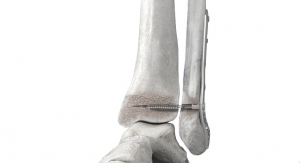
Matthew D. Putnam: The body is often able to heal itself with little or no help from biomaterials. In the case of very straightforward surgeries in healthy patients in which the native bone fragments are still intact, surgeons can use very simple bone void fillers such as chronOS Bone Graft Substitute or in some cases none at all. The main objective of these types of surgeries is to have the bony anatomy restored to its pre-injury or desired alignment, fill-in any small voids or gaps in the musculoskeletal system with a biomaterial, and then lock that down with hardware—from there the body does the rest. As the complexity of the fracture and local loss of blood supply increases with the possible added impact of co-morbidities, the chance of fracture healing decreases. In cases lacking healthy bone to build from, iliac crest autograft of VivGen becomes attractive because of its ability to provide all of the properties required for bone formation (osteoconductive, osteoinductive, osteogenic). When the bone area to be reformed is devoid of blood supply and/or infected, these issues become the primary concern and must be the first point of focus.
Barbella: What factors are currently driving innovation in orthobiologics?
Putnam: It continues to be important to meet the goals of the Triple Aim—improving outcomes while at the same time reducing costs, which is going to lead to healthier patients that give higher patient satisfaction scores to their physicians/providers. Customers are seeking these advantages and these types of products meet needs in terms of some combination of faster healing, improved OR efficiency, and ease of use.
Barbella: What interesting, new technologies are in the works?
Reed: We strong believe that there continues to be a great future for innovative allograft-derived implants, and there are some interesting developments in the area of bioactive synthetic bone void fillers as well. Biomaterials will continue to be important in helping to drive healing for patients and we will work to identify ways we can bring new innovation to market in this critical area.
The San Francisco, Calif.-based orthopedic surgeon invented the world’s first orthopedic tissue regeneration template in 1986, giving hope to countless numbers of patients with torn knee cartilage. Debuting in 2000, Stone’s collagen meniscus implant (CMI) induces the body’s self- healing prowess to repair torn or missing segments of the knee’s main shock absorber. The implant is comprised of highly purified collagen, which acts as a trellis for new meniscus tissue growth.
Since gaining market admittance, Stone’s invention has helped repair more than 4,000 injured knees worldwide. Perhaps more importantly, though, it laid the groundwork for the cultivation of biological treatments for orthopedic injuries, a sector that was valued at $5 billion in 2015 and is expected to swell to $10.2 billion in 2025, according to Million Insights data. Driving this growth over the next seven years will be the planet’s aging population, technological advancements, changing (more active) lifestyles, and a steady increase in musculoskeletal-related conditions like arthritis and osteoporosis.
There will likely be no shortage of technologies available in 2025, as the orthobiologics field is already brimming with companies jockeying for market share. ODT’s January/February feature story “Biological Building Blocks” examines the trends and challenges shaping the orthobiologics market as well as some of the latest technologies available to patients. John Pracyk, M.D., Ph.D., franchise medical leader; Matthew D. Putnam, M.D., franchise medical director; and Tim Reed, director of biomaterials marketing; were among the DePuy Synthes Companies experts interviewed for the feature; their full input is provided in the following Q&A.
Michael Barbella: Please discuss the current trends in orthobiologics. What forces are driving these trends? Have they changed in recent years?
Tim Reed: As a company, we are consistently looking at the many ways we can help customers achieve their Triple Aim goals—improving patient care while reducing treatment costs and therefore improving patient satisfaction. Biomaterials play a role in helping to achieve these goals. In today’s environment biomaterials must be effective as customers are seeking the advantages of reducing OR time and getting patients ambulatory and out of the care facility sooner. Using a bone graft substitute instead of harvesting autograft from the patient’s iliac crest is a great example of this. ViviGen Cellular Bone Matrix, a differentiated cellular autograft for the repair or reconstruction of musculoskeletal defects that DePuy Synthes launched in collaboration with LifeNet Health, is a product that can be used in these types of cases. On a more micro-level, we’re seeing customers continue to demand less costly alternatives to BMP (bone morthogenetic protein) based products, driven by price sensitivity etc. This is why VivGen has been trending so well since its launch—it’s an innovative, natural solution that doesn’t involve recombinant proteins.
