Sam Brusco, Associate Editor10.02.17
It’s usually not a promising prognosis for patients once the connection between the brain and muscles becomes severed or disrupted in some way. Whether through spinal cord injury, a stroke, or a gait disability, mobility suffers once the brain and muscles lose communication. The fallout can range from loss of fine motor skills or paraplegia, to complete paralysis. Complete or even partial recovery can be rare in these instances, hindering quality of life for potentially the remainder of patients’ lives.
However, as of late, brain-computer interface (BCI) technology has risen to the occasion to pick up the slack of a damaged neuromuscular system. For those unfamiliar with a BCI system, a computer captures and “learns” patterns of electroencephalographic (EEG) activity via either a head-worn or implantable sensor. A software algorithm then translates those patterns of EEG brain activity into actionable commands—for example, moving a certain part of the body. From there, the commands could be transmitted to a powered assistive device like a robotic exoskeleton to help those with neuromuscular damage regain fine motor control, or grant those with some form of paralysis the ability to move a previously immobile body part.
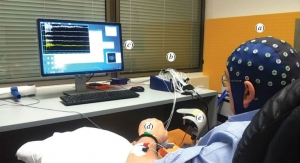
In late August, University of Adelaide researchers showed that training with a BCI system can boost stroke patients’ motor function in a damaged hand. This particular system used a suite of sensors worn as a cap to detect EEG activity on the scalp surface and translated the signals into sensory feedback via a robotic manipulator.
“In the majority of strokes, the area of the brain that sends motor commands to the muscles becomes partly damaged and thereby degrades motor functions of the affected parts,” said Dr. Sam Darvishi, who completed the BCI work during his Ph.D. in the University of Adelaide’s School of Electrical and Electronic Engineering. “During the early phases of motor learning (such as when we are toddlers), our brain and body learn how to work in harmony when the brain commands the target muscles and then receives feedback via seeing and feeling each body movement. After a stroke, the brain needs to re-train the lost skills.”
BCIs have been suggested as an alternative therapy for stroke patients for some time. Thus far, they have shown some promise in regaining motor control, but to date, haven’t been particularly effective. Finding a solution to this issue was largely the subject of Dr. Darvishi’s work.
“Our theory is that to achieve clinical results with BCIs, we need to have the right feedback to the brain at the right time,” he explained. “We need to provide the same feedback that we receive during natural motor learning, when we are seeing and feeling the body’s movement. We also found there should be a short delay between the brain activation and the activation of target muscles.”
The BCI designed by the University of Adelaide researchers proved to be remarkably effective in one patient’s case study. A 36 percent improvement in hand function was achieved with only ten 30-minute sessions. Though it was only a single patient and as such can’t be generalized to the whole stroke population, Dr. Darvishi was hopeful about the study’s implications.
“It certainly shows enough promise for a larger study of stroke patients to see if this could be a feasible therapy for stroke rehabilitation,” he said. “This would be a major step toward helping stroke patients recover from debilitating damage.”
Paraplegics may also be granted the chance to walk again using a BCI thanks to a five-year, $8 million Cyber-Physical Systems Frontier grant from the National Science Foundation. This grant intends to fund development of a fully implantable BCI to restore lower extremity sensation and the ability to walk. This BCI will transmit commands to a robotic exoskeleton for walking, which will then transmit sensory information back to the brain.
“The restoration of walking is a very significant goal for patients after spinal cord injury,” said Charles Liu, M.D., Ph.D., principal investigator at USC’s Keck School of Medicine (one of the institutions receiving the NSF funds), and professor of clinical neurological surgery and neurology and director of the USC Neurorestoration Center. “New solutions are possible with the recent advances in neuroprosthetics and regenerative medicine. We’re at the point where we can create solutions similar in concept to Tony Stark’s Iron Man suit, which is neurally integrated with him. Tony Stark’s brain interacts with the suit, and the suit interacts with his brain. Everything the suit feels, his brain feels. That’s the idea.”
The project will be carried out in phases, drawing upon the combined engineering, neuroscience, and computer science expertise of all the collaborating researchers. First, signals from the brain telling the legs to walk will be decoded. Then epilepsy patients who had electrodes implanted in the brain by Liu (as part of their workup for surgical epilepsy treatment) will have their signals recorded while walking. Finally, these signals will be translated to control a wearable robotic exoskeleton.
