Sean Fenske, Editor-in-Chief03.21.18
Last week (as of this writing), I attended the 2018 Annual Meeting of the American Academy of Orthopaedic Surgeons in New Orleans. It is likely one of the largest collections of orthopedic industry stakeholders, from key surgical opinion leaders to leading device OEMs to consultants, hospitals, universities, and even some patients. It’s a rare opportunity to gain access to representatives of the entire supply and care chain within the orthopedic industry.
For me personally, it’s a chance to catch up with contacts, see new technologies, hear about success stories, and discuss challenges encountered at every level. Fortunately, this year’s event proved to be no exception. I had several fantastic conversations with device companies as well as their supply partners.
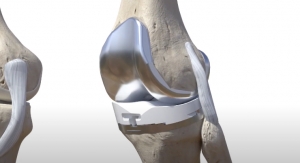
One such meeting occurred with Dr. Zhonglin (John) Zhu, chief technology officer and general manager for Okani Medical Technology, and Dane Waund, global marketing manager of healthcare for Solvay Specialty Polymers. As a provider of polyetheretherketone (PEEK) material, Solvay has had a hand in a few technologies I’ve mentioned in previous Editor’s Letters, but those were focused on spine implant solutions. Okani, however, is in the process of using the plastic to fabricate an all-polymer knee implant called ORGKnee. The results were fairly impressive too. Zhu explained that the properties of PEEK more closely resembled those of cortical bone, as compared to titanium, which could improve patient comfort. Also, since the PEEK material could be injection molded, the implants can be fabricated in a much more rapid fashion. The wear of the implant was also significantly reduced again compared to its metal counterpart. Okani sees the implant as a potentially more affordable option for those in, for example, rural areas of China, a country with a larger possible patient population (i.e., those considered in need of a knee replacement) than the U.S. The company expects to enter pre-clinical trials in April and standard clinical trials in September. It is setting up for a commercial launch in China in 2020 following the country’s Food and Drug Administration approval.
Dane and I then made our way to see James Schultz, executive vice president for sales and marketing at ECA Medical Instruments. As you may have read in previous issues of ODT, ECA provides disposable instruments to orthopedic device manufacturers, a still relatively new proposition for the industry. The company was showcasing its new Cervical-One kit, which is offered for spinal procedures. Without the need to worry about sterilization and potential infection considerations, instruments are discarded following each procedure. I did ask about the environmental impact of disposable instruments, but Jim reminded me of the information he’s shared with me of the comparison between those and the impact of cleaning reusable instruments. The argument for disposable instruments can certainly be made when including the patient safety aspects as well.
I had a brief encounter with Steve Seger, Sr. VP of sales and marketing for Orthosensor. The company provides a knee implant balancing solution that he explained resulted in a 97 percent patient satisfaction record one-year post-op. Steve joked we balance the tires on our car with more technology than we do a knee implant going into our body. He explained that most doctors balance a knee by feel and when invited to compare their “feel” to the sensor readings, some surgeons were well outside an acceptable level. If the technology enables better patient satisfaction following knee implant procedures, it’s certainly worth a closer look by both surgeons and the firms making those implants to see if it’s a complimentary technology to their system.
Another of my stops offered an educational experience, which is one of the primary reasons for many in attendance (although a visit to The Big Easy may have been reason enough for some). At the United Orthopedic Corporation (UOC) booth, Mark Froimson, M.D., MBA, 27th president of the American Association of Hip and Knee Surgeons, and a principal at Riverside Health Advisors, led a presentation on bundled payments. While the talk was targeted toward the surgeon attendees, it was good information on a topic that’s going to impact virtually all stakeholders within the orthopedic industry (and eventually much further beyond that). In a follow-up discussion with Calvin Lin, president of UOC USA, I was surprised to hear that he thinks there are still quite a few surgeons unfamiliar with the aspects of bundled payments, and offering information and education about it can only help. The company’s strategy of reducing the number of trays required for an orthopedic surgical procedure couple perfectly with the bundled payments rollout, allowing for a healthcare facility to capture more revenue for procedures.
