08.15.18
$9.3 Billion
KEY EXECUTIVES:
Alex Gorsky, Chairman and CEO
Dominic J. Caruso, Exec. VP and CFO
Jennifer Taubert, Company Group Chairman, Johnson & Johnson
Ashley McEvoy, Exec. VP, Worldwide Chairman, Medical Devices
Sandra E. Peterson, Exec. VP, Group Worldwide Chairman
Kathryn E. Wengel, Exec. VP, Chief Global Supply Chain Officer
Paulus Stoffels, Exec. VP, Chief Scientific Officer
Michael H. Ullmann, Exec. VP, General Counsel
Ronald A. Kapusta, Corporate Controller, Chief Accounting Officer
Number OF EMPLOYEES: 18,000
GLOBAL HEADQUARTERS: Warsaw, Ind.; Raynham, Mass.; and West Chester, Pa.
Some companies chase the future. Others shape it.
Apple clearly falls into the latter category. From its first computers—the rudimentary but revolutionary pre-assembled Apple 1 and graphical user interface-touting Lisa (the one that introduced the “mouse”)—to the pocket-sized iPod, touch computing iPhone and netbook-killing iPad, Apple truly has changed the way humans socialize and communicate.
Google and Tesla are top morrow moulders as well. The former entity essentially cataloged the Web, giving structure to the jumbled mess of digital data that once littered the information superhighway. Its algorithms and index updates over the last 20 years have turned the tech giant into the go-to source of information (and quite a popular transitive verb). Tesla, on the other hand, is transforming the auto industry with its electric cars and autonomous (self-driving) vehicles, while also becoming a poster child for sustainability (its Powerwall rechargeable home battery system is guaranteed to store 70 percent of its rated power output at 10 years).
Johnson & Johnson considers itself a future forger too. Actually, the company’s chief executive claims the multinational conglomerate has dedicated its life to defining healthcare’s future. “Through the decades, Johnson & Johnson has advanced with the evolution of science and technology...” Chairman and CEO Alex Gorsky reminds shareholders at the start of the firm’s 2017 annual report. “In fact, every year for the past 132 years, Johnson & Johnson has been involved in defining the future of healthcare.”
Maybe not every year, admittedly. But certainly more often than not, J&J has been at the forefront of significant medical breakthroughs: It introduced the world to sterile surgical dressings and sutures (1887); first aid kits (1888); affordable dental floss (1898); BAND-AID adhesive bandages (1921); synthetic sterile sutures (1969); a disposable skin stapler (1978); coated sutures (1979); extended-wear disposable contact lenses (1987); minimally invasive gall bladder removal (1988); low-temperature gas plasma sterilization (1992); and a topical skin adhesive glue (1998).
The company picked up the pace of innovation with the millennium’s arrival, developing a coated antibacterial suture (2003); silicone gel implants (2006); a tissue expander with suturing tabs (2008); and a single-handed balloon sinuplasty device (2012), among others. Over the last few years, however, J&J has somewhat shifted its focus to better target (and shape) both digital healthcare and value-based medicine. In 2015, for example, the company teamed up with IBM’s Watson Health unit to create consumer-focused virtual coaching apps for knee replacement patients. Using Watson’s cognitive computing, the app predicts outcomes, suggests treatment plans, and provides personalized encouragement to patients during recovery.
Through its Ethicon subsidiary, J&J also has partnered with Alphabet Inc.’s Verily Life Sciences to develop a new robotic-assisted surgical solutions platform for improved access to minimally invasive surgery, greater hospital efficiency, and enhanced patient outcomes. Since the pairing, the partners have developed a surgical platform prototype encompassing robotics, visualization, advanced instrumentation, data analytics, and connectivity—an innovation the duo is calling “Surgery 4.0.”
J&J’s value-based healthcare initiatives aim to ease the transition to alternative reimbursement models and reinforce patient engagement, preventative care, and operational efficiency. Its CareAdvantage initiative is a holistic, data-driven approach designed to help companies shift to payment models tied to quality and/or value. The plan uses data-driven analysis and insights, on-site assessments, and focused action plans to improve operational efficiency in various medical fields—including orthopedics—based on goal-specific targeted metrics.
CareAdvantage’s orthopedic component combines a cloud-based platform for patient interaction; episode data analysis; infection risk reduction best practices; pre- and post-operative care (setting) expectations; and a standardized hip fracture care program. Its Patient Athlete plan helps patients prepare for and recover from orthopedic surgery through science-based principles from the Johnson & Johnson Human Performance Institute.
