Sam Brusco, Associate Editor03.21.18
The way Americans manage their pain has been has become an agonizing issue in recent years. The longtime practice of prescribing opioids as a first-line treatment has come under much scrutiny as more and more Americans fit the criteria for opioid use disorder. The consequent “opioid crisis” has garnered enough attention to receive mention in the President’s State of the Union address.
According to Centers for Disease Control and Prevention (CDC) research, as many as one in four patients receiving long-term therapy in primary care settings may struggle with opioid use disorder. The number of opioids prescribed and sold in the United States has increased fourfold since 1999, with over 165,000 reported deaths from prescription opioid-related overdoses.
“The issue of opioid misuse and abuse remains one of my highest priorities,” FDA Commissioner Scott Gottlieb proclaimed in a statement. “We believe it’s going to take carefully developed, sustained, and coordinated action by everyone involved to reduce the tide of opioid addiction and death afflicting our communities; while maintaining appropriate prescribing for patients in medical need. We recognize both the urgency and complexity of this crisis and are taking several steps today toward these goals.”
While narcotic drugs help manage pain in the short term, there’s not enough information available about their long-term risks and benefits. The information coming to light concerning long term use for chronic pain, however, is troublesome. NIH statistics report roughly 21 to 29 percent of patients prescribed opioids for chronic pain misuse them, and between 8 and 12 percent develop an opioid use disorder.
In response to this threat, last year the CDC released its “Guideline for Prescribing Opioids for Chronic Pain,” outlining steps primary care clinicians should take in treating adult patients for chronic pain in outpatient settings. It exhorts clinicians to use nonpharmacologic therapy or non-opioid pharmacologic therapy (medications like acetaminophen or ibuprofen) as the preferred treatment method for chronic pain, to establish goals for pain and function, and to use immediate-release opioids when beginning treatment, among several other guidelines.
Remediating the opioid abuse issue in this way sounds great on paper, but assuaging the crisis is not so simple. The guidelines suggest “non-opioid therapy is preferred for chronic pain outside of active cancer, palliative, and end-of-life care,” and anyone falling into the “other” category should seek alternative treatments.
People in the “other” category struggle with conditions like chronic low back pain, post-surgical and/or chronic knee pain, or arthritis that make daily functions quite difficult. Patients suffering from these conditions may not be fully aware of safer alternatives to a painkiller regimen. They may be under the impression that those pharmaceuticals are the only means to reach a manageable level of pain. Education is critical here—both patients and physicians should be made aware of viable treatment strategies that either lessen or remove opioids from the treatment regimen. For example, methods patients might consider to manage long-term pain could include anesthetic injections to affected joints (which at most last about 18 hours), or ablation of the troublesome nerve.
Radiofrequency ablation (or RFA) is a procedure in which radio wave-generated electricity heats a section of nerve tissue via a microelectrode inserted through a needle to diminish pain signals from a specific area. RFA has found success in treating chronic low back and neck pain, as well as arthritis-related joint deterioration. There is some question about the degree—but according to the Mayfield Clinic, pain relief can last from nine months to over two years.1
For chronic pain persisting beyond that span of time, implantable or surgical nerve stimulation (neurostimulation, or neurostim) devices could be a viable treatment option. Neurostimulation therapy (also known as spinal cord stimulation, or SCS) involves application of low voltage electricity to nerves in the spine via a neurostim implant or instrument in order to restrict pain signals to the brain, often replacing them with a tingling sensation (paresthesia).
Chronic low back pain (CLBP) is one such prevalent pain condition requiring a great deal of healthcare resources, including a troubling amount of opioid use. Adding to that, the condition might not be well-managed due to limited knowledge of chronic pain mechanisms and a lack of universally accepted treatment guidelines. Further complicating matters, CLBP can be broken down into two subcategories, each requiring a tweak in the regimen. The source of the pain for each can be quite difficult to check, especially without physical signs.
“In many patients with CLBP, imaging reveals no defect that can be addressed by surgery,” explained Prashant Rawat, chief operating officer of Mainstay Medical, a Dublin, Ireland-based medical device company focused on bringing to market an implantable neurostimulation system, ReActiv8, for people with disabling CLBP. “Consequently, these patients have had no prior surgery and are not suitable candidates for surgery. The etiology of their low back pain can be deduced to be predominantly mechanical (nociceptive) in nature through the use of imaging, physical medicine-based office tests, and the nature of pain symptoms. These patients are said to suffer from Chronic Mechanical Low Back Pain (CMLBP) or predominantly nociceptive low back pain.”
