Sean Fenske, Michael Barbella, and Sam Brusco08.15.17
Sam Brusco • Associate Editor
Much like in the clinical world, investment in a specialty is where the big bucks can be made in medtech. The problem with orthopedics is that it’s already somewhat of a niche market—albeit one poised to reach $43.1 billion by 2024, according to a Grand View Research report. Well-established companies like DePuy Synthes, Stryker Orthopaedics, and Zimmer Biomet already dominate the large joint and spine markets. Unless a fledgling orthopedic company can cater to the gaps in these firms’ portfolios or vastly upgrade a legacy product, it must find an auspicious market alcove and moor itself there.
The companies chosen for this article have done just that. Each has selected a massive orthopedic growth market—in these cases, 3D printing and extremities—and anchored there.
An Additive Approach
3D printing has only begun to live up to its own hype and deliver commercially available products. Frisco, Texas-based 4WEB Medical, however, has capitalized on the hype and built its entire business on 3D-printed products.
Founded in 2008 and armed with 30 years of topological dimension theory research, the company’s eponymous “4WEB” truss design is simultaneously high-strength and lightweight, which is ideal for load-bearing orthopedic implants. However, it’s not just implant strength and architecture that 3D printing optimizes. It’s also quite adept at forming the porous surface texture optimal for osseointegration, guiding bone formation with structural mechanics rather than an externally applied coating and reducing the chance of migration.
The company’s initial focus was the spinal fusion market, for which 4WEB has four U.S. Food and Drug Administration (FDA)-cleared implant systems: The Posterior, ALIF, Cervical, and Lateral Spine Truss Systems. Each features a load distribution that minimizes stress risers and subsidence (caving in or sinking)-related complications. The implants’ open architectures allow for up to 75 percent of the implant to be filled with graft material to further maximize bone incorporation. The truss design limits imaging artifact, and bi-convex surfaces bring the implant closer to adjacent bone. According to the company, the Lateral spine implant (cleared by the FDA in late July) has three times the textured surface topography to other spine implants.

4WEB’s 3D Printed Lateral Spine Truss System was first used in surgery in the U.S. in July. Image courtesy of 4WEB Medical.
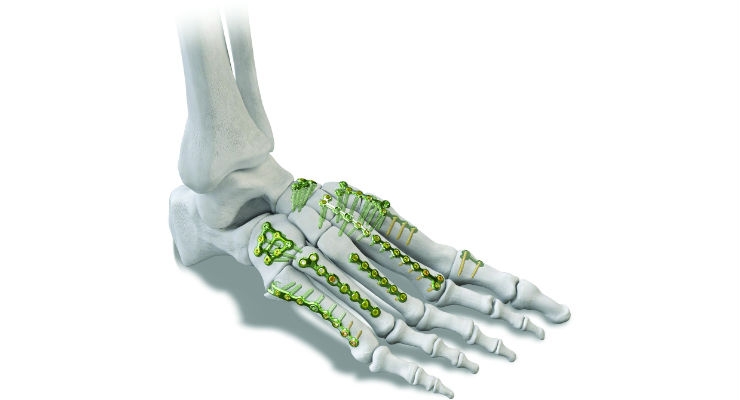
However, 4WEB soon realized its technology could be expanded beyond spinal applications. Similar in design to the spinal implant portfolio, The Osteotomy Truss System for foot and ankle fixation comes in 74 different sizing options in one set to accommodate the various tiny bones of the feet. Further, in some cases the design of the WEB Cotton, Evans, and Utility foot and ankle cages may replace the necessity for allograft wedges altogether.
4WEB intends to tailor its truss implant design for large joints in the near future. The company is actively developing implant designs for knee, hip, trauma, and patient specific orthopedic procedures.
Gaining a Foothold
Orthopedics is difficult for startups to break into. Most of the market is occupied by multi-billion dollar companies with intimidatingly large product mixes. Any orthopedic startup seeking success essentially has to find an unsaturated niche and let innovative products fly into the market before another company stakes its claim.