“...we are convinced that technology will not replace but rather will extend human caring...Ahead we see a future that lies beyond the intersection of science and technology,” Gorsky wrote in his shareholder letter. “We are rocketing toward that future, toward the very edge of imagination and possibility. That’s exciting, of course, and just a little daunting at the same time. Getting to that future will require urgency, boldness and vision—characteristics most often attributed to very successful startups. However, I believe it is also a great description of this company today—a 132-year-old startup.”
Not many “startups,” however, are as fiscally fruitful as J&J. Last year, the New Brunswick, N.J.-based company increased its total revenue 6.3 percent to $76.4 billion and grew its per-share stock price by $24.51, or 21.3 percent (ending 2017 at $139.72). Gains occurred in each of J&J’s three product franchises, with Pharmaceutical posting the largest rise (8.3 percent) followed by Medical Devices (5.9 percent) and Consumer (2.2 percent).
The sales surge in Medical Devices is a redemption of sorts, for past franchise sins. Declining revenue in recent years prompted the company to implement a massive restructuring in 2016 and replace its slower-growing businesses with more profitable technologies—a strategy that surely induced the $1.045 billion sale of DePuy Synthes’ Codman Neurosurgery business last February and the $4.33 billion purchase of Abbott Medical Optics in September 2016. The swap-out scheme also was responsible for a handful of smaller bolt-on acquisitions last year that augmented DePuy Synthes’ minimally invasive surgical solutions portfolio and expanded its capabilities in expandable spinal cage technology, 3D printing, and bone defect remedies.
The spinal cage technology came from Interventional Spine Inc., an Irvine, Calif.-based developer of expandable cages and minimally invasive surgical techniques. The company’s cage technology provides optimal tactile feedback for surgeons and features an adjustable design that customizes the device height to individual patient anatomy. The implants are designed to maximize bone graft contact and containment, which helps promote bone growth and fusion. In addition to the expandable cage technology, DePuy picked up Interventional Spine’s facet screw system designed for percutaneoous spine surgery, a minimally invasive procedure that uses small skin incisions without cutting the underlying muscle. Clinical data show such surgeries can help reduce the risk of complications and improve spinal fusion outcomes.
DePuy turned to Tissue Regeneration Systems Inc. to personalize healthcare solutions in Trauma, cherry picking the early-stage firm’s 3D printing and implant coating technologies for patient-specific, bioresorbable implant development. Tissue Regeneration Systems’ (TRS) devices are based on computed tomography (CT) scans for optimal customization and feature a strong, porous scaffold that integrates into bone as well as supports heavy loads during the healing process. The implants’ mineral coating (dubbed a “plate-like nanostructure”) resembles living tissue and helps regrow new bone.
“The acquisition of TRS technology by DePuy Synthes is a testament to our ability to identify and work collaboratively with promising early-stage companies and entrepreneurs to accelerate bringing innovative new products to market,” Robert G. Urban, global head of Johnson & Johnson Innovation, said in disclosing the April 2017 deal. “We are excited at the potential this technology holds to help improve patient outcomes.”
That excitement level rose precipitously last June upon DePuy’s entrance into the nerve localization market via its purchase of Innovative Surgical Solutions LLC, conducting business as Sentio LLC. The deal strengthened DePuy’s spine portfolio in both decompression and lateral operative procedures, and bolsters its ambulatory surgical reach, as Sentio’s platform has a minimal OR footprint. The system provides real-time nerve location feedback to surgeons using adhesive skin sensors; in animal models, Sentio’s technology found nerves faster than one competitive system and at lower levels of stimulation.
“We’ve been divesting slower-growth areas or areas that we think are better off in someone else’s hands and investing in higher growth areas and new technologies,” executive vice president and CFO Dominic J. Caruso told investors last spring. “Our entire medical devices business has largely been grown by these smaller, tuck-in, bolt-on acquisitions with technologies that advance the standard of care...”
Indeed, bolt-on acquisitions proved valuable to J&J last year, as did a 5.7 percent operational increase, 0.2 percent positive currency impact, and 21.3 percent spike in market share price. The company’s Medical Devices business also benefited from solid gains in three divisions (Cardiovascular, Vision, and Surgery) to achieve a 6 percent sales increase (to $26.6 billion).
The Cardiovascular and Surgery franchises posted double-digit growth (46 percent and 13.4 percent, respectively) but Orthopaedics barely budged from its 2016 total, slipping $76 million (0.8 percent) to $9.26 billion. Just two of the four franchise segments increased sales—Hips and Trauma—yet neither hike could completely offset stagnant knee and declining spine proceeds.