CMLBP is usually caused by damage to body tissue and manifests in an aching or throbbing. It can occur following trauma, surgery, or as a result of arthritis. Although nociceptive pain can ease as the injury heals, in more severe cases, it can persist into a chronic condition.
“After conventional management has failed them, the severely affected CMLBP patients frequently end up on long-term medication, including opioids,” said Rawat.
Chronic knee pain also afflicts a staggering amount of people—according to the American Osteopathic Association, it’s the number two cause of chronic pain and over a third of Americans report being affected by it. It can arise from an injury, too much weight on the knee joint, improper technique during activity, or can manifest following a knee replacement. There are a variety of non-opioid treatments to lessen the pain—patients might consider alternating warm and cool treatments, incorporating strength and mobility training, stretching, or a change in footwear to manage the main, among many other options.
“For chronic knee pain, the treatment algorithm might involve rehab, prescription pain medication, joint injections/nerve blocks, nerve ablation, arthroscopic knee surgery, total knee arthroplasty (TKA), and spinal cord stimulation,” commented Mark Geiger, global director of marketing, implantables, for Bioness Inc., a Valencia, Calif- based medical device company that provides technologies helping people regain mobility and independence. The company produces implantable and external neuromodulation systems, robotic systems, and software-based therapy programs providing functional and therapeutic benefits for individuals affected by pain, central nervous system disorders, and orthopedic injuries.
“There are downsides to every therapy on this list,” Geiger went on. “Opioids have side effects and can lead to a dangerous addiction. There are diminishing marginal returns of nerve blocks and joint injections. Nerve ablation is non-reversible in some cases and doesn’t always address chronic pain. Some patients have worse knee pain after arthroscopic knee surgery. There are 700,000 TKAs performed in the U.S. per year, with 170,000 of those patients still having chronic pain after their total knee replacement. Finally, spinal cord stimulation is a major surgery with a large, global implant.”
Strategic Stimulation
The first foray into neurostim as a treatment for pain dates back to 46 A.D. in Mesopotamia, with use of the torpedo fish (also referred to as the electric ray). Healers back then had a rudimentary understanding of the nervous system, but knew enough to use the fish as a proto-TENS (transcutaneous electrical nerve stimulation) device—patients either applied the electric ray to the painful area directly or placed the aching extremity in a pool of water containing the fish.
The first implanted neurostimulator for pain management application arrived nearly two millennia later in 1964 (then called the dorsal column stimulator), placed in the spine for pain relief. Today, implanting a neurostim device is an outpatient procedure, and the system is reversible should the patient seek other options. Patients can control their level of stimulation with a handheld device, and in the latest products are able to program their device to customize their pain relief.
Released last October, the Proclaim DRG neurostim system developed by medtech multinational Abbott Laboratories’ Neuromodulation division, which was a part of the St. Jude Medical acquisition, operates via dorsal root ganglion (DRG) stimulation. The DRG—a group of epidural and intraspinal nerve cells adjacent to the dorsal nerve root—is a promising target of stimulation because it houses primary sensory neurons (PSN). These PSNs represent a processing point of sensory information and facilitate sensory information flow from the periphery to the central nervous system.
Proclaim DRG is used to alleviate lower limb and hip pain in complex regional pain syndrome (CRPS) patients. It also incorporates Bluetooth wireless technology and software to use a tablet as the system’s controller. Like many implantable neuromodulation or cardiac rhythm management systems of the day, it can also safely endure an MRI scan.
“Both clinical and real world data have shown that DRG stimulation produces long-term, meaningful pain relief for patients with complex regional pain syndromes—like those resulting after total knee arthroplasty, foot surgery, or hernia surgery,” said Timothy Deer, M.D., an interventional pain physician, president, and CEO of the Spine and Nerve Center of the Virginias in Charleston, W.V. “The ability to offer DRG stimulation on the Proclaim platform is a very exciting advancement for those of us who treat patients with these debilitating conditions and want to provide relief from hard-to-treat neuropathic conditions.”
Nevro is a Redwood City, Calif.-based medical device company that began its foray into SCS technology in 2010 with the CE marked Senza system—FDA approved as of 2015—which applied the company’s proprietary HF10 SCS treatment. HF10 gets its name from combining high frequency (10 kHz) stimulation with a specific waveform and treatment algorithm. It has demonstrated considerable success in treating both chronic leg and back pain, and provides relief without paresthesia. Nevro’s Senza II system, which is a notably smaller size than its predecessor, won an FDA nod this past January.
