09.15.15
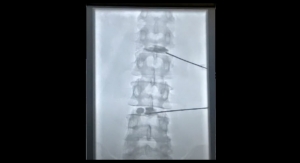
Wright Medical Group Inc. received an approval from the U.S. Food and Drug Administration (FDA) for the premarket approval application (PMA) of its Augment bone graft. The decision means the agency feels the Augment bone graft is safe and effective as an alternative to autograft for ankle and/or hindfoot fusion indications. Wright can now initiate commercial sale and distribution of the Augment bone graft in the United States.
“The FDA approval of Augment marks a capstone achievement that demonstrates the strength of our science and provides a breakthrough therapeutic option as an alternative to autograft in ankle and hindfoot fusion procedures,” said Robert Palmisano, president and CEO of Wright. “We will begin commercial sale and distribution of Augment in the U.S. and believe this product, as well as the PDGF (platelet-derived growth factor) technology platform, will be important drivers of the long-term growth of our business for years to come.”
The decision also is a vindication of sorts for the company, which had its PMA for Augment rejected in August 2013. At the time, agency officials said that the population enrolled in Wright Medical’s clinical trial was too low-risk and may not have warranted the use of either autograft or Augment bone graft. FDA officials said they would re-evaluate the use of Augment after a new clinical trial that evaluated the use of the product in a high-risk target population, where the use of an autograft transfer would be clinically warranted.
Wright Medical acquired Augment as part of its $380 million purchase of Nashville, Tenn.-based BioMimetic Therapeutics Inc. in November 2012.
The company continues to anticipate U.S. Augment revenue to be in the range of $10 million to $12 million in the first seven to eight months post-approval. Revenue is expected to build gradually during the first six months following the launch of the product as activities, such as review by hospital value analysis committees and physician education, are initiated, officials said. The company expects to begin selling product in the United States in the next several weeks once inventory is moved into its U.S. distribution network.
“This approval also underscores the significant effort and perseverance from our clinical trial investigators and Wright’s clinical, regulatory and legal teams to bring the product to market,” Palmisano added. “This success can be attributed to everyone involved in the clinical trial, especially the study investigators and coordinators; our R&D, regulatory and clinical teams; our partners and suppliers; and most importantly, the patients who participated in the landmark prospective, randomized study comparing Augment bone graft to autograft.”
Christopher DiGiovanni, M.D., lead U.S. investigator for Augment’s American pivotal trial in North America and chief of the foot and ankle service in the department of orthopedic surgery at Massachusetts General Hospital, Harvard Medical School, said: “The FDA approval of Augment provides a valuable new therapeutic healing option as an alternative to autograft in ankle and/or hindfoot fusion procedures, which is especially important since the outcomes of these interventions can at times be complicated by delayed union or non-union. This approval is based on strong clinical data showing that Augment offers clear patient benefit by enabling a healing rate and safety profile equivalent to autogenous bone graft—while simultaneously avoiding the additional surgery required to harvest autograft bone graft tissue that can result in site-specific complications and/or prolonged harvest site pain in some patients.”
According to the company, Augment is the first clinically proven protein therapeutic to come to the U.S. orthopedics market in more than a decade, offering an alternative to autograft in ankle and/or hindfoot fusion procedures, which translates into an estimated market opportunity of approximately $300 million in the United States.
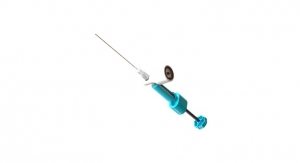
The combination of two components, recombinant human platelet-derived growth factor (rhPDGF) and beta-tricalcium phosphate (Beta-TCP), is key to the product’s activities. According to Wright officials, rhPDGF provides a biological stimulus for the recruitment and proliferation of cells, including osteoblasts, which are responsible for the formation of bone, while Beta-TCP provides a framework or scaffold for new bone growth to occur.
Based just outside Memphis, Tenn., Wright Medical Group is a specialty orthopedic company that provides extremity and biologic products.
“The FDA approval of Augment marks a capstone achievement that demonstrates the strength of our science and provides a breakthrough therapeutic option as an alternative to autograft in ankle and hindfoot fusion procedures,” said Robert Palmisano, president and CEO of Wright. “We will begin commercial sale and distribution of Augment in the U.S. and believe this product, as well as the PDGF (platelet-derived growth factor) technology platform, will be important drivers of the long-term growth of our business for years to come.”
The decision also is a vindication of sorts for the company, which had its PMA for Augment rejected in August 2013. At the time, agency officials said that the population enrolled in Wright Medical’s clinical trial was too low-risk and may not have warranted the use of either autograft or Augment bone graft. FDA officials said they would re-evaluate the use of Augment after a new clinical trial that evaluated the use of the product in a high-risk target population, where the use of an autograft transfer would be clinically warranted.
Wright Medical acquired Augment as part of its $380 million purchase of Nashville, Tenn.-based BioMimetic Therapeutics Inc. in November 2012.
The company continues to anticipate U.S. Augment revenue to be in the range of $10 million to $12 million in the first seven to eight months post-approval. Revenue is expected to build gradually during the first six months following the launch of the product as activities, such as review by hospital value analysis committees and physician education, are initiated, officials said. The company expects to begin selling product in the United States in the next several weeks once inventory is moved into its U.S. distribution network.
“This approval also underscores the significant effort and perseverance from our clinical trial investigators and Wright’s clinical, regulatory and legal teams to bring the product to market,” Palmisano added. “This success can be attributed to everyone involved in the clinical trial, especially the study investigators and coordinators; our R&D, regulatory and clinical teams; our partners and suppliers; and most importantly, the patients who participated in the landmark prospective, randomized study comparing Augment bone graft to autograft.”
Christopher DiGiovanni, M.D., lead U.S. investigator for Augment’s American pivotal trial in North America and chief of the foot and ankle service in the department of orthopedic surgery at Massachusetts General Hospital, Harvard Medical School, said: “The FDA approval of Augment provides a valuable new therapeutic healing option as an alternative to autograft in ankle and/or hindfoot fusion procedures, which is especially important since the outcomes of these interventions can at times be complicated by delayed union or non-union. This approval is based on strong clinical data showing that Augment offers clear patient benefit by enabling a healing rate and safety profile equivalent to autogenous bone graft—while simultaneously avoiding the additional surgery required to harvest autograft bone graft tissue that can result in site-specific complications and/or prolonged harvest site pain in some patients.”
According to the company, Augment is the first clinically proven protein therapeutic to come to the U.S. orthopedics market in more than a decade, offering an alternative to autograft in ankle and/or hindfoot fusion procedures, which translates into an estimated market opportunity of approximately $300 million in the United States.
The combination of two components, recombinant human platelet-derived growth factor (rhPDGF) and beta-tricalcium phosphate (Beta-TCP), is key to the product’s activities. According to Wright officials, rhPDGF provides a biological stimulus for the recruitment and proliferation of cells, including osteoblasts, which are responsible for the formation of bone, while Beta-TCP provides a framework or scaffold for new bone growth to occur.
Based just outside Memphis, Tenn., Wright Medical Group is a specialty orthopedic company that provides extremity and biologic products.