Maria Shepherd, President and Founder, Medi-Vantage02.12.18
The neurostimulation market is big and only getting bigger. Its current main application is to treat chronic pain. The number of patients afflicted with chronic pain is enormous and matches the size of the diabetes market in global size and scope. Despite advances by medical device companies, the chronic pain market is still underserved and has significant room for growth. Other applications for neurostimulation cross a broad spectrum of disease states, and there are many different configurations of neurostim devices treating chronic pain. They include, but are not limited to devices designed for spinal cord (SCS), peripheral nerve (PNS), deep brain (DBS), and sacral nerve (SNS) stimulation, among others.
Why This Is Important
Chronic pain is an immense global problem. Studies estimate that 1.5 billion adults1 suffer from chronic pain worldwide. In 2011, it was estimated2 that 100 million U.S. adults experienced chronic pain, defined as having pain every day for the previous three months (Table 1).

Table 1
It’s a Crowded Market
The top three device types in terms of market size and growth over the next five years are SCS, DBS, and SNS. Current indications for these devices include (but are not limited to) chronic pain (SCS), Parkinson’s disease, epilepsy and obsessive-compulsive disorder (DBS), and urinary and fecal incontinence (SNS). Many medical device companies are competing aggressively in this space to gain share in these markets. Despite barriers to entry, such as high device costs, difficulty in gaining reimbursement, and low knowledge levels among referring physicians, such as primary care physicians and neurologists, the market for these three device types is forecasted to grow from approximately $4.5 billion in 2015 to $13.7 billion in 20233 (Table 2).
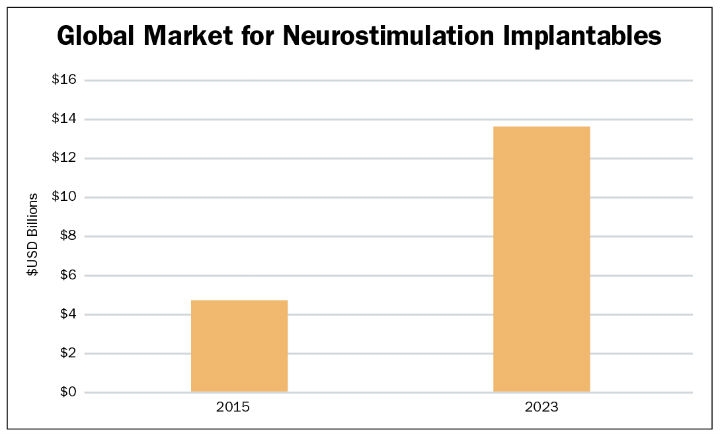
Table 2
Future Applications for Neurostimulation
It’s no wonder that it’s “bare knuckles fighting” for penetration in this market. The big three—Medtronic, St. Jude Medical (Abbott), and Boston Scientific—are skirmishing for market share4 because they know the future applications for neurostimulation include some of the largest and most challenging disease states (Table 3). These are, among others, depression, obesity, stroke recovery, Alzheimer’s, congestive heart failure, hypertension, headache, sleep apnea, rheumatoid arthritis, GERD, diabetes, and tinnitus. There is no doubt the list of disease states treatable by neurostimulation implantables will get longer.
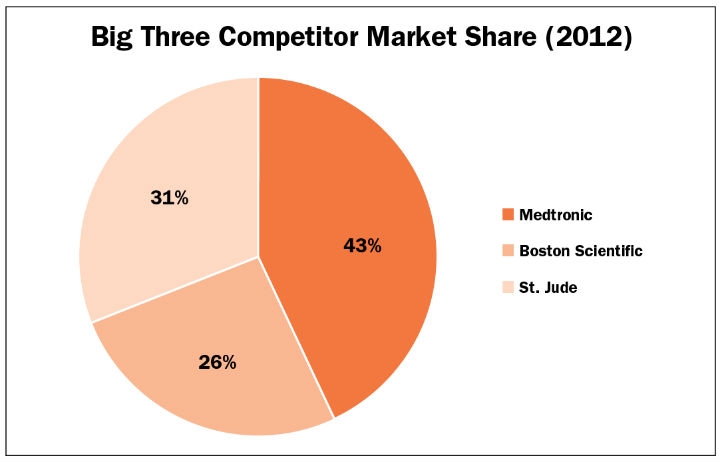
Table 3
The Medi-Vantage Perspective
There are still many unmet needs in implantable neurostimulation devices. Our research tells us that the most important requirement is to improve designs to reduce or eliminate the pain associated with the implant procedure, which currently requires waking up the patient to test the pain response. The method of action of some SCS devices is to exchange the pain sensation for paresthesia—ideally, a comfortable tingling. Patients report coping with paresthesia, but a significant segment describes the paresthesia sensation as uncomfortable, especially with positional changes. Batteries or external power supplies need improvement for certain product designs. In addition, there is a need to design for MRI compatibility and miniaturize the overall product. And, while chronic pain is managed for certain indications, there is still significant unmet need in other pain sources, especially upper limb and neck pain.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.
Why This Is Important
Chronic pain is an immense global problem. Studies estimate that 1.5 billion adults1 suffer from chronic pain worldwide. In 2011, it was estimated2 that 100 million U.S. adults experienced chronic pain, defined as having pain every day for the previous three months (Table 1).