Hips revenue swelled 2.4 percent to $1.4 billion, aided in part by two summertime co-marketing agreements and the autumn launch of a hip repair system. One of the agreements covered JointPoint Inc.’s surgical navigation software platform, featuring non-invasive computer guidance, pre-operative digital templating, case planning, and feedback for total hip arthroplasty (THA). The other pact involved the ME1000 surgical impactor from Medical Enterprises Distribution LLC, a battery-powered device designed to replace handheld mallets used in THAs. The impactor delivers constant, stable energy to automate bone preparation, implant assembly, and positioning; it is only compatible with DePuy Synthes hip systems, with adapters available for anterior or posterior approaches.
Two months after negotiating the co-marketing agreements, DePuy Synthes launched its TFNA augmentation system, designed specifically for hip fracture patients with poor bone quality. The solution—billed as the “first and only” polymethylmethacrylate cement with indications for specific traumas—aims to enhance implant fixation in potential cut-out cases.
DePuy debuted its TFNA system around the same time it launched its Maxframe multi-axial limb correction solution for bone/soft tissue deformity treatment in the arm, leg, foot, or ankle.
Maxframe features 3D planning software that creates accurate patient treatment plans and reduces both the number of required X-rays and the procedural complexity of treatments.
Reduced procedural complexity is also the goal of DePuy’s PUREVUE Visualization System, launched in December 2017. Designed for minimally invasive endoscopic surgery, PUREVUE combines a light source, image processor, and local image management for a reduced operating room footprint; it also includes an autoclavable camera head, touch-screen navigation, and customizable settings for minimally invasive surgical procedures.
“With Americans living longer and seeking shorter recovery times, the call for minimally invasive procedures is accelerating,” Stephanie Chamberlain, vice president and global platform leader of DePuy Synthes Mitek Sports Medicine, said when the PUREVUE system was unveiled. “We are committed to bringing forward innovation that responds to evolving patient needs...”
One such innovation that also acknowledges surgeons’ needs is the TRUMATCH Titanium 3D-Printed maxillofacial implants for facial reconstruction and orthognathic (corrective jaw) procedures. Debuting in the United States last fall, the personalized implants are based on CT skull scans and the outcomes of computer-aided surgical planning. The devices are part of a total solution—TRUMATCH Orthognathics, and are the first 3D-printed titanium maxillofacial implants to receive U.S. Food and Drug Administration (FDA) clearance.
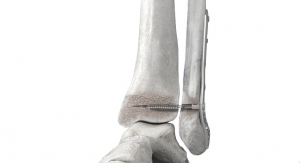
Another double-duty innovation introduced in 2017 was the Distal Radius Sterile Kit for wrist fracture treatment. The kit consists of pre-sterilized single-use instruments and implants, including the Variable Angle LCP Two-Column Volar Distal Radius Plate, sizing templates, T-8 screwdriver, 2.4mm Cortex and variable angle locking screws, L-Plates, and T-Plates. Besides eliminating sterilization costs, the kits are designed to drive repeatable, streamlined, more consistent procedures, and help improve operating room efficiency.
The Distal Radius kit was the only new product launch last year within the Trauma franchise, which increased sales 1.8 percent to $2.6 billion. The small gain, however, was neutralized by losses in Spine (down 4 percent to $3.72 billion) and Knees sales (sinking $1 million from 2016 to $1.52 billion). DePuy Synthes attributed the loss in spinal revenue to pricing and competitive pressures, but the deficit is somewhat surprising considering the number of product launches/reveals that occurred during 2017.
In February, DePuy Synthes—in collaboration with LifeNet Health—released its ViviGen Formable cellular bone matrix, a second-generation cellular allograft used for bone formation in spinal fusion surgery. The matrix consists of onsteoinductive, demineralized fibers that create a connected, putty-like consistency to achieve formable handling for open void cases such as posterolateral fusion. This handling reportedly allows surgeons to mold the allograft, helping the ViviGen Formable matrix to conform to the surgical site.
Four months after introducing the ViviGen matrix, DePuy Synthes launched the Viper and Expedium fenestrated screw systems in the United States (the products received FDA clearance in late January). When used with the company’s Confidence high-viscosity spinal cement, the screws can help stabilize spinal columns impaired by advanced stage tumors. Both systems have a hollow shaft and fenestrations above the screw tip to better control cement delivery into the vertebra and ensure immediate fixation; the products also are designed for open or percutaneous spinal fusion.
DePuy Synthes’ two other spinal product freshmen bowed in the final quarter of 2017: The VIPER PRIME System, which made its entrance during the EUROSPINE Congress in Ireland in October, combines multiple instruments into one pedicle screw inserter tool to reduce the number of instrument passes. The CONCORDE Clear MIS Discectomy Tool, meanwhile, hit the EMEA market in mid-December. CONCORDE allows surgeons to complete the degenerated disc-clearing process faster on average and more efficiently compared to traditional discectomy tools while increasing the amount of disc material removed.
