08.14.19
AT A GLANCE
Rank: #8 (Last year: #8)
$713 Million
Prior Fiscal: $636 Million
Percentage Change: +12.1%
No. of Employees: 1,800
Global Headquarters: Audubon, Pa.
KEY EXECUTIVES:
David C. Paul, Exec. Chairman
David M. Demski, CEO
Daniel T. Scavilla, Sr. VP and CFO
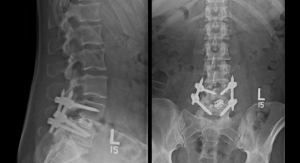
Globus Medical’s ExcelsiusGPS is a lesser-known surgical robot but is undoubtedly significant—at launch, it was the only robotic system combining surgical navigation and robotic guidance for spinal surgery. It supports both minimally invasive (MIS) and open screw placement procedures, seamlessly integrating Globus implants and instruments. ExcelsiusGPS is compatible with CT, intra-operative CT, and fluoroscopic imaging, and minimizes radiation exposure, streamlines workflow, and reproducibly assists in implant placement.
The first procedures using ExcelsiusGPS—performed at The Johns Hopkins Hospital in Baltimore, Md., and St. Mark’s Hospital in Salt Lake City, Utah—took place in October 2017. Last June, Globus recorded its 3,000th successful screw implantation using the system for a variety of spine procedures.
“Having completed installations and surgeon training programs through the first half of 2018, we see surgeons using this technology in open and minimally invasive spine procedures including posterior cervical fusion, sacroiliac joint fusion, and thoracolumbar fusion with posterior and single position lateral approaches,” Globus CEO Dave Demski told the press.
Globus made its purchase of Nemaris Inc., developer of the Surgimap surgical software platform, with ExcelsiusGPS in mind last September. Surgimap lets users plan and simulate potential surgical outcomes in the course of treating complex deformities through a cloud-based infrastructure with predictive algorithms and visual guides. Healthcare professionals can also share medical imaging globally. Using the software platform in tandem with ExcelsiusGPS both streamlines workflow and enables superior data analytics during spine surgery.
“Software-enhanced solutions are critical components of the future of computer-assisted surgery, and Surgimap is the leading surgical planning software technology in the spine market today,” Demski told the press. “We are very excited about the strategic fit of this acquisition, which will allow us to further enhance the capability of our ExcelsiusGPS navigation and robotic system to assist surgeons in planning complex spine procedures and placing screws more accurately and less invasively.”
ExcelsiusGPS first crossed the Atlantic last December for a procedure at the Metropolitan Hospital in Neo Faliro, Athens, Greece. System installations were also completed in several European countries, including Italy and Germany. The computer-assisted surgical system is a significant portion of Globus’ enabling technologies franchise, which is comprised of imaging, navigation, and robotic-assisted surgery solutions. The segment garnered $47 million in sales last year, skyrocketing 330 percent over the prior year due to ExcelsiusGPS’ U.S. launch in the fourth quarter of 2017.
This figure is a small but significant portion of Globus’ total revenue, which swelled 12.1 percent last year to $713 million. Increased spine product sales were the primary driver for the $64 million sales hike. Penetration in existing territories, navigation and robotic surgery solutions proceeds, and associated implant and robotic instrument sales helped reinforce spine revenue. Globus drew $594 million of its revenue from U.S. sales and $119 million from international; earnings from those regions rose 12.1 percent and 12.2 percent respectively.
Globus’ musculoskeletal solutions business is comprised of implantable devices, biologics, accessories, and surgical instruments used in spinal, orthopedic, and neurosurgical procedures. The segment generated a $666 million payoff, growing 6.5 percent over the prior year.
Globus proclaimed its entry into the orthopedic trauma market by showcasing 11 recently FDA-cleared products at the American Association of Orthopaedic Surgeons’ annual meeting. These include fracture plates, compression screws, cannulated screws, intramedullary nails, and external fixation solutions. The portfolio can treat a wide variety of fracture patterns, and accommodates anatomies in the upper and lower extremities as well as hip and long bones.
“It has been impressive watching Globus move from design to development to deployment of their trauma solutions line,” Dr. Andrew N. Pollak, Chief of Orthopaedics at the University of Maryland Medical System, told the press. “I have been very impressed with the ability of Globus’ engineers to translate surgeon ideas and concepts into functional and easy-to- use implants for a wide variety of fracture patterns.”
Globus introduced two lateral expandable interbody spacers with integrated screw fixation—ELSA AL and ELSA-ATP—last April. ELSA AL improves sagittal balance and improves stability with adjustable lordotic technology that allows up to 30 degrees of lordosis while maintaining an MIS corridor. It also exhibits less disruption compared to static spacers, according to the company.
The ELSA-ATP expandable spacer utilizes a modified lateral lumbar interbody fusion (LLIF) procedure called anterior-to-psoas for placement. The tweaked LLIF approach is used to avoid the psoas, a large muscle responsible for leg movement and flexion; avoiding this muscle and its integrated nerves can help reduce post-op complications like thigh pain. ELSA-ATP’s angled integrated screws provide direct fixation to adjacent vertebrae. The spacer can be inserted at a minimal height and expanded in-situ to minimize tissue disruption and maximize indirect compression.