Englewood, Colo.-based Paragon 28 took the literal approach to getting its foot in the door of the orthopedics market. Founded in 2010, the company established itself as an exclusively foot and ankle company (seriously, “exclusively foot & ankle” is right there in the company’s logo), with the “28” in its name representing the number of bones in the foot. According to the company’s website, to date Paragon 28 has filed 62 patents and supported 32,970 surgeries.
You name a foot or ankle injury or deformity, and chances are Paragon 28 has a corresponding fixation product in its arsenal. The company offers technologies for arthrodesis, deformity correction, flatfoot correction, fractures, hallux valgus (more commonly known as a bunion), and hammertoe, as well as biologic solutions to support these procedures. The company’s steady stream of products has been giving it quite a foothold in the extremities market—in 2017 alone, Paragon 28 launched nine new foot and ankle products.
“As we continue to add more products, we maintain a fine level of detail to every feature of every product we design,” Paragon 28’s mission states. “The goal is to give options to the foot and ankle surgeon, knowing there is more than one way to achieve a great result. We have listened to surgeons and will continue to do so in order to understand and address their specific needs.”
For example, the Baby Gorilla Mini Plating system launched in January builds upon the modularity, optionality, and specificity of the company’s flagship Gorilla Plating system with a comprehensive set of implants for procedures requiring narrower plates or screws matching the size and anatomy of the small bones within the foot and ankle. The Titan 3D Wedge system launched in April consists of porous titanium wedges that can offer surgeons an alternative to autograft and allograft bone. And the Phantom Lapidus Intramedullary Nail released in June is the first dedicated nail of its kind to be used specifically for bunion correction of the first TMT (tarsometatarsal) joint.
Michael Barbella • Managing Editor
There’s a scene in the 1986 film “Star Trek IV: The Voyage Home” where Dr. Leonard McCoy sneaks into a San Francisco hospital to rescue the seriously injured Pavel Chekov. With Admiral James T. Kirk at his side, he barges into the operating room and stops the surgeon from drilling into the Starfleet officer’s skull to relieve arterial pressure.
“My God man, drilling holes in his head’s not the answer!” McCoy shouts, appalled at the barbaric elements of late 20th-century healthcare. After a brief exchange, he reverses Chekov’s traumatic brain injury within seconds using a pocket-sized medical device, angrily muttering, “Chemotherapy...fundoscopic examinations...[we’re] dealing with Medievalism here.”
Indeed, scalpels, bone drills, cranial shunts, and intracellular poisons are Gothic fixes compared to the curative powers of 23rd-century intergalactic medicine. They also seem a bit primitive today—a mere three decades removed from Chekov’s miraculous resurrection—as technology taps into the body’s innate ability to heal itself.
dCELL-erating Healing Time
The science behind Tissue Regenix Group plc’s products sounds like it belongs aboard the Starship Enterprise: The company’s dCELL technology enables the body to regenerate diseased or damaged tissue from either human (allograft) or animal (xenograft) tissue sources without compromising tissue strength and structure.
Developed at The University of Leeds (West Yorkshire, England), Tissue Regenix’s patented dCELL platform strips DNA and other cellular material from the tissue, leaving behind an intact acellular matrix that serves as a framework for repopulation by the patient’s own cells. “With dCELL technology, the body recognizes the tissue matrix as a collagen scaffold, it doesn’t matter that it might come from a pig,” explained Drew Distin, vice president of Orthopedics, North America. “The patient’s own cells will grow into it.”
Tissue Regenix’s dCELL technology can be used in a wide range of medical applications, though it only has secured regulatory approval thus far for its chronic/acute wound care product. Produced from donated cadaveric human tissue and more than 99 percent DNA-free, DermaPure closely mimics the structure and function of the native tissue it is replacing. The product signals a bio-normal response by reducing inflammation and prompting the body to progress through the natural healing phases, repairing wounds in as little as one use.
dCELL technology’s next promising application lies in ACL repair, a procedure performed nearly 1 million times annually in the United States and Europe. Anterior cruciate ligament tears or ruptures are the second most common type of knee injury and can lead to long-term damage as well as additional surgeries if not reconstructed. The firm’s OrthoPure XM (porcine meniscus), OrthoPure XT (porcine tendon), OrthoPure HM (allograft meniscus), and OrthoPure HT (allograft tendon) products could restore knee stability and allow patients to return to their pre-injury lifestyles.