“2017 was a year of transition in our Medical Devices business. We’ve taken a lot of action regarding innovation, we’ve taken a lot of action pruning our portfolio, and we’ve taken action as well to improve execution in the field,” Gorsky said in a January 2018 earnings call. “What we are pleased about is the fact that we saw an increase in the number of [product] launches, and improved execution.”
The increase in product launches, however, didn’t always result in higher sales. Knees revenue suffered a similar fate to Spine despite its various innovations in 2017. At the American Academy of Orthopaedic Surgeons (AAOS) Annual Meeting in March, Mitek Sports Medicine unveiled a new suite of knee arthroscopy solutions to improve OR efficiency, simplify ACL and meniscus repair, and make the surgeries more reproducible. The platform included:
The SPEEDTRAP Graft Prep System, which allows surgeons to easily and quickly (within 20 seconds) whipstitch, or suture, one tendon end without using a needle. The timeframe is at least 77 percent faster than traditional techniques, according to DePuy Synthes, and offers strong tension.
The TRUESPAN Meniscal Repair System, which streamlines arthroscopic meniscal repair through a unique delivery system featuring an ergonomic handle, single trigger, and auto-reloading mechanism for quick, simple one-handed use. The TRUESPAN System also features the only 24-degree curved needle option on the market, which may provide better access to challenging tear locations.
The TWISTR Retrograde Reamer, used to drill the tunnels for graft placement during ACL reconstruction. The TWISTR Reamer can be set to drill 13 different tunnel diameters making it the only one-size-fits-all device of its kind on the market. The adjustable design simplifies inventory management by eliminating the need to stock multiple reamer sizes and helps reduce cost during cases where more than one size reamer is needed.
The RIGIDLOOP Adjustable Cortical System, a titanium cortical button and adjustable loop implant that holds a graft in place in the femoral tunnel. The design with adjustable loops eliminates need for multiple size implants. Its one-handed tensioning technique allows surgeons to advance graft to completely fill the socket with stronger fixation and less graft displacement compared to similar adjustable loop devices.
The INTRAFIX ADVANCE Tibial Fastener System provides rigid fixation of the graft in the tibial tunnel. The sheath and screw implant duo is designed to protect soft tissue grafts and promote integration with the surrounding bone to achieve a strong and stable fixation.
DePuy also showcased its ATTUNE Revision Knee System at AAOS. The System features the GRADIUS Curve (to address femur-tibia sliding) and GLIDERIGHT Articulation, which enables the ATTUNE design to mimic the normal patellofemoral kinematics of a human knee.
Any hopes DePuy executives had for ATTUNE’s revenue-generating potential ended abruptly in the spring after a Journal of Knee Surgery study showed a high rate of ATTUNE tibial loosening at the implant-cement interface. Researchers identified 15 cases from three hospitals and referenced 21 additional reports of tibial loosening in the MAUDE database within a two-month period as well as “numerous other tibial failures” also reported without the mechanism for failure specified.
DePuy, of course, questioned the study’s accuracy, noting the absence of a rate calculation and radiographic technique or positioning. The company also took issue with the researchers’ use of MAUDE database statistics, claiming it conflicts with the intent of the database and FDA guidance. “The FDA’s guidance is that, ‘MDR data alone cannot be used to establish rates of events, evaluate a change in event rates over time or compare event rates between devices. The number of reports cannot be interpreted or used in isolation to reach conclusions about the existence, severity, or frequency of problems associated with devices,’ “DePuy said in a formal response to the study.
More bad publicity surfaced for ATTUNE in the fall with the filing of a federal lawsuit alleging the system to be defective and “unreasonably dangerous.” The Louisiana patient who filed the suit attributed ATTUNE’s defect to mechanical loosening, which typically is caused by a weakened or failing bond between the tibial baseplate and implant-cement surface. When such loosening occurs, the artificial knee can detach from existing bone, resulting in failure.
Although DePuy did not respond directly to the lawsuit, the company has released volumes of ATTUNE study data confirming low revision rates and high implant survivorship. Statistics from the National Joint Registry for England, Wales, Northern Ireland and the Isle of Man, for example, show an estimated cumulative revision rate of 1.3 percent at four years (98.7 percent implant survivorship) for 10,605 ATTUNE knees, which compares favorably to the 1.9 percent cumulative revision rate (98.1 percent implant survivorship) for the overall class of knee replacements.
In addition, a radiostereometric (RSA) analysis conducted by the Canadian RSA Network found the ATTUNE tibial base to be stable, moving only 0.17 mm between one and two years. The results are consistent with implants that have acceptable revision rates due to aseptic loosening. A second, randomized-controlled RSA study conducted in the Netherlands compared ATTUNE implant migration to patients implanted with SIGMA knees and reported no difference in implant migration.
