08.09.16
$883 Million
KEY EXECUTIVES:
Peter J. Arduini, President, CEO, and Director
Stuart M. Essig, Ph.D., Chairman of the Board
Mark Augusti, Corp. VP and President of Orthopedics and Tissue Technologies
Kenneth Burhop, Ph.D., Corp. VP and Chief Scientific Officer
Glenn Coleman, Corp. VP and Chief Financial Officer
Robert T. Davis, Jr., Corp. VP and President of Specialty Surgical Solutions
John Mooradian, Corp. VP, Global Operations and Supply Chain
Judith E. O’Grady, R.N., M.S.N., R.A.C., Corp. VP, Global Regulatory Affairs
Dan Reuvers, Corp. VP and President – International
Joseph Vinhais, Corp. VP, Global Quality Assurance
NUMBER OF EMPLOYEES: 3,500
HEADQUARTERS: Plainsboro, N.J.
Integra is a company in transition. It’s still very much dedicated to providing medical technology to enhance healthcare, but its focus seems to be, in part, shifting away from the orthopedic sector focus it once maintained. Or, perhaps more accurately, it is separating itself from spine technologies within that sector. The company is still active with device solutions for tissue repair and regeneration as well as small bone fixation and joint replacement solutions.
On July 1, 2015, Integra saw the launch of SeaSpine, a company that will focus on surgical solutions for the treatment of spinal disorders. Spun off as a stand-alone firm, SeaSpine was formed out of Integra’s orthobiologics and spinal fusion hardware offerings.
“We believe that an independent SeaSpine will focus its investments on research and development and sales and marketing to drive faster growth. Similarly, the separation will result in faster growth for the remaining Integra businesses, Specialty Surgical Solutions and Orthopedics and Tissue Technologies,” Peter Arduini, Integra’s president and CEO, said in a company release.
A couple of months before the formal split from Integra, SeaSpine announced that Keith Valentine would be named CEO. Prior to that, Valentine had served as president and chief operating officer of NuVasive Inc., a position he had held since January 2007. Joining him, John Bostjancic was appointed as chief financial officer for the spinal company. Previously, Bostjancic had been senior vice president of global supply chain for Integra since February 2012 as well as senior vice president of financial planning and analysis from 2008 to 2012.
“I am thrilled to have the opportunity to lead SeaSpine following the spin-off from Integra,” Valentine said in a release issued at the time of the announcement of his new position.
“SeaSpine has the ability to leverage its leading orthobiologics products and broad spinal fusion hardware portfolio, as well as invest in a pipeline of differentiated products to drive growth. I look forward to working with our customers, our strong management team, employees, and our future investors in the coming weeks and months.”
Since the spin-off from Integra, SeaSpine has launched several new products for the orthopedic spine market. In 2015, the company began selling the Ventura NanoMetalene transforaminal intervertebral body fusion device and the Cambria NanoMetalene cervical interbody body fusion device. In June 2016, it announced its Hollywood VI NanoMetalene transforaminal lumbar interbody fusion device was available for use in spinal fusion procedures at one or two contiguous levels (L2-S1) in skeletally mature patients with degenerative disc disease.
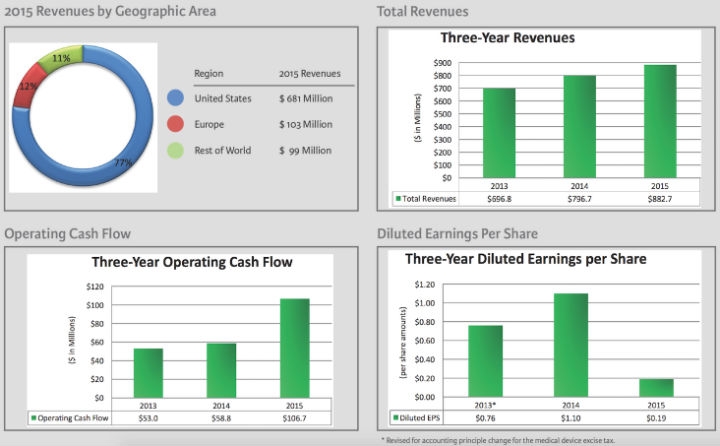
Meanwhile, Integra continued on its long-term quest to become a multi-billion dollar, diversified medical device company. In a letter to stockholders, Arduini indicated that in 2015, the company made significant progress toward that end. The company’s strong financial results were reflected in revenue growth of 10.8 percent over 2014’s figure of $796.7 million, representing $882.7 million in 2015. That revenue was the combined total from the company’s two segments—Specialty Surgical Solutions, which offers instrumentation for a range of specialties including neurosurgery and critical care, and Orthopedics & Tissue Technologies, which is the division responsible for aforementioned tissue repair and regeneration products, as well as small bone fixation and joint replacement solutions. This was a shift from the company’s previous three segments, which changed as a result of the spin-off of the SeaSpine business.