“My colleagues and I are excited about the approval of the Senza II SCS system,” said Dr. Tom Simopoulos, director of pain medicine and co-director of the Spine Center at Beth Israel Deaconess Medical Center (an affiliate of the Harvard Medical School) in Boston, Mass. “The reduced size and optimized design of the Senza II implantable pulse generator (IPG) allow for greater patient comfort and placement options. Most importantly, it delivers HF10 therapy, a non-opioid treatment option that provides profound and paresthesia-free pain relief for patients.”
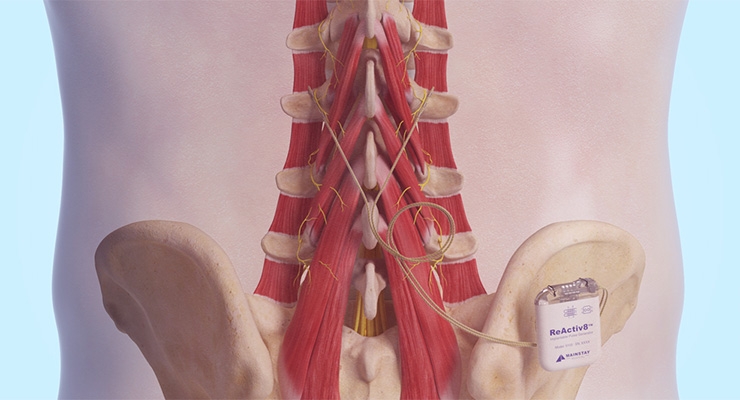
Mainstay Medical’s ReActiv8 device operates on an entirely different principle of neurostimulation. According to the company, studies have shown that restoring muscle activation can be a significant step in treating CLBP. The ReActiv8 implant works by activating a key set of muscles responsible for stabilizing the lower back. The method of treatment is similar to exercise programs that recover muscular control—patients deliver half-hour sessions twice daily via a handheld, wireless remote control.
ReActiv8 operates on the idea that repeated stimulation will eventually “teach” the brain and body to control the muscles in between sessions. Restoring this control and spine stabilization may ultimately allow the body to recover from CMLBP. The ReActiv8 implant is currently CE marked and indicated for investigational use in the United States. It is currently undergoing the ReActiv8-B U.S. pivotal trial—enrollment is expected to end in Q2 of this year, with a full data readout toward the end of the year.
“To date, neurostimulation therapies for chronic pain have focused on masking the sensation of pain,” explained Rawat. “ReActiv8 uses a new approach, which targets the underlying functional cause of pain. ReActiv8 is not a pain management strategy in the conventional sense, in that it does not mask the pain. Instead, it targets the underlying cause of pain by facilitating restoration of functional segmental stability. The treatment targets patients who specifically have severe and disabling CMLBP despite conventional management, including physical therapy and existing pain management strategies.”
The multifidus muscle attaches to the spine at multiple levels, and is the strongest stabilizer of the lower back. It is activated segmentally, coordinated by the central nervous system in response to sensory and proprioceptive stimuli. Joint pain originating from a strain or sprain of ligaments, tendons, joint capsule, or discs disrupts motor control and inhibits the multifidus muscle, allowing spine joints to move in a manner that exceed the “pain free zone.” The result is a cycle of chronic pain.
Ultrasound image-guided exercise programs targeting the multifidus muscle have been found to improve low back pain. But when that method fails to bring patients to a manageable pain level, electrical stimulation of these muscles could reactivate motor control and lead to improvements in CMLBP.
“Though motor control exercises are often prescribed for this purpose, many patients find them impossible or too painful to learn, and they often fail to reactivate the multifidus muscle which is normally not under volitional control,” noted Rawat. “ReActiv8 stimulates the nerves that innervate the multifidus muscles to elicit repetitive contractions. The hypothesized mechanism of action is that proprioception caused by the muscle contractions override the inhibitory reflex and facilitate restoration of muscle control, and eventually segmental stability.”
Peripheral nerve stimulation (PNS) is another common approach to treat chronic pain. In this method, the patient undergoes surgery to place a small, wire-like electrode next to a peripheral nerve—these are located beyond the brain or spinal cord. This electrode delivers electrical pulses to mask pain with paresthesia. PNS was invented before even SCS with roots in the mid-1960s, and has found success in treating complex regional pain syndromes and pain resulting from injuries to the peripheral nerves.