KEY EXECUTIVES:
Alex Gorsky, Chairman and CEO
Dominic J. Caruso, Exec. VP and CFO
Jennifer Taubert, Company Group Chairman, Johnson & Johnson
Ashley McEvoy, Exec. VP, Worldwide Chairman, Medical Devices
Sandra E. Peterson, Exec. VP, Group Worldwide Chairman
Kathryn E. Wengel, Exec. VP, Chief Global Supply Chain Officer
Paulus Stoffels, Exec. VP, Chief Scientific Officer
Michael H. Ullmann, Exec. VP, General Counsel
Ronald A. Kapusta, Corporate Controller, Chief Accounting Officer
Number OF EMPLOYEES: 18,000
GLOBAL HEADQUARTERS: Warsaw, Ind.; Raynham, Mass.; and West Chester, Pa.
Some companies chase the future. Others shape it.
Apple clearly falls into the latter category. From its first computers—the rudimentary but revolutionary pre-assembled Apple 1 and graphical user interface-touting Lisa (the one that introduced the “mouse”)—to the pocket-sized iPod, touch computing iPhone and netbook-killing iPad, Apple truly has changed the way humans socialize and communicate.
Google and Tesla are top morrow moulders as well. The former entity essentially cataloged the Web, giving structure to the jumbled mess of digital data that once littered the information superhighway. Its algorithms and index updates over the last 20 years have turned the tech giant into the go-to source of information (and quite a popular transitive verb). Tesla, on the other hand, is transforming the auto industry with its electric cars and autonomous (self-driving) vehicles, while also becoming a poster child for sustainability (its Powerwall rechargeable home battery system is guaranteed to store 70 percent of its rated power output at 10 years).
Johnson & Johnson considers itself a future forger too. Actually, the company’s chief executive claims the multinational conglomerate has dedicated its life to defining healthcare’s future. “Through the decades, Johnson & Johnson has advanced with the evolution of science and technology...” Chairman and CEO Alex Gorsky reminds shareholders at the start of the firm’s 2017 annual report. “In fact, every year for the past 132 years, Johnson & Johnson has been involved in defining the future of healthcare.”
Maybe not every year, admittedly. But certainly more often than not, J&J has been at the forefront of significant medical breakthroughs: It introduced the world to sterile surgical dressings and sutures (1887); first aid kits (1888); affordable dental floss (1898); BAND-AID adhesive bandages (1921); synthetic sterile sutures (1969); a disposable skin stapler (1978); coated sutures (1979); extended-wear disposable contact lenses (1987); minimally invasive gall bladder removal (1988); low-temperature gas plasma sterilization (1992); and a topical skin adhesive glue (1998).
The company picked up the pace of innovation with the millennium’s arrival, developing a coated antibacterial suture (2003); silicone gel implants (2006); a tissue expander with suturing tabs (2008); and a single-handed balloon sinuplasty device (2012), among others. Over the last few years, however, J&J has somewhat shifted its focus to better target (and shape) both digital healthcare and value-based medicine. In 2015, for example, the company teamed up with IBM’s Watson Health unit to create consumer-focused virtual coaching apps for knee replacement patients. Using Watson’s cognitive computing, the app predicts outcomes, suggests treatment plans, and provides personalized encouragement to patients during recovery.
Through its Ethicon subsidiary, J&J also has partnered with Alphabet Inc.’s Verily Life Sciences to develop a new robotic-assisted surgical solutions platform for improved access to minimally invasive surgery, greater hospital efficiency, and enhanced patient outcomes. Since the pairing, the partners have developed a surgical platform prototype encompassing robotics, visualization, advanced instrumentation, data analytics, and connectivity—an innovation the duo is calling “Surgery 4.0.”
J&J’s value-based healthcare initiatives aim to ease the transition to alternative reimbursement models and reinforce patient engagement, preventative care, and operational efficiency. Its CareAdvantage initiative is a holistic, data-driven approach designed to help companies shift to payment models tied to quality and/or value. The plan uses data-driven analysis and insights, on-site assessments, and focused action plans to improve operational efficiency in various medical fields—including orthopedics—based on goal-specific targeted metrics.
CareAdvantage’s orthopedic component combines a cloud-based platform for patient interaction; episode data analysis; infection risk reduction best practices; pre- and post-operative care (setting) expectations; and a standardized hip fracture care program. Its Patient Athlete plan helps patients prepare for and recover from orthopedic surgery through science-based principles from the Johnson & Johnson Human Performance Institute.