(Graphics courtesy of Integra LifeSciences)
The surgical segment contributed 66 percent to the total revenue figure, representing $587 million. The orthopedic and tissue division offered 34 percent, or $296 million. The company’s sales are primarily driven by the U.S. market, which saw 77 percent of the revenue for $681 million. Europe and the rest of the world followed a distant second and third, posting $103 million (12 percent) and $99 million (11 percent) respectively.
While a number of factors contributed to the positive growth of Integra in 2015, the company really only had one new product release highlight for the orthopedic space. Just as the new fiscal year began for the company in January, it announced the full market release of the Integra Reinforcement Matrix, a biological implant composed of porcine dermis that may be used for any type of tendon injury that requires surgical reconstruction. The product, according to the company, leverages a material that has been used in implants for over 14 years and in more than 40,000 surgical procedures. In addition, it is indicated for use in tendon injuries in the extremities. That includes disorders such as an ankle sprain, ruptured Achilles tendon, or flexor tendon injury of the hand.
The company should be able to remedy the new product release drought seen last year in the coming future with its dedication to internal development of new technologies. Integra devoted a healthy 5.8 percent of revenue to research and development in 2015. That figure translated to $50.9 million, a total that was higher than either of the previous two years. The company explained the increase in expenditures was primarily attributed to additional spending on new product development and clinical studies, as well as acquisition activity.
Executives expect the percentage of revenue for R&D expenditures in 2016 will continue to be similarly in the range of 5.5 percent to 6 percent.
Regarding that acquisition activity, the company was quite busy in 2015, following a series of purchases over the last few years that saw the company absorb Tarsus Medical Inc. (January 2013), Confluent Surgical Inc. (from Covidien Group in January 2014), and Medtronic Xomed Instrumentation (from Medtronic in October 2014). Further, it made the largest buy in Integra’s history in July 2015. TEI Biosciences Inc. and TEI Medical Inc. were acquired for approximately $312.4 million ($210.4 million and $102 million, respectively). TEI Bio developed and sold biologic devices for soft tissue repair and regenerative applications, including dura and hernia repair and plastic and reconstructive surgery. TEI Med actually licensed technology from TEI Bio for wound healing and orthopedics.
“This acquisition broadens our presence in regenerative wound care and tissue repair and represents a significant push forward toward our growth objectives for 2015 and beyond. The addition of TEI is an important, strategic next step for both our channel and international expansion priorities. We are enthusiastic about both TEI’s product development and commercial expertise, which accelerates our ability to establish an immediate presence in the diabetic foot ulcer space,” Arduini said in a company release announcing the deal.
According to Integra, a number of factors made the purchase particularly attractive. Most notable was TEI’s PriMatrix Dermal Repair Scaffold, which enhanced Integra’s offering in the regenerative would care and tissue repair space. The firm generated 2014 revenues of $63.5 million.
“It is an exciting time for TEI, and I am confident in Integra’s ability to grow our leading platform technology to drive broader expansion into regenerative medicine including wound care, plastic and reconstructive surgery, and other soft tissue repair and reconstruction applications,” Dr. Yiannis Monovoukas, Ph.D., chairman, president, and CEO of TEI, added in the same company release.
The TEI transaction was just the largest of a number of acquisitions Integra made in 2015. The company kicked off the year in January with the announcement that it had acquired Metasurg’s foot and ankle product portfolio in the month prior. The buy enhanced Integra’s lower extremity product offering while also providing the opportunity to distribute a biologic allograft derived from human placental tissue as a compliment to the company’s wound care offerings. Posting revenues of approximately $6 million, Integra expects the product line to return to double-digit growth numbers following the transition period.