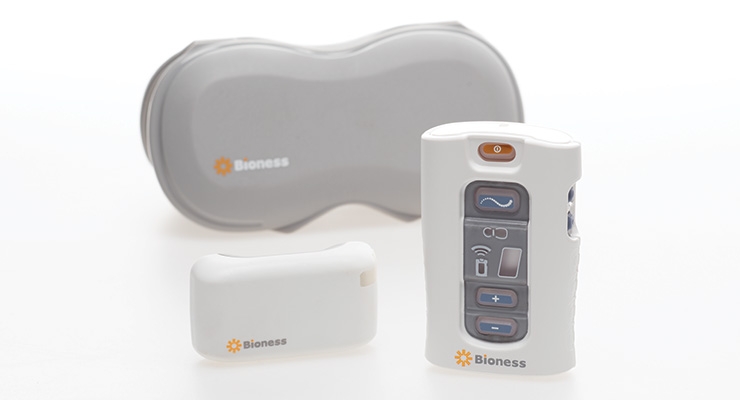
Bioness Inc.’s StimRouter neuromodulation system employs PNS therapy to treat adults with severe intractable pain originating from a peripheral nerve, working as an adjunct to other modes of therapy. Applications can include chronic pain conditions located at or related to the upper or lower limbs, entrapment syndromes, intercostal neuralgias, or other peripheral injuries or diseases. StimRouter received FDA 510(k) clearance in February 2015, and the first U.S. procedure took place in September of that year. The first European implantations came about in February 2017.
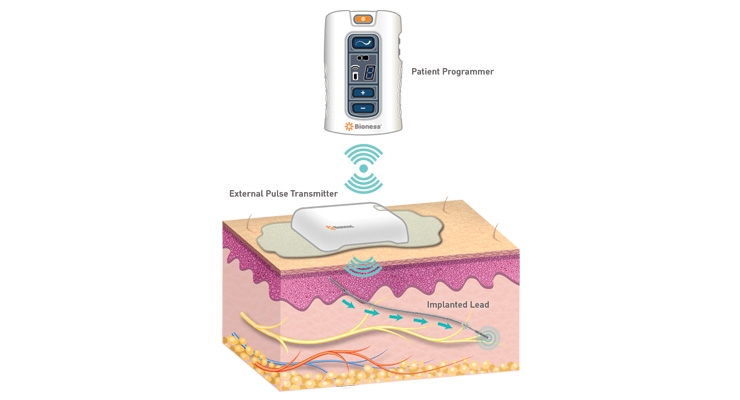
“The StimRouter is a small, thin, 15 cm passive lead implanted in a percutaneous fashion with a small incision under local anesthesia,” explained Geiger. “The tip of the lead is placed near the target peripheral nerve that’s causing the chronic pain. The ‘receiver’ portion of the lead is left near the surface of the skin (subcutaneously) so the ‘wearable’ can communicate with it through the skin. The patient turns the system on with a Patient Programmer that resembles a small remote control. The External Pulse Transmitter broadcasts a wave of energy through the skin that is picked up by the implanted receiver and ‘narrow-casted’ to the electrodes that stimulate the target peripheral nerve. The patient feels a tingling sensation instead of their pain and the patient can turn the device on, off, up, down, and change programs to deliver on-demand pain relief.”
For other types of rehab, Bioness also offers wearable systems like the L300 Foot Drop System (for those living with foot drop, a gait abnormality causing difficulty in lifting the front part of the foot), L300 Plus System (for foot drop plus thigh weakness), and the H200 Hand Rehabilitation System for hand paralysis therapy. The company’s Vector Gait & Safety System provides physical and occupational therapy, and Bioness completes the set with an integrated multi-disciplinary therapy solution with 24 programs and four standardized assessments.
The only implanted component of StimRouter is the thin, wire-like electrode. It therefore eliminates many of the risks associated with an SCS implant, which can include implant migration as well as other surgical complications. Serious injuries are relatively rare with implanted neurostim devices, and are commonly due to device complications rather than physical problems. In these cases, reprogramming, surgical lead replacement, or corrective surgery may be required.
StimRouter began receiving coverage from insurer Aetna Inc. in March of last year. This was considered a huge win in an environment where there’s no fast track for reimbursement regarding innovative pain management technologies. Further, devices that work in a similar manner to an already existing device, but are more inexpensive, can be difficult to get through the Centers for Medicare & Medicaid Services (CMS) because the system isn’t conducive to finding less costly solutions. Further, since the clinical mindset of “one-size fits all” approach to opioid prescription for chronic pain remains an option—albeit, one declining in favor since recent increased FDA attention to the epidemic—clear benefits of long-term usage of a device must be presented with a wealth of data to prove efficacy over traditional treatments.