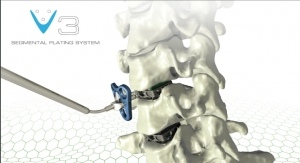
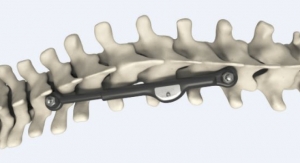
Last June saw the launch of the CREO fenestrated screw system, which is used to help treat advanced-stage thoracolumbar spinal tumors. The fenestrated screws can be combined with the firm’s FORTRESS radiopaque bone cement to restore the spine’s structural integrity at vertebral levels impacted by metastatic bone disease. CREO fenestrated screws are available with threaded, non-threaded, and MIS screw head options to accommodate surgeon preferences, and are cannulated with a series of fenestrations along the shank for targeted cement dispersion. They also rigidly attach to specialized instruments for controlled cement delivery.
Globus added two new products to its growing trauma portfolio last July. The ANTHEM ankle fracture system offers seven plating options to treat virtually any ankle fracture, and according to the company, is over 25 percent thinner than the market leader’s plate. ANTHEM’s low-profile plates help minimize soft tissue irritation from implant prominence. The efficient and comprehensive system rolls anatomically contoured plates, extensive screw options, and ankle-specific instrumentation into one.
The ANTHEM proximal humerus fracture system to treat shoulder fractures touts polyaxial screw technology, which permits more accurate dense calcar bone targeting to enhance fixation independent of plate position. Large suture holes simplify suture attachment for soft tissue or rotator cuff repair. Small fragment instruments and retractors help streamline the procedure and aid in fracture site visibility.
Last Halloween, the Globus subsidiary Human Biologics of Texas (HBT), which produces the company’s ViaCell allograft, received an FDA warning letter concerning issues found during a facility inspection last April. In the review, FDA inspectors cited failures to document and investigate ViaCell production deviations, as well as trends of any variations away from core current good tissue practice requirements. HBT responded by enacting additional validation activities last May, but the FDA took issue with the company’s reliance on sterility testing and possible contamination during processing.
The FDA also slammed HBT for not excluding or discarding samples from donors positive for Clostridium, Streptococcus pyogenes, or “any other microorganism that you have determined to be difficult to eliminate, unless you have a terminal sterilization process validated to a sterility assurance level of 10-6.” Finally, the agency referenced a lack of positive results from all environmental and personnel monitoring sample testing, claiming it was “highly unusual, especially when your sterility failure rates were so high.” The FDA gave HBT 15 days to respond to the issues. (No further information has come to light regarding HBT at this time.)
Rank: #8 (Last year: #8)
$713 Million
Prior Fiscal: $636 Million
Percentage Change: +12.1%
No. of Employees: 1,800
Global Headquarters: Audubon, Pa.
KEY EXECUTIVES:
David C. Paul, Exec. Chairman
David M. Demski, CEO
Daniel T. Scavilla, Sr. VP and CFO
Globus Medical’s ExcelsiusGPS is a lesser-known surgical robot but is undoubtedly significant—at launch, it was the only robotic system combining surgical navigation and robotic guidance for spinal surgery. It supports both minimally invasive (MIS) and open screw placement procedures, seamlessly integrating Globus implants and instruments. ExcelsiusGPS is compatible with CT, intra-operative CT, and fluoroscopic imaging, and minimizes radiation exposure, streamlines workflow, and reproducibly assists in implant placement.
The first procedures using ExcelsiusGPS—performed at The Johns Hopkins Hospital in Baltimore, Md., and St. Mark’s Hospital in Salt Lake City, Utah—took place in October 2017. Last June, Globus recorded its 3,000th successful screw implantation using the system for a variety of spine procedures.
“Having completed installations and surgeon training programs through the first half of 2018, we see surgeons using this technology in open and minimally invasive spine procedures including posterior cervical fusion, sacroiliac joint fusion, and thoracolumbar fusion with posterior and single position lateral approaches,” Globus CEO Dave Demski told the press.
Globus made its purchase of Nemaris Inc., developer of the Surgimap surgical software platform, with ExcelsiusGPS in mind last September. Surgimap lets users plan and simulate potential surgical outcomes in the course of treating complex deformities through a cloud-based infrastructure with predictive algorithms and visual guides. Healthcare professionals can also share medical imaging globally. Using the software platform in tandem with ExcelsiusGPS both streamlines workflow and enables superior data analytics during spine surgery.
“Software-enhanced solutions are critical components of the future of computer-assisted surgery, and Surgimap is the leading surgical planning software technology in the spine market today,” Demski told the press. “We are very excited about the strategic fit of this acquisition, which will allow us to further enhance the capability of our ExcelsiusGPS navigation and robotic system to assist surgeons in planning complex spine procedures and placing screws more accurately and less invasively.”
