08.09.16
$2.9 Billion
KEY EXECUTIVES:
Omar Ishrak, Chairman and CEO
Karen L. Parkill, Exec. VP and Chief Financial Officer
Richard E. Kuntz, M.D., M.SC., Sr. VP and Chief Scientific, Clinical, and Regulatory Officer
Luann Pendy, Sr. VP, Global Quality
Mike Genau, Sr. VP and President, Americas Region
Robert ten Hoedt, Exec. VP and President, EMEAC
Bob White, Sr. VP and President, Asia Pacific Region
Chris Lee, Sr. VP and President, Greater China Region
Geoffrey S. Martha, Exec. VP and Group President, Restorative Therapies Group
NUMBER OF EMPLOYEES: 5,000
HEADQUARTERS: Memphis, Tenn.
It’s going to be difficult, if not downright impossible, for Medtronic plc to top its 2015 fiscal year. During that historic 12-month period, the company purchased Covidien plc for a record $50 billion, creating the world’s second-largest medical device company (Johnson & Johnson still leads, even with slumping device revenue) and sparking a national debate over the deal’s structure. Under terms of the all cash-and-stock purchase, Medtronic redomiciled overseas, reducing its corporate tax rate in a controversial practice known as an inversion. The move was among a slew of inverted deals that eventually prompted the U.S. Treasury Department to impose limits on re-incorporations and restrict related-party debt for American subsidiaries.
Besides trimming its tax rate, the blockbuster merger enabled Medtronic to increase its fiscal year revenue by 19 percent and helped the company become more diversified and balanced, given Covidien’s focus on surgical tools and hospital-based technologies. It also expanded Medtronic’s exposure to smaller cities in emerging markets, which currently constitute 12.7 percent of total revenue, up slightly from 12.3 percent in FY14. And it better positions the company to lead the push toward value-based healthcare by integrating patient care services that balance cost and access challenges.
“[Omar] Ishrak changed the ethos of the company, expanding it from a device maker into one that also offers services and partners with healthcare providers,” Raj Denhoy, a medical technology research analyst at Jefferies LLC told Barron’s last fall. “It was a bold move.”
And a necessary one as well. With the U.S. healthcare industry favoring risk- and value-based business models over traditional fee-for-service systems (thanks to Obamacare), medtech companies are increasingly reducing their dependence solely on medical devices to sustain growth. At Medtronic, that newfound freedom has inspired collaborations with hospital systems, payers, governments, and companies to develop integrated health solutions that complement and enhance product value through traditional wraparound services and solutions.
In FY15, those solutions targeted diabetes and patient home monitoring. Medtronic partnered with both Sanofi S.A. and IBM on diabetes treatment and management, leveraging its devices and care management products with its cohorts’ existing solutions to improve patient outcomes. The company is matching its insulin pumps and glucose monitoring technology with Sanofi’s insulin products to ameliorate type 2 diabetes management, particularly for patients having trouble controlling their blood sugar levels. The pair has been battling type 1 diabetes with a similar agreement, offering an implantable insulin delivery system to European patients.
Medtronic and Sanofi also are working together on care management services, developing an education plan for type 2 diabetics who cannot control their glucose levels through medication. The program will guide these patients through the initial phase of insulin treatment.
Medtronic’s hookup with IBM aims to improve outcomes as well, albeit via personalized care plans that provide decision support for healthcare providers and patients. The companies also are exploring ways to leverage IBM’s Watson Health Cloud platform to improve Medtronic and other partners’ closed loop algorithms, which attempt to mimic the functions of a healthy pancreas.
While certainly a sound investment, partnerships like those between Medtronic and IBM are most likely to increase as the medtech behemoth embraces emerging bundle payment and risk-sharing business models, and distances itself from product-only growth strategies. “Of all the healthcare industries, medical devices faces the biggest challenge from the shift to a fee-for-outcome system. Hospitals are gaining clout when it comes to deciding if a medical device gets used by doctors and the price they pay,” Jean Hynes, manager of the Vanguard Health Care Fund (a Medtronic shareowner), explained to Barrons. “Omar (Ishrak) is way ahead of the curve in figuring out this new world in terms of how to compete.”