Table 1
It’s a Crowded Market
The top three device types in terms of market size and growth over the next five years are SCS, DBS, and SNS. Current indications for these devices include (but are not limited to) chronic pain (SCS), Parkinson’s disease, epilepsy and obsessive-compulsive disorder (DBS), and urinary and fecal incontinence (SNS). Many medical device companies are competing aggressively in this space to gain share in these markets. Despite barriers to entry, such as high device costs, difficulty in gaining reimbursement, and low knowledge levels among referring physicians, such as primary care physicians and neurologists, the market for these three device types is forecasted to grow from approximately $4.5 billion in 2015 to $13.7 billion in 20233 (Table 2).

Table 2
Future Applications for Neurostimulation
It’s no wonder that it’s “bare knuckles fighting” for penetration in this market. The big three—Medtronic, St. Jude Medical (Abbott), and Boston Scientific—are skirmishing for market share4 because they know the future applications for neurostimulation include some of the largest and most challenging disease states (Table 3). These are, among others, depression, obesity, stroke recovery, Alzheimer’s, congestive heart failure, hypertension, headache, sleep apnea, rheumatoid arthritis, GERD, diabetes, and tinnitus. There is no doubt the list of disease states treatable by neurostimulation implantables will get longer.

Table 3
The Medi-Vantage Perspective
There are still many unmet needs in implantable neurostimulation devices. Our research tells us that the most important requirement is to improve designs to reduce or eliminate the pain associated with the implant procedure, which currently requires waking up the patient to test the pain response. The method of action of some SCS devices is to exchange the pain sensation for paresthesia—ideally, a comfortable tingling. Patients report coping with paresthesia, but a significant segment describes the paresthesia sensation as uncomfortable, especially with positional changes. Batteries or external power supplies need improvement for certain product designs. In addition, there is a need to design for MRI compatibility and miniaturize the overall product. And, while chronic pain is managed for certain indications, there is still significant unmet need in other pain sources, especially upper limb and neck pain.
References
- http://bit.ly/odt180101
- Institute of Medicine Report from the Committee on Advancing Pain Research, Care, and Education: Relieving Pain in America, A Blueprint for Transforming Prevention, Care, Education and Research. The National Academies Press, 2011.
- http://bit.ly/odt180103
- http://bit.ly/odt180104
Maria Shepherd has more than 20 years of leadership experience in medical device/life-science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing and business strategy as well as innovation research for the medical device industry. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.msbiv.com). She can be reached at 855-343-3100, ext. 102, or at mshepherd@medi-vantage.com. Visit her website at www.medi-vantage.com.