“There is no doubt in today’s global environment, it is a challenge to obtain reimbursement for any new treatment addressing an unserved patient population,” stated Rawat. “This challenge is not limited to just treatments for chronic pain conditions. However, if the benefit of a treatment is compelling not only to patients, but also to future healthcare costs, the ability to return to work, and the reduction of the opioid crisis, we believe insurance companies will cover such treatments.”
The clamor is rising from patients and physicians to make these options affordable, but the challenge of coverage for innovative devices meets the roadblock of being deemed “experimental,” and therefore they are ineligible for reimbursement. For example, Nevro’s Senza SCS implant received an experimental designation from insurers in Highmark BCBS Pennsylvania and Blue Cross Blue Shield Tennessee in 2016. Although CMS officially determines reimbursement from Medicare and Medicaid, private insurers will often follow CMS’s lead. There is some hope, however, that tides are beginning to turn with regard to neurostimulation technologies to treat chronic pain thanks to the reinforcement of strong clinical data proving its safety and efficacy.
“Reimbursement is a challenge, but Medicare and private payers are realizing that PNS is not only effective, but also economical compared to many other neuromodulation therapies,” declared Geiger. “Sometimes these technologies cost six to seven times more than the StimRouter. In fact, reimbursement for StimRouter has increased significantly two years in a row for both the facility and physician payments. We believe this is a clear signal about the benefits of PNS.”
One can only hope that patients, physicians, and payers are receiving these signals as well.
Reference
According to Centers for Disease Control and Prevention (CDC) research, as many as one in four patients receiving long-term therapy in primary care settings may struggle with opioid use disorder. The number of opioids prescribed and sold in the United States has increased fourfold since 1999, with over 165,000 reported deaths from prescription opioid-related overdoses.
“The issue of opioid misuse and abuse remains one of my highest priorities,” FDA Commissioner Scott Gottlieb proclaimed in a statement. “We believe it’s going to take carefully developed, sustained, and coordinated action by everyone involved to reduce the tide of opioid addiction and death afflicting our communities; while maintaining appropriate prescribing for patients in medical need. We recognize both the urgency and complexity of this crisis and are taking several steps today toward these goals.”
While narcotic drugs help manage pain in the short term, there’s not enough information available about their long-term risks and benefits. The information coming to light concerning long term use for chronic pain, however, is troublesome. NIH statistics report roughly 21 to 29 percent of patients prescribed opioids for chronic pain misuse them, and between 8 and 12 percent develop an opioid use disorder.
In response to this threat, last year the CDC released its “Guideline for Prescribing Opioids for Chronic Pain,” outlining steps primary care clinicians should take in treating adult patients for chronic pain in outpatient settings. It exhorts clinicians to use nonpharmacologic therapy or non-opioid pharmacologic therapy (medications like acetaminophen or ibuprofen) as the preferred treatment method for chronic pain, to establish goals for pain and function, and to use immediate-release opioids when beginning treatment, among several other guidelines.
Remediating the opioid abuse issue in this way sounds great on paper, but assuaging the crisis is not so simple. The guidelines suggest “non-opioid therapy is preferred for chronic pain outside of active cancer, palliative, and end-of-life care,” and anyone falling into the “other” category should seek alternative treatments.
People in the “other” category struggle with conditions like chronic low back pain, post-surgical and/or chronic knee pain, or arthritis that make daily functions quite difficult. Patients suffering from these conditions may not be fully aware of safer alternatives to a painkiller regimen. They may be under the impression that those pharmaceuticals are the only means to reach a manageable level of pain. Education is critical here—both patients and physicians should be made aware of viable treatment strategies that either lessen or remove opioids from the treatment regimen. For example, methods patients might consider to manage long-term pain could include anesthetic injections to affected joints (which at most last about 18 hours), or ablation of the troublesome nerve.
Radiofrequency ablation (or RFA) is a procedure in which radio wave-generated electricity heats a section of nerve tissue via a microelectrode inserted through a needle to diminish pain signals from a specific area. RFA has found success in treating chronic low back and neck pain, as well as arthritis-related joint deterioration. There is some question about the degree—but according to the Mayfield Clinic, pain relief can last from nine months to over two years.1
For chronic pain persisting beyond that span of time, implantable or surgical nerve stimulation (neurostimulation, or neurostim) devices could be a viable treatment option. Neurostimulation therapy (also known as spinal cord stimulation, or SCS) involves application of low voltage electricity to nerves in the spine via a neurostim implant or instrument in order to restrict pain signals to the brain, often replacing them with a tingling sensation (paresthesia).
