Michael Barbella, Managing Editor06.24.19
Tony Finau has learned quite a lot about himself over the past 12 months. Besides gaining new insights into his fortitude, humility, and confidence, the 29-year-old professional golfer discovered a keen sense of humor he perhaps never before realized (or cared to admit).
The latter trait was a particular surprise to Finau’s fans, considering the clever manner in which it was divulged.
Shortly before the 2019 Master’s Tournament, Nike and Finau teamed up to create a signature shoe he could wear at the year’s first major should the 6-foot, 4-inch Samoan decide to partake in another disastrous hole-in-one victory run (Finau injured his left ankle during last year’s Master’s celebrating an ace at the seventh hole). Debuting on April Fool’s Day, the fairway-colored, incredibly high-topped Finau1 boot came with a three-and-a-half minute “mockumentary” that poked fun at Finau and his injury.
“Not a lot of people come back from that—not the injury but the embarrassment,” Carl Madore, a Nike “footwear designer,” quips in the video. Finau adds further comic relief with lines such as, “Like any elite athlete, I had to get back in the lab and rebuild my body from ground up,” and “I thought to myself, ‘What if we designed a golf shoe for stability in my ankle? Why not?’”
“I felt like if you can’t laugh at yourself in something like that, then I think you’re doing it all wrong,” Finau told Star Tribune (Minneapolis) sports columnist Jim Souhan at the start of the Master’s. “I thought it was quite funny after the fact, celebrating a hole-in-one and ruining my ankle for a few months.”
Few in Finau’s inner circle were laughing after the mishap last spring, though. Thankfully, however, Finau sustained only soft tissue damage and some torn ligaments; he rebounded the following day to shoot a four-under-par 68 in the opening round and briefly led the tournament before slipping behind three-time major winner Jordan Spieth to tie for second place with Matthew Kuchar. He finished the contest at T10, winning $286,000.
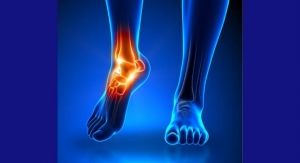
Although Finau now makes light of his injury, ankle sprains are no laughing matter: Studies have shown such traumas can have lasting consequences, leading to joint damage, chronic instability, and ACL troubles. Sprains have become one of the most common sports injuries and demand for non-invasive, simple solutions is expected to drive the global foot and ankle treatment market over the next half-decade. Other factors like rising osteoporosis rates and an aging world population also will contribute to the growth.
ODT’s May/June feature “A Little Extreme” examines the innovation being developed in this burgeoning sector (and the overall worldwide extremities market) that eventually will contribute to its solid growth over the next half-decade.
Robert T. Davis Jr., corporate vice president and president, Orthopedics and Tissue Technologies for Integra LifeSciences, a global provider of regenerative technologies, neurosurgical and extremity orthopedic solutions, was interviewed for the story; his input in the following Q&A, provides an addendum to the feature.
Michael Barbella: What trends/factors are currently impacting the global extremities market?
Robert T. Davis Jr.: The global extremities market has seen a rise in higher expectations among the aging population on post-operative function and the ability to return to their normal, active lifestyle.
Barbella: What factors are driving growth in this market? What segment is the growth most prolific—upper extremity, lower extremity, small joint (fingers, toes), etc., or is it fairly even across the board?
Davis: The shoulder and ankle replacement market are fast-growing segments in this space, with ankle growing in the low double-digits. This growth is driven in part by more companies entering the total ankle replacement space and funneling resources into innovation and peer lead surgeon education. The volume of ankle replacement procedures per year is comparatively low to a knee or hip replacement. This factor can lend itself to a longer learning curve for the procedure. Hands-on cadaveric training programs for surgeons have reduced this learning curve and expanded the procedural volume. The introduction of revision solutions in both the ankle and shoulder market is giving surgeons more tools for future options for their younger patients. The continued adoption of reverse shoulder arthroplasty is also driving growth in this market. With more published studies on reverse as a viable choice for bad humeral head fractures, more surgeons are starting to use this higher average selling price (ASP) solution.