“...we are convinced that technology will not replace but rather will extend human caring...Ahead we see a future that lies beyond the intersection of science and technology,” Gorsky wrote in his shareholder letter. “We are rocketing toward that future, toward the very edge of imagination and possibility. That’s exciting, of course, and just a little daunting at the same time. Getting to that future will require urgency, boldness and vision—characteristics most often attributed to very successful startups. However, I believe it is also a great description of this company today—a 132-year-old startup.”
Not many “startups,” however, are as fiscally fruitful as J&J. Last year, the New Brunswick, N.J.-based company increased its total revenue 6.3 percent to $76.4 billion and grew its per-share stock price by $24.51, or 21.3 percent (ending 2017 at $139.72). Gains occurred in each of J&J’s three product franchises, with Pharmaceutical posting the largest rise (8.3 percent) followed by Medical Devices (5.9 percent) and Consumer (2.2 percent).
The sales surge in Medical Devices is a redemption of sorts, for past franchise sins. Declining revenue in recent years prompted the company to implement a massive restructuring in 2016 and replace its slower-growing businesses with more profitable technologies—a strategy that surely induced the $1.045 billion sale of DePuy Synthes’ Codman Neurosurgery business last February and the $4.33 billion purchase of Abbott Medical Optics in September 2016. The swap-out scheme also was responsible for a handful of smaller bolt-on acquisitions last year that augmented DePuy Synthes’ minimally invasive surgical solutions portfolio and expanded its capabilities in expandable spinal cage technology, 3D printing, and bone defect remedies.
The spinal cage technology came from Interventional Spine Inc., an Irvine, Calif.-based developer of expandable cages and minimally invasive surgical techniques. The company’s cage technology provides optimal tactile feedback for surgeons and features an adjustable design that customizes the device height to individual patient anatomy. The implants are designed to maximize bone graft contact and containment, which helps promote bone growth and fusion. In addition to the expandable cage technology, DePuy picked up Interventional Spine’s facet screw system designed for percutaneoous spine surgery, a minimally invasive procedure that uses small skin incisions without cutting the underlying muscle. Clinical data show such surgeries can help reduce the risk of complications and improve spinal fusion outcomes.
DePuy turned to Tissue Regeneration Systems Inc. to personalize healthcare solutions in Trauma, cherry picking the early-stage firm’s 3D printing and implant coating technologies for patient-specific, bioresorbable implant development. Tissue Regeneration Systems’ (TRS) devices are based on computed tomography (CT) scans for optimal customization and feature a strong, porous scaffold that integrates into bone as well as supports heavy loads during the healing process. The implants’ mineral coating (dubbed a “plate-like nanostructure”) resembles living tissue and helps regrow new bone.
“The acquisition of TRS technology by DePuy Synthes is a testament to our ability to identify and work collaboratively with promising early-stage companies and entrepreneurs to accelerate bringing innovative new products to market,” Robert G. Urban, global head of Johnson & Johnson Innovation, said in disclosing the April 2017 deal. “We are excited at the potential this technology holds to help improve patient outcomes.”
That excitement level rose precipitously last June upon DePuy’s entrance into the nerve localization market via its purchase of Innovative Surgical Solutions LLC, conducting business as Sentio LLC. The deal strengthened DePuy’s spine portfolio in both decompression and lateral operative procedures, and bolsters its ambulatory surgical reach, as Sentio’s platform has a minimal OR footprint. The system provides real-time nerve location feedback to surgeons using adhesive skin sensors; in animal models, Sentio’s technology found nerves faster than one competitive system and at lower levels of stimulation.
“We’ve been divesting slower-growth areas or areas that we think are better off in someone else’s hands and investing in higher growth areas and new technologies,” executive vice president and CFO Dominic J. Caruso told investors last spring. “Our entire medical devices business has largely been grown by these smaller, tuck-in, bolt-on acquisitions with technologies that advance the standard of care...”
Indeed, bolt-on acquisitions proved valuable to J&J last year, as did a 5.7 percent operational increase, 0.2 percent positive currency impact, and 21.3 percent spike in market share price. The company’s Medical Devices business also benefited from solid gains in three divisions (Cardiovascular, Vision, and Surgery) to achieve a 6 percent sales increase (to $26.6 billion).
The Cardiovascular and Surgery franchises posted double-digit growth (46 percent and 13.4 percent, respectively) but Orthopaedics barely budged from its 2016 total, slipping $76 million (0.8 percent) to $9.26 billion. Just two of the four franchise segments increased sales—Hips and Trauma—yet neither hike could completely offset stagnant knee and declining spine proceeds.