Chronic low back pain (CLBP) is one such prevalent pain condition requiring a great deal of healthcare resources, including a troubling amount of opioid use. Adding to that, the condition might not be well-managed due to limited knowledge of chronic pain mechanisms and a lack of universally accepted treatment guidelines. Further complicating matters, CLBP can be broken down into two subcategories, each requiring a tweak in the regimen. The source of the pain for each can be quite difficult to check, especially without physical signs.
“In many patients with CLBP, imaging reveals no defect that can be addressed by surgery,” explained Prashant Rawat, chief operating officer of Mainstay Medical, a Dublin, Ireland-based medical device company focused on bringing to market an implantable neurostimulation system, ReActiv8, for people with disabling CLBP. “Consequently, these patients have had no prior surgery and are not suitable candidates for surgery. The etiology of their low back pain can be deduced to be predominantly mechanical (nociceptive) in nature through the use of imaging, physical medicine-based office tests, and the nature of pain symptoms. These patients are said to suffer from Chronic Mechanical Low Back Pain (CMLBP) or predominantly nociceptive low back pain.”
CMLBP is usually caused by damage to body tissue and manifests in an aching or throbbing. It can occur following trauma, surgery, or as a result of arthritis. Although nociceptive pain can ease as the injury heals, in more severe cases, it can persist into a chronic condition.
“After conventional management has failed them, the severely affected CMLBP patients frequently end up on long-term medication, including opioids,” said Rawat.
Chronic knee pain also afflicts a staggering amount of people—according to the American Osteopathic Association, it’s the number two cause of chronic pain and over a third of Americans report being affected by it. It can arise from an injury, too much weight on the knee joint, improper technique during activity, or can manifest following a knee replacement. There are a variety of non-opioid treatments to lessen the pain—patients might consider alternating warm and cool treatments, incorporating strength and mobility training, stretching, or a change in footwear to manage the main, among many other options.
“For chronic knee pain, the treatment algorithm might involve rehab, prescription pain medication, joint injections/nerve blocks, nerve ablation, arthroscopic knee surgery, total knee arthroplasty (TKA), and spinal cord stimulation,” commented Mark Geiger, global director of marketing, implantables, for Bioness Inc., a Valencia, Calif- based medical device company that provides technologies helping people regain mobility and independence. The company produces implantable and external neuromodulation systems, robotic systems, and software-based therapy programs providing functional and therapeutic benefits for individuals affected by pain, central nervous system disorders, and orthopedic injuries.
“There are downsides to every therapy on this list,” Geiger went on. “Opioids have side effects and can lead to a dangerous addiction. There are diminishing marginal returns of nerve blocks and joint injections. Nerve ablation is non-reversible in some cases and doesn’t always address chronic pain. Some patients have worse knee pain after arthroscopic knee surgery. There are 700,000 TKAs performed in the U.S. per year, with 170,000 of those patients still having chronic pain after their total knee replacement. Finally, spinal cord stimulation is a major surgery with a large, global implant.”
Strategic Stimulation
The first foray into neurostim as a treatment for pain dates back to 46 A.D. in Mesopotamia, with use of the torpedo fish (also referred to as the electric ray). Healers back then had a rudimentary understanding of the nervous system, but knew enough to use the fish as a proto-TENS (transcutaneous electrical nerve stimulation) device—patients either applied the electric ray to the painful area directly or placed the aching extremity in a pool of water containing the fish.
The first implanted neurostimulator for pain management application arrived nearly two millennia later in 1964 (then called the dorsal column stimulator), placed in the spine for pain relief. Today, implanting a neurostim device is an outpatient procedure, and the system is reversible should the patient seek other options. Patients can control their level of stimulation with a handheld device, and in the latest products are able to program their device to customize their pain relief.
Released last October, the Proclaim DRG neurostim system developed by medtech multinational Abbott Laboratories’ Neuromodulation division, which was a part of the St. Jude Medical acquisition, operates via dorsal root ganglion (DRG) stimulation. The DRG—a group of epidural and intraspinal nerve cells adjacent to the dorsal nerve root—is a promising target of stimulation because it houses primary sensory neurons (PSN). These PSNs represent a processing point of sensory information and facilitate sensory information flow from the periphery to the central nervous system.