Barbella: What types of extremity solutions are popular with surgeons? Why are they popular?
Davis: Bone-sparing and convertible solutions continue to attract interest from surgeons as they allow for greater preservation of the patient’s anatomy and easier future revision pathways.
Barbella: What makes the extremity market an attractive alternative to the large joint and spine sectors? Why is this one of the fastest-growing areas of orthopedics?
Davis: In some respects, the extremities market is where the hip and knee markets were over a decade ago. There are three key reasons why the extremity market is an attractive alternative: 1) the extremities market is not as crowded with competitors as the large joint and spine sectors, 2) there are still opportunities for innovation, and 3) it’s attractive to surgeons who are looking to specialize within a practice. As we see more technological advancements in this sector, options like reverse shoulders and revision ankle prostheses have increased both the number of surgeons performing these procedures, as well as the indications and patient population.
Barbella: What new innovations/technologies are in the works? What technologies hold the most promise for patients? For market growth?
Davis: The market is seeing investments in research for new alternatives in joint replacement such as pyrocarbon and 3D printed implants. These new innovations provide solutions to patients who may fall outside the average age range for a traditional joint replacement, or have unique anatomy or significant bone loss. New materials have the potential to increase the longevity of the lifetime of the prosthesis, thus opening up the market to younger patients, where surgeons may have previously held off on performing a joint replacement due to concerns of future revisions. The market is starting to see trends with the introduction of 3-D printed implants that can be customized specific to a patient’s CT scans. This opens the door to more options during revision surgery, offering the patient a custom implant component in some cases, instead of going straight to a fusion.
Barbella: How does your company stay innovative in this increasingly crowded market? How do you differentiate yourself from other players in the sector?
Davis: We tend to focus our innovation based on what our customers are telling us, around their unmet needs and opportunities. When developing our portfolio pipeline, Integra works closely with customers to identify pain points in their surgical practice and areas where they see opportunity to improve patient outcomes. Sometimes innovation means making a procedure more simple and reproducible for the surgeon and their OR team. Integra is unique in that it offers a broad portfolio in extremity orthopedics as well as regenerative technology such as nerve conduits, skin substitutes, and amniotic solutions for the extremity surgeon.
We also have a strong focus on peer-to-peer learning and partnerships with design surgeons to advance technology. We have a 56,000-square-foot Austin, Texas, Center of Excellence (COE) where we have incorporated a state-of-the-art product development lab, skills and training area designed to maximize the overall customer experience. This COE is one of Integra’s hubs for innovation, attracting leading orthopedic surgeons from all over the world. Since the launch of this new COE, we have greatly expanded our educational offerings to train surgeons on the safe and efficacious use of Integra products.
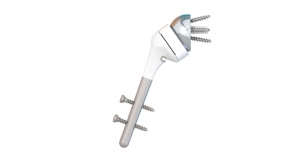
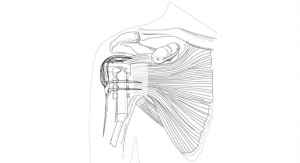
Last year, we launched innovative new products such as the Integra XT Revision Total Ankle Replacement System, and expanded the commercial availability of Cadence Total Ankle System, our ankle prosthesis developed in partnership with world-leading foot and ankle surgeons. In addition, we launched the Integra Panta 2 TTC Arthrodesis Nail System for tibiotalo-calcaneal fusion of the ankle due to severe arthritis. We strengthened our shoulder portfolio with the full market release of the Integra Titan Reverse Shoulder System-S and are collaborating with the Consortium of Focused Orthopedists to develop a short-stem and stemless shoulder system.