ExcelsiusGPS first crossed the Atlantic last December for a procedure at the Metropolitan Hospital in Neo Faliro, Athens, Greece. System installations were also completed in several European countries, including Italy and Germany. The computer-assisted surgical system is a significant portion of Globus’ enabling technologies franchise, which is comprised of imaging, navigation, and robotic-assisted surgery solutions. The segment garnered $47 million in sales last year, skyrocketing 330 percent over the prior year due to ExcelsiusGPS’ U.S. launch in the fourth quarter of 2017.
This figure is a small but significant portion of Globus’ total revenue, which swelled 12.1 percent last year to $713 million. Increased spine product sales were the primary driver for the $64 million sales hike. Penetration in existing territories, navigation and robotic surgery solutions proceeds, and associated implant and robotic instrument sales helped reinforce spine revenue. Globus drew $594 million of its revenue from U.S. sales and $119 million from international; earnings from those regions rose 12.1 percent and 12.2 percent respectively.
Globus’ musculoskeletal solutions business is comprised of implantable devices, biologics, accessories, and surgical instruments used in spinal, orthopedic, and neurosurgical procedures. The segment generated a $666 million payoff, growing 6.5 percent over the prior year.
Globus proclaimed its entry into the orthopedic trauma market by showcasing 11 recently FDA-cleared products at the American Association of Orthopaedic Surgeons’ annual meeting. These include fracture plates, compression screws, cannulated screws, intramedullary nails, and external fixation solutions. The portfolio can treat a wide variety of fracture patterns, and accommodates anatomies in the upper and lower extremities as well as hip and long bones.
“It has been impressive watching Globus move from design to development to deployment of their trauma solutions line,” Dr. Andrew N. Pollak, Chief of Orthopaedics at the University of Maryland Medical System, told the press. “I have been very impressed with the ability of Globus’ engineers to translate surgeon ideas and concepts into functional and easy-to- use implants for a wide variety of fracture patterns.”
Globus introduced two lateral expandable interbody spacers with integrated screw fixation—ELSA AL and ELSA-ATP—last April. ELSA AL improves sagittal balance and improves stability with adjustable lordotic technology that allows up to 30 degrees of lordosis while maintaining an MIS corridor. It also exhibits less disruption compared to static spacers, according to the company.
The ELSA-ATP expandable spacer utilizes a modified lateral lumbar interbody fusion (LLIF) procedure called anterior-to-psoas for placement. The tweaked LLIF approach is used to avoid the psoas, a large muscle responsible for leg movement and flexion; avoiding this muscle and its integrated nerves can help reduce post-op complications like thigh pain. ELSA-ATP’s angled integrated screws provide direct fixation to adjacent vertebrae. The spacer can be inserted at a minimal height and expanded in-situ to minimize tissue disruption and maximize indirect compression.
Last June saw the launch of the CREO fenestrated screw system, which is used to help treat advanced-stage thoracolumbar spinal tumors. The fenestrated screws can be combined with the firm’s FORTRESS radiopaque bone cement to restore the spine’s structural integrity at vertebral levels impacted by metastatic bone disease. CREO fenestrated screws are available with threaded, non-threaded, and MIS screw head options to accommodate surgeon preferences, and are cannulated with a series of fenestrations along the shank for targeted cement dispersion. They also rigidly attach to specialized instruments for controlled cement delivery.
Globus added two new products to its growing trauma portfolio last July. The ANTHEM ankle fracture system offers seven plating options to treat virtually any ankle fracture, and according to the company, is over 25 percent thinner than the market leader’s plate. ANTHEM’s low-profile plates help minimize soft tissue irritation from implant prominence. The efficient and comprehensive system rolls anatomically contoured plates, extensive screw options, and ankle-specific instrumentation into one.
The ANTHEM proximal humerus fracture system to treat shoulder fractures touts polyaxial screw technology, which permits more accurate dense calcar bone targeting to enhance fixation independent of plate position. Large suture holes simplify suture attachment for soft tissue or rotator cuff repair. Small fragment instruments and retractors help streamline the procedure and aid in fracture site visibility.
Last Halloween, the Globus subsidiary Human Biologics of Texas (HBT), which produces the company’s ViaCell allograft, received an FDA warning letter concerning issues found during a facility inspection last April. In the review, FDA inspectors cited failures to document and investigate ViaCell production deviations, as well as trends of any variations away from core current good tissue practice requirements. HBT responded by enacting additional validation activities last May, but the FDA took issue with the company’s reliance on sterility testing and possible contamination during processing.
The FDA also slammed HBT for not excluding or discarding samples from donors positive for Clostridium, Streptococcus pyogenes, or “any other microorganism that you have determined to be difficult to eliminate, unless you have a terminal sterilization process validated to a sterility assurance level of 10-6.” Finally, the agency referenced a lack of positive results from all environmental and personnel monitoring sample testing, claiming it was “highly unusual, especially when your sterility failure rates were so high.” The FDA gave HBT 15 days to respond to the issues. (No further information has come to light regarding HBT at this time.)