Proclaim DRG is used to alleviate lower limb and hip pain in complex regional pain syndrome (CRPS) patients. It also incorporates Bluetooth wireless technology and software to use a tablet as the system’s controller. Like many implantable neuromodulation or cardiac rhythm management systems of the day, it can also safely endure an MRI scan.
“Both clinical and real world data have shown that DRG stimulation produces long-term, meaningful pain relief for patients with complex regional pain syndromes—like those resulting after total knee arthroplasty, foot surgery, or hernia surgery,” said Timothy Deer, M.D., an interventional pain physician, president, and CEO of the Spine and Nerve Center of the Virginias in Charleston, W.V. “The ability to offer DRG stimulation on the Proclaim platform is a very exciting advancement for those of us who treat patients with these debilitating conditions and want to provide relief from hard-to-treat neuropathic conditions.”
Nevro is a Redwood City, Calif.-based medical device company that began its foray into SCS technology in 2010 with the CE marked Senza system—FDA approved as of 2015—which applied the company’s proprietary HF10 SCS treatment. HF10 gets its name from combining high frequency (10 kHz) stimulation with a specific waveform and treatment algorithm. It has demonstrated considerable success in treating both chronic leg and back pain, and provides relief without paresthesia. Nevro’s Senza II system, which is a notably smaller size than its predecessor, won an FDA nod this past January.
“My colleagues and I are excited about the approval of the Senza II SCS system,” said Dr. Tom Simopoulos, director of pain medicine and co-director of the Spine Center at Beth Israel Deaconess Medical Center (an affiliate of the Harvard Medical School) in Boston, Mass. “The reduced size and optimized design of the Senza II implantable pulse generator (IPG) allow for greater patient comfort and placement options. Most importantly, it delivers HF10 therapy, a non-opioid treatment option that provides profound and paresthesia-free pain relief for patients.”
Mainstay Medical’s ReActiv8 device operates on an entirely different principle of neurostimulation. According to the company, studies have shown that restoring muscle activation can be a significant step in treating CLBP. The ReActiv8 implant works by activating a key set of muscles responsible for stabilizing the lower back. The method of treatment is similar to exercise programs that recover muscular control—patients deliver half-hour sessions twice daily via a handheld, wireless remote control.
ReActiv8 operates on the idea that repeated stimulation will eventually “teach” the brain and body to control the muscles in between sessions. Restoring this control and spine stabilization may ultimately allow the body to recover from CMLBP. The ReActiv8 implant is currently CE marked and indicated for investigational use in the United States. It is currently undergoing the ReActiv8-B U.S. pivotal trial—enrollment is expected to end in Q2 of this year, with a full data readout toward the end of the year.
“To date, neurostimulation therapies for chronic pain have focused on masking the sensation of pain,” explained Rawat. “ReActiv8 uses a new approach, which targets the underlying functional cause of pain. ReActiv8 is not a pain management strategy in the conventional sense, in that it does not mask the pain. Instead, it targets the underlying cause of pain by facilitating restoration of functional segmental stability. The treatment targets patients who specifically have severe and disabling CMLBP despite conventional management, including physical therapy and existing pain management strategies.”
The multifidus muscle attaches to the spine at multiple levels, and is the strongest stabilizer of the lower back. It is activated segmentally, coordinated by the central nervous system in response to sensory and proprioceptive stimuli. Joint pain originating from a strain or sprain of ligaments, tendons, joint capsule, or discs disrupts motor control and inhibits the multifidus muscle, allowing spine joints to move in a manner that exceed the “pain free zone.” The result is a cycle of chronic pain.
Ultrasound image-guided exercise programs targeting the multifidus muscle have been found to improve low back pain. But when that method fails to bring patients to a manageable pain level, electrical stimulation of these muscles could reactivate motor control and lead to improvements in CMLBP.
“Though motor control exercises are often prescribed for this purpose, many patients find them impossible or too painful to learn, and they often fail to reactivate the multifidus muscle which is normally not under volitional control,” noted Rawat. “ReActiv8 stimulates the nerves that innervate the multifidus muscles to elicit repetitive contractions. The hypothesized mechanism of action is that proprioception caused by the muscle contractions override the inhibitory reflex and facilitate restoration of muscle control, and eventually segmental stability.”