In 2018, we made significant investments with our channel expansion and created a dedicated sales channel for extremity orthopedics to increase our presence in the operating room and leverage our expertise to go deeper clinically with our orthopedic technologies. We’ve increased our capacity and changed our product mix to create more focus and drive efficiencies in a two-tier sales model; with specialized distributors focused on shoulder arthroplasty and upper extremity implant solutions, direct sales specialists focused on our leading ankle arthroplasty and nerve and tendon products with additional associate sales representatives to increase our presence in the OR. We are also leveraging the broader footprint of Integra to drive access with contracting including enterprise and government sales.
With our focused extremity orthopedics channel and new product introductions, we saw healthy growth rates with our shoulder and ankle portfolio. We also have a rich heritage in peripheral nerve repair and are excited about new product introductions to help improve patient outcomes.
Barbella: How is product development in extremities different than that in large joint or spine?
Davis: There are still a lot of opportunities to drive innovation in extremity orthopedics and adopt technologies from the large joint and spine space. There are still so many unanswered questions and areas for improvement. For example, whereas larger joint and spine may have lower revision rates, extremities is trying to solve problems that still don’t have a solution. In shoulder, there is still not an answer on the market for the glenoid, loosening in total shoulders and wearing in a hemi arthroplasty. Solving the problems of the glenoid will increase longevity of implants and improve shoulder patient outcomes. We also have to approach the problem and the verification differently. In some cases, standards for performance have not been created so we are not only innovating in design but also setting new standards on how extremity devices should perform.
Barbella: How is value-based healthcare and market forces like robotics, 3D printing, and computer-aided surgery impacting extremities technologies and product development?
Davis: 3-D printing, PSI/patient specific instrumentation, computer-aided navigation technology, along with patient planning surgical software are driving opportunities for better accuracy during surgery, and perception of a more reproducible procedure. These technologies can give surgeons tools to decrease the learning curve, and allow them to address more complex revision surgeries. Value-based healthcare is driving the need for surgeries to be more efficient. Integra designed the Cadence total ankle system instrumentation with this in mind, to simplify the surgical procedure allowing the potential to reduce OR time and associated costs to both the facility and patient.
Barbella: How do you expect the extremities market to evolve over the next half decade?
Davis: The mix in procedures will likely shift and see a higher rate of revision surgeries, as the market offers solutions for younger patients and innovation in revision designs. Medical education and social media are increasing the awareness of advancements in technology in this space.
**Data Sources: SmartTrak
The latter trait was a particular surprise to Finau’s fans, considering the clever manner in which it was divulged.
Shortly before the 2019 Master’s Tournament, Nike and Finau teamed up to create a signature shoe he could wear at the year’s first major should the 6-foot, 4-inch Samoan decide to partake in another disastrous hole-in-one victory run (Finau injured his left ankle during last year’s Master’s celebrating an ace at the seventh hole). Debuting on April Fool’s Day, the fairway-colored, incredibly high-topped Finau1 boot came with a three-and-a-half minute “mockumentary” that poked fun at Finau and his injury.
“Not a lot of people come back from that—not the injury but the embarrassment,” Carl Madore, a Nike “footwear designer,” quips in the video. Finau adds further comic relief with lines such as, “Like any elite athlete, I had to get back in the lab and rebuild my body from ground up,” and “I thought to myself, ‘What if we designed a golf shoe for stability in my ankle? Why not?’”
“I felt like if you can’t laugh at yourself in something like that, then I think you’re doing it all wrong,” Finau told Star Tribune (Minneapolis) sports columnist Jim Souhan at the start of the Master’s. “I thought it was quite funny after the fact, celebrating a hole-in-one and ruining my ankle for a few months.”
Few in Finau’s inner circle were laughing after the mishap last spring, though. Thankfully, however, Finau sustained only soft tissue damage and some torn ligaments; he rebounded the following day to shoot a four-under-par 68 in the opening round and briefly led the tournament before slipping behind three-time major winner Jordan Spieth to tie for second place with Matthew Kuchar. He finished the contest at T10, winning $286,000.
