Joe Randolph, President & CEO, The Innovation Institute02.19.19
It takes a specific type of know-how to determine which ideas and opportunities are worth investing in. This means professionals who have medical sense, financial finesse, and marketing expertise must determine if the market is ready for a new product, solution, or breakthrough. One might expect that everyone would be interested in a new medical product that improves patient care, but it’s not that simple.
For example, take a look at the Conforming Thermal Pack. We all agree that it’s a brilliant invention conceived by a NASA astronaut/physician. It’s a superior cold/hot therapy product that conforms to the injured joint, holds the right temperature and for the appropriate amount of time. It took someone like Dr. Scott Parazynski, a determined NASA astronaut/medical doctor who experienced the frustrations of healing his own injury, to see that a medical product doesn’t have to be just adequate. With iterative prototyping and rigorous evaluation, he developed a product that combines a proprietary shape and fill chemistry to improve thermal transfer and avoid unwanted condensation to deliver optimal therapeutic temperature for the appropriate amount of time. Because of the ingenuity and perseverance of Dr. Parazynski and other innovators, we are breaking through with new and exciting medical products.
The challenge now is to show others they don’t need to accept an inferior solution. We need to reach physicians and their patients to demonstrate to them that the next generation hot and cold therapy product is here and ready to be used now, and it’s affordable.
When determining whether to move forward on a potential breakthrough product, we are tasked with evaluating and testing each prototype for function, feasibility, safety, aesthetics, and other criteria. Often, modifications and tweaks are made until the product passes all the tests that the Innovation Lab team has constructed based on their evaluation and vetting expertise.
The next step is to communicate the solution to an audience that understands the problem and whether a breakthrough solution would be embraced. Once there are statistics and research that support the need for a new product, the Lab team gathers more feedback and refines the design until a consensus is reached to move forward with development and commercialization.
As one would guess, all these steps require staffing and funding. Taking a product to market can be expensive. Therefore, we often focus on partnering with organizations that are in the same category as our particular product through licensing or distribution arrangements.
We are currently selling our Lonnie Brace product through LabCo, Innovation Lab’s subsidiary for distributing medical goods. We are also actively seeking a larger commercial partner to license the product. The product aims to reduce pain associated with tendinitis and other injuries plaguing the elbow area. It was invented by Stephen Marino, MD, a neurologist—another inventor who saw the need based on his own experience.
The WingSling, developed by Mark Elzik, MD, an orthopedic surgeon, is now sold by CVS Health, one of the top pharmacy chains in the nation as a private label product. It is doing well. A patient from one of the health systems we are affiliated with actually tracked down the product online when she was looking for a solution to help elevate her wrist, which had been fractured, to decrease swelling and pain. Turns out that Dr. Elzik’s invention was exactly what she needed. She raved about it and shared her experience with ABC News. This kind of visibility with user endorsement is helpful to promote the product and help more people.
We’re excited about what we have in our pipeline for 2019. We are especially pleased with how we have maximized our reach with RedCrow and other pertinent partners. RedCrow’s founder is familiar with crowdfunding and happens to have been a Talking Heads band member. Jerry Harrison is not only a musician; he is an innovator and entrepreneur who previously helped raise funds for promising bands through his company GarageBand.com. It was praised as being the world’s “largest online community for independent musicians.” Now as co-founder for RedCrow, he helps raise funds for medical products he believes in. How lucky we are to be working with Harrison and his dynamic team.
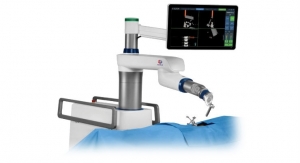
One product we are raising funds for with RedCrow is the Centerline Biomedical Intra-Operative Positioning System (IOPS), a platform technology that uses low-intensity electromagnetic tracking. The system reduces the need for fluoroscopy and minimizes the harmful effects of radiation. With its 3D enhanced visualization and GPS-like real-time navigation, surgeons can perform procedures with precision and control for accurate device placement, simplified complex procedures (and potentially decreased secondary procedures including costly re-interventions), and reduced radiation exposure for patients and healthcare professionals.
By making procedures more efficient and predictable, hospitals can perform procedures with higher profit margins, increase utilization of their operating rooms, drive revenue, and realize a greater profit. In the future, IOPS aims to reduce the costs of robotic or autonomous surgeries (a market estimated at $16.91 billion in 2021) for several applications by directly linking the patient’s anatomy to the robotic system.
This brings me back full circle to exactly where we began, as an organization founded in 2013, to focus on doing more, with less, for more people. We have been doing this by listening closely to the needs of innovators and major health systems and taking action through innovation.