Indeed, Medtronic Chairman and CEO Omar Ishrak’s ingenuity has yielded significant financial gains for his company. In fiscal 2015 (year ended April 24, 2015), revenue jumped 19 percent to $20.2 billion—due largely to the Covidien acquisition—and the company’s stock appreciated 33 percent, or 20 percentage points better than the S&P 500’s overall performance. In addition, operating margin improved by roughly 60 basis points, which corresponded to about 200 basis points of operating leverage. Operating profit, however, slipped 1.2 percent to $3.76 billion and net income fell 12.7 percent to $2.67 billion as volatile currency rates undercut earnings by $666 million.
That instability had a negligible effect on foreign sales, though. Non-U.S. developed markets revenue ballooned 13 percent to $6.37 billion, and emerging market proceeds surged 23 percent to $2.58 billion, according to Medtronic’s fiscal 2015 annual report. Executives attributed the rise in foreign sales to the fourth-quarter creation of the Minimally Invasive Therapies Group, solid gains in the company’s three other business segments, significant new product growth in the Australia-New Zealand region, and steady sales in Western Europe of catheter lab managed services. Similarly, broad-based procedural growth and new product launches bolstered U.S. sales 22 percent to $11.3 billion.
“We delivered solid results in FY15,” Ishrak wrote in his annual letter to shareholders. “The most important event of our fiscal year was the Covidien transaction. The combination of Medtronic and Covidien positions us as a clear industry leader and has set the stage for us to lead the transformation of healthcare. In many ways, this acquisition has initiated a new era for Medtronic.”
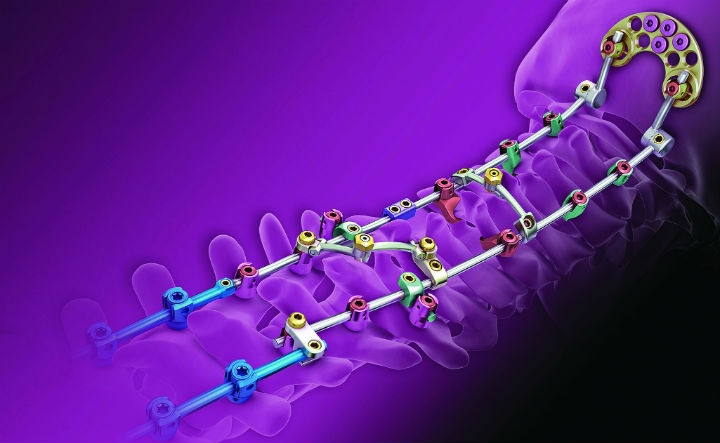
Medtronic received U.S. Food and Drug Administration clearance in February 2015 for expanded indications of the VERTEX Reconstruction System. The new clearance allows for lateral mass and pedicle screw fixation in the posterior cervical spine. (Courtesy of Medtronic plc)
The company’s new era began quite auspiciously, with solid gains reported in all business segments. Medtronic’s Restorative Therapies Group posted its best gains in five years, but the final numbers were still below management’s expectations. Growth in Surgical Technologies proceeds was partially offset by sagging spine sales and a $127 million loss to foreign currency volatility. Even so, Restorative Therapies still achieved an overall 4 percent increase compared to FY14, generating $6.75 billion in sales. The Group’s overall health was driven by the birth of the Neurovascular division (formerly part of Covidien), and solid growth in both the Surgical Technologies and Neuromodulation divisions.
Acquisitions figured prominently into Neuromodulation’s 2015 fiscal year but did not impact net sales. Proceeds rose 4 percent over FY14 to $1.97 billion on the strength of Medtronic’s gastro/urology products and deep brain/pain stimulation technology, particularly the RestoreSensor SureScan MRI system, an implantable pain-relieving device for the spinal cord.
Spine spoiled Restorative Therapy’s (and Medtronic’s) near-perfect financial report card, posting FY15’s only loss. Division sales slipped 2 percent to $2.97 billion as Interventional spine proceeds fell victim to weak demand in Europe, pricing pressures in Germany, and overall foreign currency volatility. Neither modest U.S. procedural growth nor new product launches—namely, the Prestige LP Cervical Disc and Pure Titanium Coated interbody fusion devices—could overcome domestic pricing pressures and lingering stagnation in the bone morphogenetic protein market.
Debuting in July 2014, the Prestige LP Cervical disc is intended to treat single-level diseased discs. The implant is an upgrade to the Prestige disc Medtronic released in 2007 featuring a ball-and-trough socket that maintains natural bending, rotation, and translation of the two components relative to each other.