“With this grant, Dr. Liu and his counterparts at University of California, Irvine and Caltech are poised to push the frontiers of medicine and engineering into unchartered territory,” said Rohit Varma, M.D., MPH, dean of the Keck School. “It is this spirit of innovation that drives our clinician-scientists to find novel ways to prevent, treat, or cure the most challenging health issues.”
“We want to create a paradigm shift for what’s possible for patients who are paralyzed by finding engineering solutions to medical problems,” Liu said.
However, as of late, brain-computer interface (BCI) technology has risen to the occasion to pick up the slack of a damaged neuromuscular system. For those unfamiliar with a BCI system, a computer captures and “learns” patterns of electroencephalographic (EEG) activity via either a head-worn or implantable sensor. A software algorithm then translates those patterns of EEG brain activity into actionable commands—for example, moving a certain part of the body. From there, the commands could be transmitted to a powered assistive device like a robotic exoskeleton to help those with neuromuscular damage regain fine motor control, or grant those with some form of paralysis the ability to move a previously immobile body part.
In late August, University of Adelaide researchers showed that training with a BCI system can boost stroke patients’ motor function in a damaged hand. This particular system used a suite of sensors worn as a cap to detect EEG activity on the scalp surface and translated the signals into sensory feedback via a robotic manipulator.
“In the majority of strokes, the area of the brain that sends motor commands to the muscles becomes partly damaged and thereby degrades motor functions of the affected parts,” said Dr. Sam Darvishi, who completed the BCI work during his Ph.D. in the University of Adelaide’s School of Electrical and Electronic Engineering. “During the early phases of motor learning (such as when we are toddlers), our brain and body learn how to work in harmony when the brain commands the target muscles and then receives feedback via seeing and feeling each body movement. After a stroke, the brain needs to re-train the lost skills.”
BCIs have been suggested as an alternative therapy for stroke patients for some time. Thus far, they have shown some promise in regaining motor control, but to date, haven’t been particularly effective. Finding a solution to this issue was largely the subject of Dr. Darvishi’s work.
“Our theory is that to achieve clinical results with BCIs, we need to have the right feedback to the brain at the right time,” he explained. “We need to provide the same feedback that we receive during natural motor learning, when we are seeing and feeling the body’s movement. We also found there should be a short delay between the brain activation and the activation of target muscles.”
The BCI designed by the University of Adelaide researchers proved to be remarkably effective in one patient’s case study. A 36 percent improvement in hand function was achieved with only ten 30-minute sessions. Though it was only a single patient and as such can’t be generalized to the whole stroke population, Dr. Darvishi was hopeful about the study’s implications.
“It certainly shows enough promise for a larger study of stroke patients to see if this could be a feasible therapy for stroke rehabilitation,” he said. “This would be a major step toward helping stroke patients recover from debilitating damage.”
Paraplegics may also be granted the chance to walk again using a BCI thanks to a five-year, $8 million Cyber-Physical Systems Frontier grant from the National Science Foundation. This grant intends to fund development of a fully implantable BCI to restore lower extremity sensation and the ability to walk. This BCI will transmit commands to a robotic exoskeleton for walking, which will then transmit sensory information back to the brain.
“The restoration of walking is a very significant goal for patients after spinal cord injury,” said Charles Liu, M.D., Ph.D., principal investigator at USC’s Keck School of Medicine (one of the institutions receiving the NSF funds), and professor of clinical neurological surgery and neurology and director of the USC Neurorestoration Center. “New solutions are possible with the recent advances in neuroprosthetics and regenerative medicine. We’re at the point where we can create solutions similar in concept to Tony Stark’s Iron Man suit, which is neurally integrated with him. Tony Stark’s brain interacts with the suit, and the suit interacts with his brain. Everything the suit feels, his brain feels. That’s the idea.”
The project will be carried out in phases, drawing upon the combined engineering, neuroscience, and computer science expertise of all the collaborating researchers. First, signals from the brain telling the legs to walk will be decoded. Then epilepsy patients who had electrodes implanted in the brain by Liu (as part of their workup for surgical epilepsy treatment) will have their signals recorded while walking. Finally, these signals will be translated to control a wearable robotic exoskeleton.
“With this grant, Dr. Liu and his counterparts at University of California, Irvine and Caltech are poised to push the frontiers of medicine and engineering into unchartered territory,” said Rohit Varma, M.D., MPH, dean of the Keck School. “It is this spirit of innovation that drives our clinician-scientists to find novel ways to prevent, treat, or cure the most challenging health issues.”
“We want to create a paradigm shift for what’s possible for patients who are paralyzed by finding engineering solutions to medical problems,” Liu said.