Barbella: What opportunities does this sector present to companies that operate in this space?
John Pracyk: Biomaterials are very frequently used in spine fusion procedures as well as in many other orthopedic surgeries, such as extremity cases and trauma procedures. In general, providers are often interested in working with manufacturers like DePuy Synthes and Johnson & Johnson Medical Devices Companies who can provide a full range of hardware fixation products and biomaterials solutions for all orthopedic and neurological spine surgeries across the continuum of care. Surgeons and providers rely on DePuy Synthes to deliver innovative and effective solutions, so bringing new bone graft substitutes like VivGen and ViviGen Formable Cellular Bone Matrix to market has enabled us to meet our existing customers’ evolving needs and also engage with new customers.
Barbella: What challenges/concerns are facing the orthobiologics industry? How can the industry and companies in this sector overcome these challenges?
Pracyk: Cost pressures and customer education are two of the biggest challenges we face. Continuing to research and innovate in this cost-sensitive environment is difficult, but we know there is a demand for solutions that meet the aspects of Triple Aim, and this is what keeps our teams motivated. Also, cost often needs to be considered for allograft-derived biomaterials due to the complexity and highly variable cost associated with donor recovery and processing. On the customer education side, the challenge is in helping our customers navigate the myriad of products available while ensuring our portfolio includes cost-effective solutions that can be used in less complex surgeries as well as the most challenging cases. For example, there must easily be over 100 different brands of bone void filler products on the market today, which are derived in the forms of simple allograft chips and ceramic granules to highly engineered cellular allografts, bioactive ceramics, and BMPs. It’s very challenging for customers to know the differences between products, why some are more expensive, and why certain products are better suited for particular procedures. As a leader in professional education, DePuy Synthes strives to reduce this complexity by making biomaterials an integral part of our curriculum across all orthopedic specialties and ensuring that all of our offerings have well-defined value propositions with accompanying explanations that can directly highlight the clinical benefits of each product.
Barbella: Why is it so difficult/challenging to mimic the body’s natural biological healing process for bone?
Matthew D. Putnam: The body is often able to heal itself with little or no help from biomaterials. In the case of very straightforward surgeries in healthy patients in which the native bone fragments are still intact, surgeons can use very simple bone void fillers such as chronOS Bone Graft Substitute or in some cases none at all. The main objective of these types of surgeries is to have the bony anatomy restored to its pre-injury or desired alignment, fill-in any small voids or gaps in the musculoskeletal system with a biomaterial, and then lock that down with hardware—from there the body does the rest. As the complexity of the fracture and local loss of blood supply increases with the possible added impact of co-morbidities, the chance of fracture healing decreases. In cases lacking healthy bone to build from, iliac crest autograft of VivGen becomes attractive because of its ability to provide all of the properties required for bone formation (osteoconductive, osteoinductive, osteogenic). When the bone area to be reformed is devoid of blood supply and/or infected, these issues become the primary concern and must be the first point of focus.
Barbella: What factors are currently driving innovation in orthobiologics?
Putnam: It continues to be important to meet the goals of the Triple Aim—improving outcomes while at the same time reducing costs, which is going to lead to healthier patients that give higher patient satisfaction scores to their physicians/providers. Customers are seeking these advantages and these types of products meet needs in terms of some combination of faster healing, improved OR efficiency, and ease of use.
Barbella: What interesting, new technologies are in the works?
Reed: We strong believe that there continues to be a great future for innovative allograft-derived implants, and there are some interesting developments in the area of bioactive synthetic bone void fillers as well. Biomaterials will continue to be important in helping to drive healing for patients and we will work to identify ways we can bring new innovation to market in this critical area.