The LP disc features an improved fixation mechanism that uses two rails that press into pre-drilled holes instead of the commonly used bone screws. The device is made of a proprietary titanium-ceramic composite that reportedly has demonstrated slower wear while producing less visual scatter during magnetic resonance imaging scans when compared to stainless steel. Available worldwide for a decade, U.S. Food and Drug Administration (FDA) approval of the disc occurred following a prospective, multicenter, historical-controlled investigational device exemption trial in the United States.
FDA approval of the Prestige LP disc coincided with CE marking of the pure titanium-coated Capstone PTC and Clydesdale PTC Spinal systems—devices intended to help reduce pressure on spinal cords or nerve roots during recovery from spinal surgery. The Clydesdale system comprises tantalum markers and PEEK (polyether ether ketone) cages of various widths and heights that can be inserted between two lumbar or lumbosacral vertebral bodies for support and correction during lumbar interbody fusion surgeries. The implants’ hollow geometry allows them to be packed with autogenous bone graft, according to Medtronic. Both systems received FDA clearance in March 2014 and hit the U.S. market in August that year.
Rounding out Spine’s string of July (2014) christenings was the Cornerstone-SR titanium-coated cervical cage, which received CE mark approval and began selling in Western Europe. The device is designed to treat cervical disc disease at one level from the C2-C3 disc to the C7-T1 disc.
The Cornerstone’s American counterpart—the titanium-coated Anatomic PEEK Cervical Fusion System—launched in September 2014 and kicked off the company’s autumn product introductions. Nipping at the heels of the Anatomic system was the Kyphon Express II Balloon Kyphoplasty platform, including the next-generation Kyphon Cement Delivery system.
The Kyphon platform features new 700 psi balloons as well as a redesigned cement delivery system that enables one balloon to remain inflated during contralateral cement fill, thereby helping to reduce fractures. An expanded tools portfolio also makes the system generally more adaptable; a Quick Release button, for example, stops bone cement flow on demand, giving surgeons precise control over the amount of cement used in procedures. “…physicians can now treat more spine levels by choosing the sizes, length, and volume most appropriate for the individual patient’s anatomy and fracture,” proclaimed Dr. Doug Beall, chief of radiology services for Clinical Radiology of Oklahoma.
Medtronic followed up the Kyphon platform release with the U.S. debut (in October 2014) of the Divergence Anterior Cervical Fusion system for single-level cervical disc disease treatment. Featuring a plate and interbody cage that can be inserted simultaneously, the system incorporates a laterally divergent screw insertion technique, which reportedly requires less retraction compared to traditional medially convergent screw insertion techniques used with other anterior cervical plating systems.
The company bounced back from a four-month drought in new product releases with the FDA clearance for expanded indications of the Vertex Reconstruction system. Initially, the system was approved for use in upper thoracic screw fixation, but the most recent clearance (in February 2015) allows for lateral mass and pedicle screws to be used as a form of fixation to treat different pathologies of the cervical spine.
KEY EXECUTIVES:
Omar Ishrak, Chairman and CEO
Karen L. Parkill, Exec. VP and Chief Financial Officer
Richard E. Kuntz, M.D., M.SC., Sr. VP and Chief Scientific, Clinical, and Regulatory Officer
Luann Pendy, Sr. VP, Global Quality
Mike Genau, Sr. VP and President, Americas Region
Robert ten Hoedt, Exec. VP and President, EMEAC
Bob White, Sr. VP and President, Asia Pacific Region
Chris Lee, Sr. VP and President, Greater China Region
Geoffrey S. Martha, Exec. VP and Group President, Restorative Therapies Group
NUMBER OF EMPLOYEES: 5,000
HEADQUARTERS: Memphis, Tenn.
It’s going to be difficult, if not downright impossible, for Medtronic plc to top its 2015 fiscal year. During that historic 12-month period, the company purchased Covidien plc for a record $50 billion, creating the world’s second-largest medical device company (Johnson & Johnson still leads, even with slumping device revenue) and sparking a national debate over the deal’s structure. Under terms of the all cash-and-stock purchase, Medtronic redomiciled overseas, reducing its corporate tax rate in a controversial practice known as an inversion. The move was among a slew of inverted deals that eventually prompted the U.S. Treasury Department to impose limits on re-incorporations and restrict related-party debt for American subsidiaries.
Besides trimming its tax rate, the blockbuster merger enabled Medtronic to increase its fiscal year revenue by 19 percent and helped the company become more diversified and balanced, given Covidien’s focus on surgical tools and hospital-based technologies. It also expanded Medtronic’s exposure to smaller cities in emerging markets, which currently constitute 12.7 percent of total revenue, up slightly from 12.3 percent in FY14. And it better positions the company to lead the push toward value-based healthcare by integrating patient care services that balance cost and access challenges.