Peripheral nerve stimulation (PNS) is another common approach to treat chronic pain. In this method, the patient undergoes surgery to place a small, wire-like electrode next to a peripheral nerve—these are located beyond the brain or spinal cord. This electrode delivers electrical pulses to mask pain with paresthesia. PNS was invented before even SCS with roots in the mid-1960s, and has found success in treating complex regional pain syndromes and pain resulting from injuries to the peripheral nerves.
Bioness Inc.’s StimRouter neuromodulation system employs PNS therapy to treat adults with severe intractable pain originating from a peripheral nerve, working as an adjunct to other modes of therapy. Applications can include chronic pain conditions located at or related to the upper or lower limbs, entrapment syndromes, intercostal neuralgias, or other peripheral injuries or diseases. StimRouter received FDA 510(k) clearance in February 2015, and the first U.S. procedure took place in September of that year. The first European implantations came about in February 2017.
“The StimRouter is a small, thin, 15 cm passive lead implanted in a percutaneous fashion with a small incision under local anesthesia,” explained Geiger. “The tip of the lead is placed near the target peripheral nerve that’s causing the chronic pain. The ‘receiver’ portion of the lead is left near the surface of the skin (subcutaneously) so the ‘wearable’ can communicate with it through the skin. The patient turns the system on with a Patient Programmer that resembles a small remote control. The External Pulse Transmitter broadcasts a wave of energy through the skin that is picked up by the implanted receiver and ‘narrow-casted’ to the electrodes that stimulate the target peripheral nerve. The patient feels a tingling sensation instead of their pain and the patient can turn the device on, off, up, down, and change programs to deliver on-demand pain relief.”
For other types of rehab, Bioness also offers wearable systems like the L300 Foot Drop System (for those living with foot drop, a gait abnormality causing difficulty in lifting the front part of the foot), L300 Plus System (for foot drop plus thigh weakness), and the H200 Hand Rehabilitation System for hand paralysis therapy. The company’s Vector Gait & Safety System provides physical and occupational therapy, and Bioness completes the set with an integrated multi-disciplinary therapy solution with 24 programs and four standardized assessments.
The only implanted component of StimRouter is the thin, wire-like electrode. It therefore eliminates many of the risks associated with an SCS implant, which can include implant migration as well as other surgical complications. Serious injuries are relatively rare with implanted neurostim devices, and are commonly due to device complications rather than physical problems. In these cases, reprogramming, surgical lead replacement, or corrective surgery may be required.
StimRouter began receiving coverage from insurer Aetna Inc. in March of last year. This was considered a huge win in an environment where there’s no fast track for reimbursement regarding innovative pain management technologies. Further, devices that work in a similar manner to an already existing device, but are more inexpensive, can be difficult to get through the Centers for Medicare & Medicaid Services (CMS) because the system isn’t conducive to finding less costly solutions. Further, since the clinical mindset of “one-size fits all” approach to opioid prescription for chronic pain remains an option—albeit, one declining in favor since recent increased FDA attention to the epidemic—clear benefits of long-term usage of a device must be presented with a wealth of data to prove efficacy over traditional treatments.
“There is no doubt in today’s global environment, it is a challenge to obtain reimbursement for any new treatment addressing an unserved patient population,” stated Rawat. “This challenge is not limited to just treatments for chronic pain conditions. However, if the benefit of a treatment is compelling not only to patients, but also to future healthcare costs, the ability to return to work, and the reduction of the opioid crisis, we believe insurance companies will cover such treatments.”
The clamor is rising from patients and physicians to make these options affordable, but the challenge of coverage for innovative devices meets the roadblock of being deemed “experimental,” and therefore they are ineligible for reimbursement. For example, Nevro’s Senza SCS implant received an experimental designation from insurers in Highmark BCBS Pennsylvania and Blue Cross Blue Shield Tennessee in 2016. Although CMS officially determines reimbursement from Medicare and Medicaid, private insurers will often follow CMS’s lead. There is some hope, however, that tides are beginning to turn with regard to neurostimulation technologies to treat chronic pain thanks to the reinforcement of strong clinical data proving its safety and efficacy.
“Reimbursement is a challenge, but Medicare and private payers are realizing that PNS is not only effective, but also economical compared to many other neuromodulation therapies,” declared Geiger. “Sometimes these technologies cost six to seven times more than the StimRouter. In fact, reimbursement for StimRouter has increased significantly two years in a row for both the facility and physician payments. We believe this is a clear signal about the benefits of PNS.”
One can only hope that patients, physicians, and payers are receiving these signals as well.
Reference
- http://bit.ly/odt180490 [PDF]