Maria Shepherd, President and Founder, Medi-Vantage11.21.17
The global obesity epidemic continues to grow. In 2016, it was estimated that greater than 39 percent of adults1 (up from 35.7 percent of adults2) are obese (Table 1). Obesity can bring a quadruple whammy of co-morbidities including cardiovascular disease (the leading cause of death in 2012); diabetes; musculoskeletal disorders (especially osteoarthritis), and cancer (including breast, colon, endometrial, gallbladder, kidney, liver, ovarian, and prostate cancer).1 CDC statistics3 estimate the medical costs associated with obesity in 2008 was $147 billion.
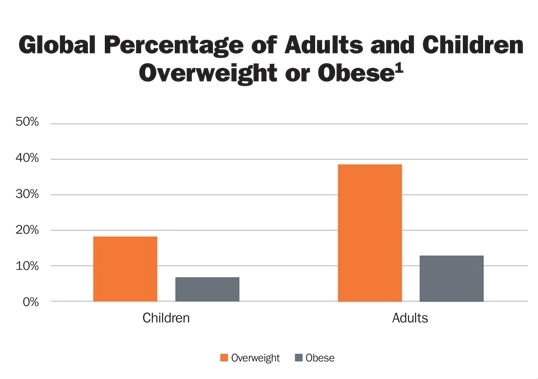
Table 1
Why This Is Important
Obesity drives the rate of musculoskeletal disorders, particularly hip and knee implants. In a large study designed to evaluate the effect of body mass index (BMI) in outcomes in primary total knee arthroplasty (n=2,180), researchers found that 93 percent of patients were either overweight or obese.4 The authors also stated that even though there was no consensus on this finding, there was growing evidence that increasing BMI, especially in patients with a BMI greater than 40, results in inferior clinical outcomes.
The mean BMI in the study was 31.86 (range: 18-52). Overweight and obese patients had more difficult recoveries, as measured by total WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) scores. Researchers found no statistically significant difference in the pre- or post-op WOMAC scores of the normal weight cohort (BMI<25) as compared to patients with a BMI ≥25. At six-months post-op, patients were assessed in the cohorts for BMI 25-29 (overweight), BMI 30-34, and BMI 35-39. Significant differences in outcomes were found.
Thirty-two patients in the obese or overweight cohorts (1.47 percent) had superficial infections, nine (0.41 percent) were diagnosed with deep infections, and 19 (0.87 percent) revisions were performed. Later follow-up added 10 cases of deep vein thrombosis (0.46 percent) and nine pulmonary embolisms (0.41 percent) (Table 2).
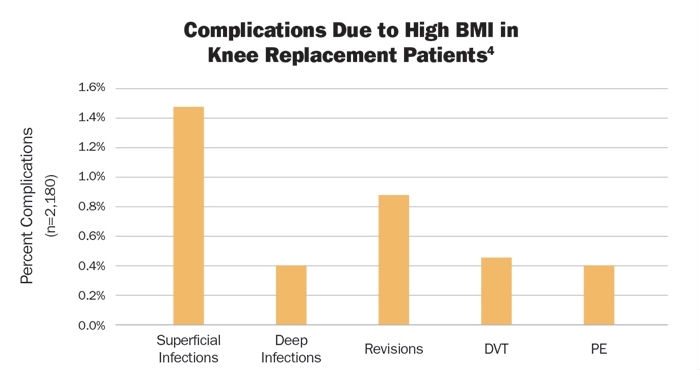
Table 2
Approximately 700,000 knee replacement procedures are performed annually in the United States.5 This number is projected to increase to 3.48 million procedures per year by 2030 (Table 3).6 Between 2000 and 2010, the number of hip replacement procedures among inpatients aged 45 and over had more than doubled—from 138,700 to 310,800 procedures per year.7 By 2030, these rates are projected to increase to 572,000.6
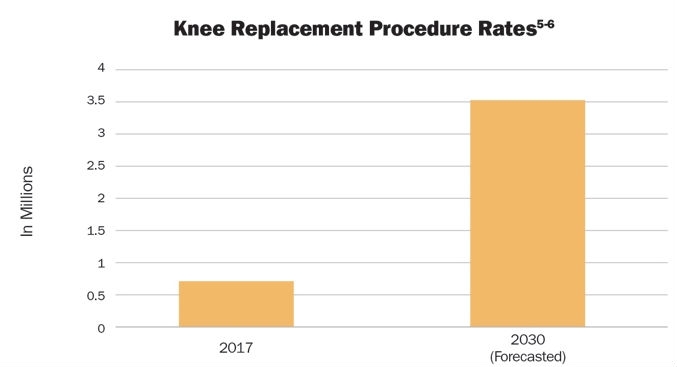
Table 3
From the Medi-Vantage Point
The issue of obesity is not going away anytime soon. So why not consider the strategies discussed by diabetes device industry leaders and develop implants that can reduce the complications associated with obese patients?
For example, in a recent article,8 Jeffrey Brewer, president and CEO of Bigfoot Biomedical—a firm dedicated to bettering the lives of people with diabetes through smart technology—stated, “We treat all patients of a given chronic disease category as if they’re the same. They should use the same tool, the same drugs, be treated by the same kind of doctor, and use the same therapy, and the world just doesn’t work like that.”
“We’re all different,” he added. “Different things are important to different people, and that includes chronic disease. There are some people that are willing to give a lot to it, to optimize their health. And then there are people who just simply refuse to do that. They are going to prioritize other things in their lives, and I think we should have solutions that meet the needs of both kinds of patients.”
Conclusion and Recommendations
Sure, it takes a long time to get a new implant on the market, designed for the specific needs of the obese population. But in a world of CJR (comprehensive care for joint replacement) and commoditized hip and knee implants, doesn’t that objective make sense?
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.