Hips revenue swelled 2.4 percent to $1.4 billion, aided in part by two summertime co-marketing agreements and the autumn launch of a hip repair system. One of the agreements covered JointPoint Inc.’s surgical navigation software platform, featuring non-invasive computer guidance, pre-operative digital templating, case planning, and feedback for total hip arthroplasty (THA). The other pact involved the ME1000 surgical impactor from Medical Enterprises Distribution LLC, a battery-powered device designed to replace handheld mallets used in THAs. The impactor delivers constant, stable energy to automate bone preparation, implant assembly, and positioning; it is only compatible with DePuy Synthes hip systems, with adapters available for anterior or posterior approaches.
Two months after negotiating the co-marketing agreements, DePuy Synthes launched its TFNA augmentation system, designed specifically for hip fracture patients with poor bone quality. The solution—billed as the “first and only” polymethylmethacrylate cement with indications for specific traumas—aims to enhance implant fixation in potential cut-out cases.
DePuy debuted its TFNA system around the same time it launched its Maxframe multi-axial limb correction solution for bone/soft tissue deformity treatment in the arm, leg, foot, or ankle.
Maxframe features 3D planning software that creates accurate patient treatment plans and reduces both the number of required X-rays and the procedural complexity of treatments.
Reduced procedural complexity is also the goal of DePuy’s PUREVUE Visualization System, launched in December 2017. Designed for minimally invasive endoscopic surgery, PUREVUE combines a light source, image processor, and local image management for a reduced operating room footprint; it also includes an autoclavable camera head, touch-screen navigation, and customizable settings for minimally invasive surgical procedures.
“With Americans living longer and seeking shorter recovery times, the call for minimally invasive procedures is accelerating,” Stephanie Chamberlain, vice president and global platform leader of DePuy Synthes Mitek Sports Medicine, said when the PUREVUE system was unveiled. “We are committed to bringing forward innovation that responds to evolving patient needs...”
One such innovation that also acknowledges surgeons’ needs is the TRUMATCH Titanium 3D-Printed maxillofacial implants for facial reconstruction and orthognathic (corrective jaw) procedures. Debuting in the United States last fall, the personalized implants are based on CT skull scans and the outcomes of computer-aided surgical planning. The devices are part of a total solution—TRUMATCH Orthognathics, and are the first 3D-printed titanium maxillofacial implants to receive U.S. Food and Drug Administration (FDA) clearance.
Another double-duty innovation introduced in 2017 was the Distal Radius Sterile Kit for wrist fracture treatment. The kit consists of pre-sterilized single-use instruments and implants, including the Variable Angle LCP Two-Column Volar Distal Radius Plate, sizing templates, T-8 screwdriver, 2.4mm Cortex and variable angle locking screws, L-Plates, and T-Plates. Besides eliminating sterilization costs, the kits are designed to drive repeatable, streamlined, more consistent procedures, and help improve operating room efficiency.
The Distal Radius kit was the only new product launch last year within the Trauma franchise, which increased sales 1.8 percent to $2.6 billion. The small gain, however, was neutralized by losses in Spine (down 4 percent to $3.72 billion) and Knees sales (sinking $1 million from 2016 to $1.52 billion). DePuy Synthes attributed the loss in spinal revenue to pricing and competitive pressures, but the deficit is somewhat surprising considering the number of product launches/reveals that occurred during 2017.
In February, DePuy Synthes—in collaboration with LifeNet Health—released its ViviGen Formable cellular bone matrix, a second-generation cellular allograft used for bone formation in spinal fusion surgery. The matrix consists of onsteoinductive, demineralized fibers that create a connected, putty-like consistency to achieve formable handling for open void cases such as posterolateral fusion. This handling reportedly allows surgeons to mold the allograft, helping the ViviGen Formable matrix to conform to the surgical site.
Four months after introducing the ViviGen matrix, DePuy Synthes launched the Viper and Expedium fenestrated screw systems in the United States (the products received FDA clearance in late January). When used with the company’s Confidence high-viscosity spinal cement, the screws can help stabilize spinal columns impaired by advanced stage tumors. Both systems have a hollow shaft and fenestrations above the screw tip to better control cement delivery into the vertebra and ensure immediate fixation; the products also are designed for open or percutaneous spinal fusion.
DePuy Synthes’ two other spinal product freshmen bowed in the final quarter of 2017: The VIPER PRIME System, which made its entrance during the EUROSPINE Congress in Ireland in October, combines multiple instruments into one pedicle screw inserter tool to reduce the number of instrument passes. The CONCORDE Clear MIS Discectomy Tool, meanwhile, hit the EMEA market in mid-December. CONCORDE allows surgeons to complete the degenerated disc-clearing process faster on average and more efficiently compared to traditional discectomy tools while increasing the amount of disc material removed.