Although Finau now makes light of his injury, ankle sprains are no laughing matter: Studies have shown such traumas can have lasting consequences, leading to joint damage, chronic instability, and ACL troubles. Sprains have become one of the most common sports injuries and demand for non-invasive, simple solutions is expected to drive the global foot and ankle treatment market over the next half-decade. Other factors like rising osteoporosis rates and an aging world population also will contribute to the growth.
ODT’s May/June feature “A Little Extreme” examines the innovation being developed in this burgeoning sector (and the overall worldwide extremities market) that eventually will contribute to its solid growth over the next half-decade.
Robert T. Davis Jr., corporate vice president and president, Orthopedics and Tissue Technologies for Integra LifeSciences, a global provider of regenerative technologies, neurosurgical and extremity orthopedic solutions, was interviewed for the story; his input in the following Q&A, provides an addendum to the feature.
Michael Barbella: What trends/factors are currently impacting the global extremities market?
Robert T. Davis Jr.: The global extremities market has seen a rise in higher expectations among the aging population on post-operative function and the ability to return to their normal, active lifestyle.
Barbella: What factors are driving growth in this market? What segment is the growth most prolific—upper extremity, lower extremity, small joint (fingers, toes), etc., or is it fairly even across the board?
Davis: The shoulder and ankle replacement market are fast-growing segments in this space, with ankle growing in the low double-digits. This growth is driven in part by more companies entering the total ankle replacement space and funneling resources into innovation and peer lead surgeon education. The volume of ankle replacement procedures per year is comparatively low to a knee or hip replacement. This factor can lend itself to a longer learning curve for the procedure. Hands-on cadaveric training programs for surgeons have reduced this learning curve and expanded the procedural volume. The introduction of revision solutions in both the ankle and shoulder market is giving surgeons more tools for future options for their younger patients. The continued adoption of reverse shoulder arthroplasty is also driving growth in this market. With more published studies on reverse as a viable choice for bad humeral head fractures, more surgeons are starting to use this higher average selling price (ASP) solution.
Barbella: What types of extremity solutions are popular with surgeons? Why are they popular?
Davis: Bone-sparing and convertible solutions continue to attract interest from surgeons as they allow for greater preservation of the patient’s anatomy and easier future revision pathways.
Barbella: What makes the extremity market an attractive alternative to the large joint and spine sectors? Why is this one of the fastest-growing areas of orthopedics?
Davis: In some respects, the extremities market is where the hip and knee markets were over a decade ago. There are three key reasons why the extremity market is an attractive alternative: 1) the extremities market is not as crowded with competitors as the large joint and spine sectors, 2) there are still opportunities for innovation, and 3) it’s attractive to surgeons who are looking to specialize within a practice. As we see more technological advancements in this sector, options like reverse shoulders and revision ankle prostheses have increased both the number of surgeons performing these procedures, as well as the indications and patient population.
Barbella: What new innovations/technologies are in the works? What technologies hold the most promise for patients? For market growth?
Davis: The market is seeing investments in research for new alternatives in joint replacement such as pyrocarbon and 3D printed implants. These new innovations provide solutions to patients who may fall outside the average age range for a traditional joint replacement, or have unique anatomy or significant bone loss. New materials have the potential to increase the longevity of the lifetime of the prosthesis, thus opening up the market to younger patients, where surgeons may have previously held off on performing a joint replacement due to concerns of future revisions. The market is starting to see trends with the introduction of 3-D printed implants that can be customized specific to a patient’s CT scans. This opens the door to more options during revision surgery, offering the patient a custom implant component in some cases, instead of going straight to a fusion.
Barbella: How does your company stay innovative in this increasingly crowded market? How do you differentiate yourself from other players in the sector?