“[Omar] Ishrak changed the ethos of the company, expanding it from a device maker into one that also offers services and partners with healthcare providers,” Raj Denhoy, a medical technology research analyst at Jefferies LLC told Barron’s last fall. “It was a bold move.”
And a necessary one as well. With the U.S. healthcare industry favoring risk- and value-based business models over traditional fee-for-service systems (thanks to Obamacare), medtech companies are increasingly reducing their dependence solely on medical devices to sustain growth. At Medtronic, that newfound freedom has inspired collaborations with hospital systems, payers, governments, and companies to develop integrated health solutions that complement and enhance product value through traditional wraparound services and solutions.
In FY15, those solutions targeted diabetes and patient home monitoring. Medtronic partnered with both Sanofi S.A. and IBM on diabetes treatment and management, leveraging its devices and care management products with its cohorts’ existing solutions to improve patient outcomes. The company is matching its insulin pumps and glucose monitoring technology with Sanofi’s insulin products to ameliorate type 2 diabetes management, particularly for patients having trouble controlling their blood sugar levels. The pair has been battling type 1 diabetes with a similar agreement, offering an implantable insulin delivery system to European patients.
Medtronic and Sanofi also are working together on care management services, developing an education plan for type 2 diabetics who cannot control their glucose levels through medication. The program will guide these patients through the initial phase of insulin treatment.
Medtronic’s hookup with IBM aims to improve outcomes as well, albeit via personalized care plans that provide decision support for healthcare providers and patients. The companies also are exploring ways to leverage IBM’s Watson Health Cloud platform to improve Medtronic and other partners’ closed loop algorithms, which attempt to mimic the functions of a healthy pancreas.
While certainly a sound investment, partnerships like those between Medtronic and IBM are most likely to increase as the medtech behemoth embraces emerging bundle payment and risk-sharing business models, and distances itself from product-only growth strategies. “Of all the healthcare industries, medical devices faces the biggest challenge from the shift to a fee-for-outcome system. Hospitals are gaining clout when it comes to deciding if a medical device gets used by doctors and the price they pay,” Jean Hynes, manager of the Vanguard Health Care Fund (a Medtronic shareowner), explained to Barrons. “Omar (Ishrak) is way ahead of the curve in figuring out this new world in terms of how to compete.”
Indeed, Medtronic Chairman and CEO Omar Ishrak’s ingenuity has yielded significant financial gains for his company. In fiscal 2015 (year ended April 24, 2015), revenue jumped 19 percent to $20.2 billion—due largely to the Covidien acquisition—and the company’s stock appreciated 33 percent, or 20 percentage points better than the S&P 500’s overall performance. In addition, operating margin improved by roughly 60 basis points, which corresponded to about 200 basis points of operating leverage. Operating profit, however, slipped 1.2 percent to $3.76 billion and net income fell 12.7 percent to $2.67 billion as volatile currency rates undercut earnings by $666 million.
That instability had a negligible effect on foreign sales, though. Non-U.S. developed markets revenue ballooned 13 percent to $6.37 billion, and emerging market proceeds surged 23 percent to $2.58 billion, according to Medtronic’s fiscal 2015 annual report. Executives attributed the rise in foreign sales to the fourth-quarter creation of the Minimally Invasive Therapies Group, solid gains in the company’s three other business segments, significant new product growth in the Australia-New Zealand region, and steady sales in Western Europe of catheter lab managed services. Similarly, broad-based procedural growth and new product launches bolstered U.S. sales 22 percent to $11.3 billion.
“We delivered solid results in FY15,” Ishrak wrote in his annual letter to shareholders. “The most important event of our fiscal year was the Covidien transaction. The combination of Medtronic and Covidien positions us as a clear industry leader and has set the stage for us to lead the transformation of healthcare. In many ways, this acquisition has initiated a new era for Medtronic.”

Medtronic received U.S. Food and Drug Administration clearance in February 2015 for expanded indications of the VERTEX Reconstruction System. The new clearance allows for lateral mass and pedicle screw fixation in the posterior cervical spine. (Courtesy of Medtronic plc)
The company’s new era began quite auspiciously, with solid gains reported in all business segments. Medtronic’s Restorative Therapies Group posted its best gains in five years, but the final numbers were still below management’s expectations. Growth in Surgical Technologies proceeds was partially offset by sagging spine sales and a $127 million loss to foreign currency volatility. Even so, Restorative Therapies still achieved an overall 4 percent increase compared to FY14, generating $6.75 billion in sales. The Group’s overall health was driven by the birth of the Neurovascular division (formerly part of Covidien), and solid growth in both the Surgical Technologies and Neuromodulation divisions.