“Autograft harvesting has always been the gold standard, because you’re using the patient’s own tendon and cells. But what happens when a patient has a multi-ligament injury?” Distin asked. “In those cases, an autograft would be a poor choice. Our dCELL technology could help to solve those challenges.”
The United Kingdom-based company currently is conducting clinical trials for OrthoPure products within the European Union, in hopes of debuting the innovation in the United States next year, initially with OrthoPure HT.
‘Play-doh’ for Bones
McCoy would likely scoff at OsteoNovus’ approach to bone repair, but the technology is impressing both regulators and corporate executives alike here in the early 21st century. The company’s primary product, NovoGro, is a regenerative biomaterial that grows robust bone with excellent handling properties in just six weeks.
Spawned by decade-old research into new bone graft materials, OsteoNovus was formed in 2013 by Sarit Bhaduri, director of the University of Toledo’s (UT) Multifunctional Materials Laboratory; Vijay Goel, UT McMaster-Gardner Endowed Chair of Orthopedic Bioengineering; and Dr. Anand Agarwal, a UT research professor, orthopedic surgeon, and CEO/co-founder of Spinal Balance Inc., an early-stage developer of spinal implants. The trio’s synthetic bone graft substitutes are intended to fill gaps or voids in the skeletal system during orthopedic surgery. Once in place, the implant dissolves and is eventually replaced by the patient’s own new bone.
“The basic technology is based on calcium phosphate, which has a unique manufacturing technology that can be used in spinal fusions and traumatic injuries with bone defects,” Agarwal said in a TechOhio video. “It can be used for delivering drugs, it can be used for delivering hormones...it can be used for whatever you like because it has some unique characteristics.”
More like futuristic: The company claims its NovoGro product feels like Play-doh, sticks to wet bone, and is resistant to lavage (body cavity washes). One of the most significant advantages of the UT-hatched material is its ability to dissolve and leave only natural bone behind. “Up until now, there has not been a very effective synthetic bone graft,” Agarwal told the Toledo Blade in April. “There is still innovation going on. We are a step further in making a better synthetic bone graft than what there is in the market.”
Regulators wholeheartedly agree. In late April (around the time Agarwal spoke to the Blade), OsteoNovus received U.S. Food and Drug Administration approval for NovoGro—a mere eight weeks after filing its application with the agency.
OsteoNovus has received funding for NovoGro through the Ohio Third Frontier project and Third Frontier partner organizations, as well as private investors. The company also is slated to close a partnership with a mid-size firm (more than $100 million in revenue) for non-exclusive private label NovoGro sales.
For example, the Baby Gorilla Mini Plating system launched in January builds upon the modularity, optionality, and specificity of the company’s flagship Gorilla Plating system with a comprehensive set of implants for procedures requiring narrower plates or screws matching the size and anatomy of the small bones within the foot and ankle. The Titan 3D Wedge system launched in April consists of porous titanium wedges that can offer surgeons an alternative to autograft and allograft bone. And the Phantom Lapidus Intramedullary Nail released in June is the first dedicated nail of its kind to be used specifically for bunion correction of the first TMT (tarsometatarsal) joint.
Sean Fenske • Editor
While providing dissimilar technologies unrelated to the other’s unique clinical application, these two companies have introduced new product offerings that could make a significant impact in their respective sectors. One enhances the patient rehabilitation experience while the other improves the orthopedic implant surface. Whether or not these innovations help carry the companies to a spot on a future ODT Top 10 list remains to be seen, but in the meantime, they should impact the orthopedic industry and benefit the patients who use them.