In September, the company announced the purchase of two product portfolios from Tornier. The Salto Talaris and Salto XT ankle replacement products, as well as the Futura silastic toe replacement products were purchased for $6 million in a cash transaction. Integra cited interest in the products as an opportunity to both enhance its lower extremities product offering and accelerate entry into the U.S. total ankle replacement market.
Lastly, in December, the company acquired the assets of Tekmed Instruments, an Italian distributor of Integra products within the country. The deal, worth $14.2 million, provided support to the growth of the Specialty Surgical Solutions segment within Italy. In addition, with Tekmed’s focus on neurosurgery and neurotrauma, the deal also enhanced the offerings for those areas.
KEY EXECUTIVES:
Peter J. Arduini, President, CEO, and Director
Stuart M. Essig, Ph.D., Chairman of the Board
Mark Augusti, Corp. VP and President of Orthopedics and Tissue Technologies
Kenneth Burhop, Ph.D., Corp. VP and Chief Scientific Officer
Glenn Coleman, Corp. VP and Chief Financial Officer
Robert T. Davis, Jr., Corp. VP and President of Specialty Surgical Solutions
John Mooradian, Corp. VP, Global Operations and Supply Chain
Judith E. O’Grady, R.N., M.S.N., R.A.C., Corp. VP, Global Regulatory Affairs
Dan Reuvers, Corp. VP and President – International
Joseph Vinhais, Corp. VP, Global Quality Assurance
NUMBER OF EMPLOYEES: 3,500
HEADQUARTERS: Plainsboro, N.J.
Integra is a company in transition. It’s still very much dedicated to providing medical technology to enhance healthcare, but its focus seems to be, in part, shifting away from the orthopedic sector focus it once maintained. Or, perhaps more accurately, it is separating itself from spine technologies within that sector. The company is still active with device solutions for tissue repair and regeneration as well as small bone fixation and joint replacement solutions.
On July 1, 2015, Integra saw the launch of SeaSpine, a company that will focus on surgical solutions for the treatment of spinal disorders. Spun off as a stand-alone firm, SeaSpine was formed out of Integra’s orthobiologics and spinal fusion hardware offerings.
“We believe that an independent SeaSpine will focus its investments on research and development and sales and marketing to drive faster growth. Similarly, the separation will result in faster growth for the remaining Integra businesses, Specialty Surgical Solutions and Orthopedics and Tissue Technologies,” Peter Arduini, Integra’s president and CEO, said in a company release.
A couple of months before the formal split from Integra, SeaSpine announced that Keith Valentine would be named CEO. Prior to that, Valentine had served as president and chief operating officer of NuVasive Inc., a position he had held since January 2007. Joining him, John Bostjancic was appointed as chief financial officer for the spinal company. Previously, Bostjancic had been senior vice president of global supply chain for Integra since February 2012 as well as senior vice president of financial planning and analysis from 2008 to 2012.
“I am thrilled to have the opportunity to lead SeaSpine following the spin-off from Integra,” Valentine said in a release issued at the time of the announcement of his new position.
“SeaSpine has the ability to leverage its leading orthobiologics products and broad spinal fusion hardware portfolio, as well as invest in a pipeline of differentiated products to drive growth. I look forward to working with our customers, our strong management team, employees, and our future investors in the coming weeks and months.”
Since the spin-off from Integra, SeaSpine has launched several new products for the orthopedic spine market. In 2015, the company began selling the Ventura NanoMetalene transforaminal intervertebral body fusion device and the Cambria NanoMetalene cervical interbody body fusion device. In June 2016, it announced its Hollywood VI NanoMetalene transforaminal lumbar interbody fusion device was available for use in spinal fusion procedures at one or two contiguous levels (L2-S1) in skeletally mature patients with degenerative disc disease.
Meanwhile, Integra continued on its long-term quest to become a multi-billion dollar, diversified medical device company. In a letter to stockholders, Arduini indicated that in 2015, the company made significant progress toward that end. The company’s strong financial results were reflected in revenue growth of 10.8 percent over 2014’s figure of $796.7 million, representing $882.7 million in 2015. That revenue was the combined total from the company’s two segments—Specialty Surgical Solutions, which offers instrumentation for a range of specialties including neurosurgery and critical care, and Orthopedics & Tissue Technologies, which is the division responsible for aforementioned tissue repair and regeneration products, as well as small bone fixation and joint replacement solutions. This was a shift from the company’s previous three segments, which changed as a result of the spin-off of the SeaSpine business.