“2017 was a year of transition in our Medical Devices business. We’ve taken a lot of action regarding innovation, we’ve taken a lot of action pruning our portfolio, and we’ve taken action as well to improve execution in the field,” Gorsky said in a January 2018 earnings call. “What we are pleased about is the fact that we saw an increase in the number of [product] launches, and improved execution.”
The increase in product launches, however, didn’t always result in higher sales. Knees revenue suffered a similar fate to Spine despite its various innovations in 2017. At the American Academy of Orthopaedic Surgeons (AAOS) Annual Meeting in March, Mitek Sports Medicine unveiled a new suite of knee arthroscopy solutions to improve OR efficiency, simplify ACL and meniscus repair, and make the surgeries more reproducible. The platform included:
The SPEEDTRAP Graft Prep System, which allows surgeons to easily and quickly (within 20 seconds) whipstitch, or suture, one tendon end without using a needle. The timeframe is at least 77 percent faster than traditional techniques, according to DePuy Synthes, and offers strong tension.
The TRUESPAN Meniscal Repair System, which streamlines arthroscopic meniscal repair through a unique delivery system featuring an ergonomic handle, single trigger, and auto-reloading mechanism for quick, simple one-handed use. The TRUESPAN System also features the only 24-degree curved needle option on the market, which may provide better access to challenging tear locations.
The TWISTR Retrograde Reamer, used to drill the tunnels for graft placement during ACL reconstruction. The TWISTR Reamer can be set to drill 13 different tunnel diameters making it the only one-size-fits-all device of its kind on the market. The adjustable design simplifies inventory management by eliminating the need to stock multiple reamer sizes and helps reduce cost during cases where more than one size reamer is needed.
The RIGIDLOOP Adjustable Cortical System, a titanium cortical button and adjustable loop implant that holds a graft in place in the femoral tunnel. The design with adjustable loops eliminates need for multiple size implants. Its one-handed tensioning technique allows surgeons to advance graft to completely fill the socket with stronger fixation and less graft displacement compared to similar adjustable loop devices.
The INTRAFIX ADVANCE Tibial Fastener System provides rigid fixation of the graft in the tibial tunnel. The sheath and screw implant duo is designed to protect soft tissue grafts and promote integration with the surrounding bone to achieve a strong and stable fixation.
DePuy also showcased its ATTUNE Revision Knee System at AAOS. The System features the GRADIUS Curve (to address femur-tibia sliding) and GLIDERIGHT Articulation, which enables the ATTUNE design to mimic the normal patellofemoral kinematics of a human knee.
Any hopes DePuy executives had for ATTUNE’s revenue-generating potential ended abruptly in the spring after a Journal of Knee Surgery study showed a high rate of ATTUNE tibial loosening at the implant-cement interface. Researchers identified 15 cases from three hospitals and referenced 21 additional reports of tibial loosening in the MAUDE database within a two-month period as well as “numerous other tibial failures” also reported without the mechanism for failure specified.
DePuy, of course, questioned the study’s accuracy, noting the absence of a rate calculation and radiographic technique or positioning. The company also took issue with the researchers’ use of MAUDE database statistics, claiming it conflicts with the intent of the database and FDA guidance. “The FDA’s guidance is that, ‘MDR data alone cannot be used to establish rates of events, evaluate a change in event rates over time or compare event rates between devices. The number of reports cannot be interpreted or used in isolation to reach conclusions about the existence, severity, or frequency of problems associated with devices,’ “DePuy said in a formal response to the study.
More bad publicity surfaced for ATTUNE in the fall with the filing of a federal lawsuit alleging the system to be defective and “unreasonably dangerous.” The Louisiana patient who filed the suit attributed ATTUNE’s defect to mechanical loosening, which typically is caused by a weakened or failing bond between the tibial baseplate and implant-cement surface. When such loosening occurs, the artificial knee can detach from existing bone, resulting in failure.
Although DePuy did not respond directly to the lawsuit, the company has released volumes of ATTUNE study data confirming low revision rates and high implant survivorship. Statistics from the National Joint Registry for England, Wales, Northern Ireland and the Isle of Man, for example, show an estimated cumulative revision rate of 1.3 percent at four years (98.7 percent implant survivorship) for 10,605 ATTUNE knees, which compares favorably to the 1.9 percent cumulative revision rate (98.1 percent implant survivorship) for the overall class of knee replacements.
In addition, a radiostereometric (RSA) analysis conducted by the Canadian RSA Network found the ATTUNE tibial base to be stable, moving only 0.17 mm between one and two years. The results are consistent with implants that have acceptable revision rates due to aseptic loosening. A second, randomized-controlled RSA study conducted in the Netherlands compared ATTUNE implant migration to patients implanted with SIGMA knees and reported no difference in implant migration.