Our measurement of success is numbers that show reduced costs for health systems and their patients; increased revenues for health systems and individuals through innovation implementation; and improved patient care that results from innovation. These numbers will improve year over year because there is so much need. There is always room for improvement, so we will continue to be called upon to do what we do.
For example, take a look at the Conforming Thermal Pack. We all agree that it’s a brilliant invention conceived by a NASA astronaut/physician. It’s a superior cold/hot therapy product that conforms to the injured joint, holds the right temperature and for the appropriate amount of time. It took someone like Dr. Scott Parazynski, a determined NASA astronaut/medical doctor who experienced the frustrations of healing his own injury, to see that a medical product doesn’t have to be just adequate. With iterative prototyping and rigorous evaluation, he developed a product that combines a proprietary shape and fill chemistry to improve thermal transfer and avoid unwanted condensation to deliver optimal therapeutic temperature for the appropriate amount of time. Because of the ingenuity and perseverance of Dr. Parazynski and other innovators, we are breaking through with new and exciting medical products.
The challenge now is to show others they don’t need to accept an inferior solution. We need to reach physicians and their patients to demonstrate to them that the next generation hot and cold therapy product is here and ready to be used now, and it’s affordable.
When determining whether to move forward on a potential breakthrough product, we are tasked with evaluating and testing each prototype for function, feasibility, safety, aesthetics, and other criteria. Often, modifications and tweaks are made until the product passes all the tests that the Innovation Lab team has constructed based on their evaluation and vetting expertise.
The next step is to communicate the solution to an audience that understands the problem and whether a breakthrough solution would be embraced. Once there are statistics and research that support the need for a new product, the Lab team gathers more feedback and refines the design until a consensus is reached to move forward with development and commercialization.
As one would guess, all these steps require staffing and funding. Taking a product to market can be expensive. Therefore, we often focus on partnering with organizations that are in the same category as our particular product through licensing or distribution arrangements.
We are currently selling our Lonnie Brace product through LabCo, Innovation Lab’s subsidiary for distributing medical goods. We are also actively seeking a larger commercial partner to license the product. The product aims to reduce pain associated with tendinitis and other injuries plaguing the elbow area. It was invented by Stephen Marino, MD, a neurologist—another inventor who saw the need based on his own experience.
The WingSling, developed by Mark Elzik, MD, an orthopedic surgeon, is now sold by CVS Health, one of the top pharmacy chains in the nation as a private label product. It is doing well. A patient from one of the health systems we are affiliated with actually tracked down the product online when she was looking for a solution to help elevate her wrist, which had been fractured, to decrease swelling and pain. Turns out that Dr. Elzik’s invention was exactly what she needed. She raved about it and shared her experience with ABC News. This kind of visibility with user endorsement is helpful to promote the product and help more people.
We’re excited about what we have in our pipeline for 2019. We are especially pleased with how we have maximized our reach with RedCrow and other pertinent partners. RedCrow’s founder is familiar with crowdfunding and happens to have been a Talking Heads band member. Jerry Harrison is not only a musician; he is an innovator and entrepreneur who previously helped raise funds for promising bands through his company GarageBand.com. It was praised as being the world’s “largest online community for independent musicians.” Now as co-founder for RedCrow, he helps raise funds for medical products he believes in. How lucky we are to be working with Harrison and his dynamic team.
One product we are raising funds for with RedCrow is the Centerline Biomedical Intra-Operative Positioning System (IOPS), a platform technology that uses low-intensity electromagnetic tracking. The system reduces the need for fluoroscopy and minimizes the harmful effects of radiation. With its 3D enhanced visualization and GPS-like real-time navigation, surgeons can perform procedures with precision and control for accurate device placement, simplified complex procedures (and potentially decreased secondary procedures including costly re-interventions), and reduced radiation exposure for patients and healthcare professionals.
By making procedures more efficient and predictable, hospitals can perform procedures with higher profit margins, increase utilization of their operating rooms, drive revenue, and realize a greater profit. In the future, IOPS aims to reduce the costs of robotic or autonomous surgeries (a market estimated at $16.91 billion in 2021) for several applications by directly linking the patient’s anatomy to the robotic system.
This brings me back full circle to exactly where we began, as an organization founded in 2013, to focus on doing more, with less, for more people. We have been doing this by listening closely to the needs of innovators and major health systems and taking action through innovation.
Our measurement of success is numbers that show reduced costs for health systems and their patients; increased revenues for health systems and individuals through innovation implementation; and improved patient care that results from innovation. These numbers will improve year over year because there is so much need. There is always room for improvement, so we will continue to be called upon to do what we do.