(Graphics courtesy of Integra LifeSciences)
The surgical segment contributed 66 percent to the total revenue figure, representing $587 million. The orthopedic and tissue division offered 34 percent, or $296 million. The company’s sales are primarily driven by the U.S. market, which saw 77 percent of the revenue for $681 million. Europe and the rest of the world followed a distant second and third, posting $103 million (12 percent) and $99 million (11 percent) respectively.
While a number of factors contributed to the positive growth of Integra in 2015, the company really only had one new product release highlight for the orthopedic space. Just as the new fiscal year began for the company in January, it announced the full market release of the Integra Reinforcement Matrix, a biological implant composed of porcine dermis that may be used for any type of tendon injury that requires surgical reconstruction. The product, according to the company, leverages a material that has been used in implants for over 14 years and in more than 40,000 surgical procedures. In addition, it is indicated for use in tendon injuries in the extremities. That includes disorders such as an ankle sprain, ruptured Achilles tendon, or flexor tendon injury of the hand.
The company should be able to remedy the new product release drought seen last year in the coming future with its dedication to internal development of new technologies. Integra devoted a healthy 5.8 percent of revenue to research and development in 2015. That figure translated to $50.9 million, a total that was higher than either of the previous two years. The company explained the increase in expenditures was primarily attributed to additional spending on new product development and clinical studies, as well as acquisition activity.
Executives expect the percentage of revenue for R&D expenditures in 2016 will continue to be similarly in the range of 5.5 percent to 6 percent.
Regarding that acquisition activity, the company was quite busy in 2015, following a series of purchases over the last few years that saw the company absorb Tarsus Medical Inc. (January 2013), Confluent Surgical Inc. (from Covidien Group in January 2014), and Medtronic Xomed Instrumentation (from Medtronic in October 2014). Further, it made the largest buy in Integra’s history in July 2015. TEI Biosciences Inc. and TEI Medical Inc. were acquired for approximately $312.4 million ($210.4 million and $102 million, respectively). TEI Bio developed and sold biologic devices for soft tissue repair and regenerative applications, including dura and hernia repair and plastic and reconstructive surgery. TEI Med actually licensed technology from TEI Bio for wound healing and orthopedics.
“This acquisition broadens our presence in regenerative wound care and tissue repair and represents a significant push forward toward our growth objectives for 2015 and beyond. The addition of TEI is an important, strategic next step for both our channel and international expansion priorities. We are enthusiastic about both TEI’s product development and commercial expertise, which accelerates our ability to establish an immediate presence in the diabetic foot ulcer space,” Arduini said in a company release announcing the deal.
According to Integra, a number of factors made the purchase particularly attractive. Most notable was TEI’s PriMatrix Dermal Repair Scaffold, which enhanced Integra’s offering in the regenerative would care and tissue repair space. The firm generated 2014 revenues of $63.5 million.
“It is an exciting time for TEI, and I am confident in Integra’s ability to grow our leading platform technology to drive broader expansion into regenerative medicine including wound care, plastic and reconstructive surgery, and other soft tissue repair and reconstruction applications,” Dr. Yiannis Monovoukas, Ph.D., chairman, president, and CEO of TEI, added in the same company release.
The TEI transaction was just the largest of a number of acquisitions Integra made in 2015. The company kicked off the year in January with the announcement that it had acquired Metasurg’s foot and ankle product portfolio in the month prior. The buy enhanced Integra’s lower extremity product offering while also providing the opportunity to distribute a biologic allograft derived from human placental tissue as a compliment to the company’s wound care offerings. Posting revenues of approximately $6 million, Integra expects the product line to return to double-digit growth numbers following the transition period.
In September, the company announced the purchase of two product portfolios from Tornier. The Salto Talaris and Salto XT ankle replacement products, as well as the Futura silastic toe replacement products were purchased for $6 million in a cash transaction. Integra cited interest in the products as an opportunity to both enhance its lower extremities product offering and accelerate entry into the U.S. total ankle replacement market.
Lastly, in December, the company acquired the assets of Tekmed Instruments, an Italian distributor of Integra products within the country. The deal, worth $14.2 million, provided support to the growth of the Specialty Surgical Solutions segment within Italy. In addition, with Tekmed’s focus on neurosurgery and neurotrauma, the deal also enhanced the offerings for those areas.




