Table 1
Why This Is Important
Obesity drives the rate of musculoskeletal disorders, particularly hip and knee implants. In a large study designed to evaluate the effect of body mass index (BMI) in outcomes in primary total knee arthroplasty (n=2,180), researchers found that 93 percent of patients were either overweight or obese.4 The authors also stated that even though there was no consensus on this finding, there was growing evidence that increasing BMI, especially in patients with a BMI greater than 40, results in inferior clinical outcomes.
The mean BMI in the study was 31.86 (range: 18-52). Overweight and obese patients had more difficult recoveries, as measured by total WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) scores. Researchers found no statistically significant difference in the pre- or post-op WOMAC scores of the normal weight cohort (BMI<25) as compared to patients with a BMI ≥25. At six-months post-op, patients were assessed in the cohorts for BMI 25-29 (overweight), BMI 30-34, and BMI 35-39. Significant differences in outcomes were found.
Thirty-two patients in the obese or overweight cohorts (1.47 percent) had superficial infections, nine (0.41 percent) were diagnosed with deep infections, and 19 (0.87 percent) revisions were performed. Later follow-up added 10 cases of deep vein thrombosis (0.46 percent) and nine pulmonary embolisms (0.41 percent) (Table 2).

Table 2
Approximately 700,000 knee replacement procedures are performed annually in the United States.5 This number is projected to increase to 3.48 million procedures per year by 2030 (Table 3).6 Between 2000 and 2010, the number of hip replacement procedures among inpatients aged 45 and over had more than doubled—from 138,700 to 310,800 procedures per year.7 By 2030, these rates are projected to increase to 572,000.6

Table 3
From the Medi-Vantage Point
The issue of obesity is not going away anytime soon. So why not consider the strategies discussed by diabetes device industry leaders and develop implants that can reduce the complications associated with obese patients?
For example, in a recent article,8 Jeffrey Brewer, president and CEO of Bigfoot Biomedical—a firm dedicated to bettering the lives of people with diabetes through smart technology—stated, “We treat all patients of a given chronic disease category as if they’re the same. They should use the same tool, the same drugs, be treated by the same kind of doctor, and use the same therapy, and the world just doesn’t work like that.”
“We’re all different,” he added. “Different things are important to different people, and that includes chronic disease. There are some people that are willing to give a lot to it, to optimize their health. And then there are people who just simply refuse to do that. They are going to prioritize other things in their lives, and I think we should have solutions that meet the needs of both kinds of patients.”
Conclusion and Recommendations
Sure, it takes a long time to get a new implant on the market, designed for the specific needs of the obese population. But in a world of CJR (comprehensive care for joint replacement) and commoditized hip and knee implants, doesn’t that objective make sense?
References
- http://bit.ly/odt171201
- Ogden CL, Carroll MD, Kit B, Flegal KM. NCHS data brief, prevalence of obesity in the United States, 2009–2010.
- http://bit.ly/odt171202
- http://bit.ly/odt171203
- http://bit.ly/odt171204
- Kurtz S, Ong K, Lau E, et al. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am 2007; 89:780.
- http://bit.ly/odt171205
- http://bit.ly/odt171206
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.