Next year’s annual meeting takes place in Las Vegas, so if you don’t have plans to attend already, “roll the dice” and see if it’s not worth your while. Maybe I’ll even see you there!
For me personally, it’s a chance to catch up with contacts, see new technologies, hear about success stories, and discuss challenges encountered at every level. Fortunately, this year’s event proved to be no exception. I had several fantastic conversations with device companies as well as their supply partners.
One such meeting occurred with Dr. Zhonglin (John) Zhu, chief technology officer and general manager for Okani Medical Technology, and Dane Waund, global marketing manager of healthcare for Solvay Specialty Polymers. As a provider of polyetheretherketone (PEEK) material, Solvay has had a hand in a few technologies I’ve mentioned in previous Editor’s Letters, but those were focused on spine implant solutions. Okani, however, is in the process of using the plastic to fabricate an all-polymer knee implant called ORGKnee. The results were fairly impressive too. Zhu explained that the properties of PEEK more closely resembled those of cortical bone, as compared to titanium, which could improve patient comfort. Also, since the PEEK material could be injection molded, the implants can be fabricated in a much more rapid fashion. The wear of the implant was also significantly reduced again compared to its metal counterpart. Okani sees the implant as a potentially more affordable option for those in, for example, rural areas of China, a country with a larger possible patient population (i.e., those considered in need of a knee replacement) than the U.S. The company expects to enter pre-clinical trials in April and standard clinical trials in September. It is setting up for a commercial launch in China in 2020 following the country’s Food and Drug Administration approval.
Dane and I then made our way to see James Schultz, executive vice president for sales and marketing at ECA Medical Instruments. As you may have read in previous issues of ODT, ECA provides disposable instruments to orthopedic device manufacturers, a still relatively new proposition for the industry. The company was showcasing its new Cervical-One kit, which is offered for spinal procedures. Without the need to worry about sterilization and potential infection considerations, instruments are discarded following each procedure. I did ask about the environmental impact of disposable instruments, but Jim reminded me of the information he’s shared with me of the comparison between those and the impact of cleaning reusable instruments. The argument for disposable instruments can certainly be made when including the patient safety aspects as well.
I had a brief encounter with Steve Seger, Sr. VP of sales and marketing for Orthosensor. The company provides a knee implant balancing solution that he explained resulted in a 97 percent patient satisfaction record one-year post-op. Steve joked we balance the tires on our car with more technology than we do a knee implant going into our body. He explained that most doctors balance a knee by feel and when invited to compare their “feel” to the sensor readings, some surgeons were well outside an acceptable level. If the technology enables better patient satisfaction following knee implant procedures, it’s certainly worth a closer look by both surgeons and the firms making those implants to see if it’s a complimentary technology to their system.
Another of my stops offered an educational experience, which is one of the primary reasons for many in attendance (although a visit to The Big Easy may have been reason enough for some). At the United Orthopedic Corporation (UOC) booth, Mark Froimson, M.D., MBA, 27th president of the American Association of Hip and Knee Surgeons, and a principal at Riverside Health Advisors, led a presentation on bundled payments. While the talk was targeted toward the surgeon attendees, it was good information on a topic that’s going to impact virtually all stakeholders within the orthopedic industry (and eventually much further beyond that). In a follow-up discussion with Calvin Lin, president of UOC USA, I was surprised to hear that he thinks there are still quite a few surgeons unfamiliar with the aspects of bundled payments, and offering information and education about it can only help. The company’s strategy of reducing the number of trays required for an orthopedic surgical procedure couple perfectly with the bundled payments rollout, allowing for a healthcare facility to capture more revenue for procedures.
Next year’s annual meeting takes place in Las Vegas, so if you don’t have plans to attend already, “roll the dice” and see if it’s not worth your while. Maybe I’ll even see you there!