Maria Shepherd, Medi-Vantage08.09.16
Metastatic cancer is commonly found in bone,1 and breast cancer is the most common primary tumor type in patients with bone metastases. Approximately one in eight American women will develop invasive breast cancer over the course of her lifetime.2 According to breastcancer.org, an estimated 247,000 new cases of invasive breast cancer will be diagnosed in 2016.
Why This Is Important
In general, malignant bone disease means a diagnosis of incurable cancer. Bone metastases often compromise quality of life.3 The good news? The clinical course of a breast cancer patient with metastatic bone disease can be extended. Research using registries in Denmark shows that one- and three-year cumulative survival rates in breast cancer patients diagnosed with malignant bone disease was 59 percent and 22 percent, respectively.
Liquid biopsy, a new technology based on a blood draw, is far less invasive than bone marrow aspiration. This may further extend survival rates by providing quicker, more frequent access to vital information that helps shape treatment for breast cancer patients.
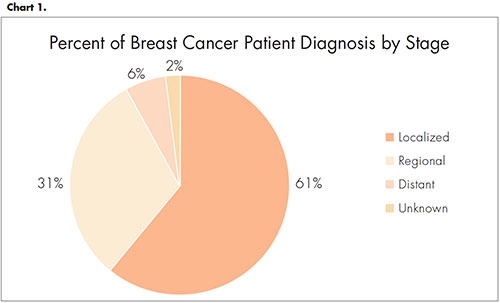
Breast cancer is the cause of 23 percent of all cancer diagnoses in women.4 Thirty-one percent and 6 percent of patients will have Stage 2 or 3 breast cancer, also called regional and distant cancer respectively (Chart 1). Patients diagnosed with Stage 3 distant cancer have metastasized cancer, so one of the treatment goals for those patients diagnosed with Stage 2 disease is to keep it from progressing to the next stage.
Liquid biopsy detects circulating nucleic acids, providing a minimally invasive, early detection method of breast cancer disease progression and re-occurrence. Liquid biopsy analyzes DNA that cancer tumors cast into the bloodstream (CTC). In a study of more than 15,000 patients with 50 different tumor types presented at the 2016 American Society of Clinical Oncology (ASCO) Annual Meeting, it was determined that liquid biopsy can accurately detect mutations in cancer DNA.5
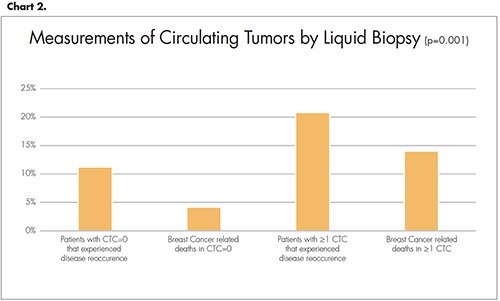
Multiple studies correlate with the findings presented at ASCO. In one study, CTCs at diagnosis were evaluated to learn if they were related to disease-free survival and overall survival.6 Blood was collected from patients and prognosis was assigned to two categories—favorable (CTC=0) or unfavorable (≥1 CTC). While not a perfect diagnostic tool, the outcomes for the Franken liquid biopsy study are seen in Chart 2.
In another study, ctDNA (circulating tumor DNA) was studied for its efficacy in identifying the risk of breast cancer survivor relapse.7 The ctDNA of 55 breast cancer patients was evaluated. Eighty percent of patients who experienced cancer reoccurrence had ctDNA detected in one or more samples. Early detection is critical to outcomes, and relapse predictions based on ctDNA occurred at an average of eight months before detection by imaging modalities.
Leveraging Liquid Biopsy
The use of liquid biopsy in patients with bone metastases is an exciting new diagnostic tool to enable early changes in treatment strategy. Can your medtech company commercialize this new tool for the disease state your company addresses?
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services and assesses prospective acquisitions. Shepherd has taught marketing and product development courses, and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 x102 or at mshepherd@medi-vantage. Visit her company’s website at www.medi-vantage.com.
Why This Is Important
In general, malignant bone disease means a diagnosis of incurable cancer. Bone metastases often compromise quality of life.3 The good news? The clinical course of a breast cancer patient with metastatic bone disease can be extended. Research using registries in Denmark shows that one- and three-year cumulative survival rates in breast cancer patients diagnosed with malignant bone disease was 59 percent and 22 percent, respectively.
Liquid biopsy, a new technology based on a blood draw, is far less invasive than bone marrow aspiration. This may further extend survival rates by providing quicker, more frequent access to vital information that helps shape treatment for breast cancer patients.
Breast cancer is the cause of 23 percent of all cancer diagnoses in women.4 Thirty-one percent and 6 percent of patients will have Stage 2 or 3 breast cancer, also called regional and distant cancer respectively (Chart 1). Patients diagnosed with Stage 3 distant cancer have metastasized cancer, so one of the treatment goals for those patients diagnosed with Stage 2 disease is to keep it from progressing to the next stage.


Liquid biopsy detects circulating nucleic acids, providing a minimally invasive, early detection method of breast cancer disease progression and re-occurrence. Liquid biopsy analyzes DNA that cancer tumors cast into the bloodstream (CTC). In a study of more than 15,000 patients with 50 different tumor types presented at the 2016 American Society of Clinical Oncology (ASCO) Annual Meeting, it was determined that liquid biopsy can accurately detect mutations in cancer DNA.5
Multiple studies correlate with the findings presented at ASCO. In one study, CTCs at diagnosis were evaluated to learn if they were related to disease-free survival and overall survival.6 Blood was collected from patients and prognosis was assigned to two categories—favorable (CTC=0) or unfavorable (≥1 CTC). While not a perfect diagnostic tool, the outcomes for the Franken liquid biopsy study are seen in Chart 2.
In another study, ctDNA (circulating tumor DNA) was studied for its efficacy in identifying the risk of breast cancer survivor relapse.7 The ctDNA of 55 breast cancer patients was evaluated. Eighty percent of patients who experienced cancer reoccurrence had ctDNA detected in one or more samples. Early detection is critical to outcomes, and relapse predictions based on ctDNA occurred at an average of eight months before detection by imaging modalities.
Leveraging Liquid Biopsy
The use of liquid biopsy in patients with bone metastases is an exciting new diagnostic tool to enable early changes in treatment strategy. Can your medtech company commercialize this new tool for the disease state your company addresses?
References
- Roodman GD. Mechanisms of bone metastasis. N Engl J Med. 2004;350(16):1655–1664.
- http://bit.ly/2016odt0801
- Lipton A. Management of bone metastases in breast cancer. Curr Treat Options Oncol. 2005;6(2):161–171.
- Abraham, J., J.L. Gulley, and C.J. Allegra, Bethesda handbook of clinical oncology. Fourth edition. ed. 2014, Philadelphia: Wolters Kluwer/ Lippincott Williams & Wilkins Health. xiv, 742 pages.
- Mack, P.; Schilsky, R.; Brody, J.; Pal, S.; June 4, 2016, presentation, American Society of Clinical Oncology annual meeting, Chicago, IL and http://bit.ly/2016odt0802.
- Franken, B., et al., Circulating tumor cells, disease recurrence and survival in newly diagnosed breast cancer. Breast Cancer Res, 2012. 14(5): p. R133.
- Garcia-Murillas, I., et al., Mutation tracking in circulating tumor DNA predicts relapse in early breast cancer. Sci Transl Med, 2015. 7(302): p. 302ra133.
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy and innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services and assesses prospective acquisitions. Shepherd has taught marketing and product development courses, and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100 x102 or at mshepherd@medi-vantage. Visit her company’s website at www.medi-vantage.com.