Davis: We tend to focus our innovation based on what our customers are telling us, around their unmet needs and opportunities. When developing our portfolio pipeline, Integra works closely with customers to identify pain points in their surgical practice and areas where they see opportunity to improve patient outcomes. Sometimes innovation means making a procedure more simple and reproducible for the surgeon and their OR team. Integra is unique in that it offers a broad portfolio in extremity orthopedics as well as regenerative technology such as nerve conduits, skin substitutes, and amniotic solutions for the extremity surgeon.
We also have a strong focus on peer-to-peer learning and partnerships with design surgeons to advance technology. We have a 56,000-square-foot Austin, Texas, Center of Excellence (COE) where we have incorporated a state-of-the-art product development lab, skills and training area designed to maximize the overall customer experience. This COE is one of Integra’s hubs for innovation, attracting leading orthopedic surgeons from all over the world. Since the launch of this new COE, we have greatly expanded our educational offerings to train surgeons on the safe and efficacious use of Integra products.
Last year, we launched innovative new products such as the Integra XT Revision Total Ankle Replacement System, and expanded the commercial availability of Cadence Total Ankle System, our ankle prosthesis developed in partnership with world-leading foot and ankle surgeons. In addition, we launched the Integra Panta 2 TTC Arthrodesis Nail System for tibiotalo-calcaneal fusion of the ankle due to severe arthritis. We strengthened our shoulder portfolio with the full market release of the Integra Titan Reverse Shoulder System-S and are collaborating with the Consortium of Focused Orthopedists to develop a short-stem and stemless shoulder system.
In 2018, we made significant investments with our channel expansion and created a dedicated sales channel for extremity orthopedics to increase our presence in the operating room and leverage our expertise to go deeper clinically with our orthopedic technologies. We’ve increased our capacity and changed our product mix to create more focus and drive efficiencies in a two-tier sales model; with specialized distributors focused on shoulder arthroplasty and upper extremity implant solutions, direct sales specialists focused on our leading ankle arthroplasty and nerve and tendon products with additional associate sales representatives to increase our presence in the OR. We are also leveraging the broader footprint of Integra to drive access with contracting including enterprise and government sales.
With our focused extremity orthopedics channel and new product introductions, we saw healthy growth rates with our shoulder and ankle portfolio. We also have a rich heritage in peripheral nerve repair and are excited about new product introductions to help improve patient outcomes.
Barbella: How is product development in extremities different than that in large joint or spine?
Davis: There are still a lot of opportunities to drive innovation in extremity orthopedics and adopt technologies from the large joint and spine space. There are still so many unanswered questions and areas for improvement. For example, whereas larger joint and spine may have lower revision rates, extremities is trying to solve problems that still don’t have a solution. In shoulder, there is still not an answer on the market for the glenoid, loosening in total shoulders and wearing in a hemi arthroplasty. Solving the problems of the glenoid will increase longevity of implants and improve shoulder patient outcomes. We also have to approach the problem and the verification differently. In some cases, standards for performance have not been created so we are not only innovating in design but also setting new standards on how extremity devices should perform.
Barbella: How is value-based healthcare and market forces like robotics, 3D printing, and computer-aided surgery impacting extremities technologies and product development?
Davis: 3-D printing, PSI/patient specific instrumentation, computer-aided navigation technology, along with patient planning surgical software are driving opportunities for better accuracy during surgery, and perception of a more reproducible procedure. These technologies can give surgeons tools to decrease the learning curve, and allow them to address more complex revision surgeries. Value-based healthcare is driving the need for surgeries to be more efficient. Integra designed the Cadence total ankle system instrumentation with this in mind, to simplify the surgical procedure allowing the potential to reduce OR time and associated costs to both the facility and patient.
Barbella: How do you expect the extremities market to evolve over the next half decade?
Davis: The mix in procedures will likely shift and see a higher rate of revision surgeries, as the market offers solutions for younger patients and innovation in revision designs. Medical education and social media are increasing the awareness of advancements in technology in this space.
**Data Sources: SmartTrak