Acquisitions figured prominently into Neuromodulation’s 2015 fiscal year but did not impact net sales. Proceeds rose 4 percent over FY14 to $1.97 billion on the strength of Medtronic’s gastro/urology products and deep brain/pain stimulation technology, particularly the RestoreSensor SureScan MRI system, an implantable pain-relieving device for the spinal cord.
Spine spoiled Restorative Therapy’s (and Medtronic’s) near-perfect financial report card, posting FY15’s only loss. Division sales slipped 2 percent to $2.97 billion as Interventional spine proceeds fell victim to weak demand in Europe, pricing pressures in Germany, and overall foreign currency volatility. Neither modest U.S. procedural growth nor new product launches—namely, the Prestige LP Cervical Disc and Pure Titanium Coated interbody fusion devices—could overcome domestic pricing pressures and lingering stagnation in the bone morphogenetic protein market.
Debuting in July 2014, the Prestige LP Cervical disc is intended to treat single-level diseased discs. The implant is an upgrade to the Prestige disc Medtronic released in 2007 featuring a ball-and-trough socket that maintains natural bending, rotation, and translation of the two components relative to each other.
The LP disc features an improved fixation mechanism that uses two rails that press into pre-drilled holes instead of the commonly used bone screws. The device is made of a proprietary titanium-ceramic composite that reportedly has demonstrated slower wear while producing less visual scatter during magnetic resonance imaging scans when compared to stainless steel. Available worldwide for a decade, U.S. Food and Drug Administration (FDA) approval of the disc occurred following a prospective, multicenter, historical-controlled investigational device exemption trial in the United States.
FDA approval of the Prestige LP disc coincided with CE marking of the pure titanium-coated Capstone PTC and Clydesdale PTC Spinal systems—devices intended to help reduce pressure on spinal cords or nerve roots during recovery from spinal surgery. The Clydesdale system comprises tantalum markers and PEEK (polyether ether ketone) cages of various widths and heights that can be inserted between two lumbar or lumbosacral vertebral bodies for support and correction during lumbar interbody fusion surgeries. The implants’ hollow geometry allows them to be packed with autogenous bone graft, according to Medtronic. Both systems received FDA clearance in March 2014 and hit the U.S. market in August that year.
Rounding out Spine’s string of July (2014) christenings was the Cornerstone-SR titanium-coated cervical cage, which received CE mark approval and began selling in Western Europe. The device is designed to treat cervical disc disease at one level from the C2-C3 disc to the C7-T1 disc.
The Cornerstone’s American counterpart—the titanium-coated Anatomic PEEK Cervical Fusion System—launched in September 2014 and kicked off the company’s autumn product introductions. Nipping at the heels of the Anatomic system was the Kyphon Express II Balloon Kyphoplasty platform, including the next-generation Kyphon Cement Delivery system.
The Kyphon platform features new 700 psi balloons as well as a redesigned cement delivery system that enables one balloon to remain inflated during contralateral cement fill, thereby helping to reduce fractures. An expanded tools portfolio also makes the system generally more adaptable; a Quick Release button, for example, stops bone cement flow on demand, giving surgeons precise control over the amount of cement used in procedures. “…physicians can now treat more spine levels by choosing the sizes, length, and volume most appropriate for the individual patient’s anatomy and fracture,” proclaimed Dr. Doug Beall, chief of radiology services for Clinical Radiology of Oklahoma.
Medtronic followed up the Kyphon platform release with the U.S. debut (in October 2014) of the Divergence Anterior Cervical Fusion system for single-level cervical disc disease treatment. Featuring a plate and interbody cage that can be inserted simultaneously, the system incorporates a laterally divergent screw insertion technique, which reportedly requires less retraction compared to traditional medially convergent screw insertion techniques used with other anterior cervical plating systems.
The company bounced back from a four-month drought in new product releases with the FDA clearance for expanded indications of the Vertex Reconstruction system. Initially, the system was approved for use in upper thoracic screw fixation, but the most recent clearance (in February 2015) allows for lateral mass and pedicle screws to be used as a form of fixation to treat different pathologies of the cervical spine.




