Digital Rehabilitation
Providing an array of braces, supports, and other products focused on rehabilitation, Breg’s interaction with patients occurs primarily post-orthopedic surgical procedure. The company claims it delivers solutions to over 1 million patients every year. The bracing technologies address virtually every major joint, such as knee, hip, elbow, and shoulder. In addition, the company offers therapy products to aid with pain management and help prevent deep vein thrombosis. None of these technologies, however, are the reason for the company’s inclusion in this feature.
The Breg Flex is a digital health product the company released in 2017 to address the rehabilitation needs for patients performing exercises at home following an orthopedic surgical procedure. The patient wears a wireless sensor that syncs to an app hosted on a mobile device, such as a tablet. Through the sensor, the app is able to track the progress being made by the patient as they cycle through their rehabilitation exercise routine. The app also provides a visual guide for the patient to ensure they are performing an exercise properly. The patient’s progress can also be shared with healthcare professionals.

Breg Flex is a first-of-its-kind mobile health solution designed to facilitate and improve patients’ at-home recovery following orthopedic surgery. Image courtesy of Breg.
“Our beta program revealed that Breg Flex has the potential to improve overall patient outcomes. It found that Breg Flex patients achieved their optimal outcome using 25 percent less visits than non-Breg Flex patients in the same diagnosis group,” said Dr. Daniel Segina, an orthopedic trauma surgeon based in Rockledge, Fla. “Breg Flex patients scored 20 percent higher on their functional improvement scores than non-Breg Flex patients, on average. Additionally, Breg Flex patients were 75 percent compliant with their therapy protocols (vs. an average of 30 percent for non-Breg Flex patients), and reported a satisfaction level of 90 percent, on average.”
As more orthopedic reimbursement systems move to a bundled payment model, enhancing patient recovery by ensuring compliance with required physical therapy regimens following a surgical procedure will help prevent additional costs that fall to the healthcare provider. As such, more facilities may look to solutions like the Breg Flex to ensure patients maintain their exercise routine, requiring less follow-up and aiding in the overall success of the treatment.
Further, it’s possible that Breg could expand the indications for which the Flex technology is used. While currently specific to the orthopedic rehabilitation space, it wouldn’t require much revision for the system to be adapted for a physical fitness type of application, broadening the appeal to a very large, although already crowded, consumer market.
Spinal Fusion Nanotechnology
Founded in 2006, Titan Spine is a relatively newer player in the orthopedic technology space. It provides a variety of products used for fusion procedures, primarily interbody devices that are implanted between vertebra to address a degenerative disc condition. Bucking an industry trend toward PEEK (polyether ether ketone) materials for device fabrication, the company has produced implants using the more traditional titanium, but with a twist. That significant variation is why the company finds itself included on this list.
Titan Spine offers a line of its Endoskeleton titanium implants featuring its proprietary nanoLOCK surface technology. The surface is generated through a subtractive process that results in an osteogenic environment, promoting bone growth. The technology received FDA clearance in late 2014, but the full product launch did not occur until the fourth quarter of 2016.
“The full U.S. launch of our nanoLOCK surface technology comes at an optimal time as the interbody market is rapidly shifting toward titanium surface-enhanced implants. It represents a major milestone for Titan Spine and is the culmination of years of preparation through scientific study, manufacturing validation, IP protection, and unique regulatory clearances through collaboration with the FDA and CMS. This effort resulted in nanoLOCK being the only nano-cleared interbody device on the market and is the only interbody device that has access to the recently created new technology ICD-10 code for a nanotextured surface on an interbody fusion device,” Kevin Gemas, president of Titan Spine, explained at the time of the announcement of the full launch of the product.
While the company’s own offerings represent an impressive product portfolio for indications in the spinal fusion space, Titan Spine could make substantial waves in the sector should the OEM decide to license the technology that generates the nanoLOCK surface treatment capability to other medical device OEMs. Its own product line is most certainly likely to grow in adoption, but well-established OEMs in the space who already have an expansive customer base could truly bring the technology into the limelight.