Mark Crawford, Contributing Writer08.15.18
Orthopedic devices remain one of the most traditional segments in medical product design and is therefore slow to adopt new technologies or innovative design solutions. Despite this resistance, orthopedic design is advancing—not just for new products, but also improving existing products and reducing their manufacturing costs. This trend is in part driven by market demands—baby boomers are retiring in record numbers and want to stay active, creating more demand for advanced products and orthopedic procedures. As a result, a fertile market exists for competitive designs and systems that result in better, safer, and faster outcomes, especially for this age group.
Although many OEMs are reluctant to push forward with new products, they still want to have a share of this tempting market. To stay involved, they are increasingly outsourcing product design to contract manufacturers (CMs) and design engineers. Smaller start-up companies are also outsourcing design because they don’t have the capital or expertise to do it on their own (including navigating regulatory pathways).
Ideally, OEMs want to partner with a trusted CM—from design through production, if possible. CMs are investing in more services, equipment, and expertise to meet these needs. To stand out from the crowd, CMs must know how to use the latest technologies and materials to design products—for example, additive manufacturing and how it can be used to customize devices and instruments. Therefore, an increasing number of small and large OEMs are partnering with CMs to bring advanced designs and products to the marketplace—without having to make heavy internal investments or create/maintain R&D departments.
“OEMs do want to be able to come up with new contributions like minimally invasive therapies, which reduce time in the OR and lead to quicker recoveries,” said Brett Gopal, senior director of innovation and network strategy with Orchid Orthopedic Solutions, a Holt, Mich.-based provider of contract manufacturing and design services for the orthopedic market. “At the end of the day it is about benefitting the patient—but OEMs would still like to make advanced products that cost less, wherever possible.”
“I attended a seminar recently where a major OEM stated that innovation and collaboration are alive and well,” said Philip Allen, director of sales and marketing for Lowell, a Minneapolis, Minn.-based contract manufacturer of complex implants for the orthopedic and cardiovascular markets. “Another said that they have more ideas than they can get to. This means design and development teams are working overtime on new projects. OEMs are using tuck-in acquisitions for new products, but are also engaged in breakthrough projects for major disease states. This creates many opportunities for us to assist in the design process.”
What OEMs Want
OEMs, of course, want everything, and at a lower cost. They want devices to be smaller, thinner, and stronger. They are interested in new materials and the latest advanced manufacturing techniques on the market, but also want their CMs to be the experts and assume the risks. OEMs seek device designs that are more anatomically correct. They want to add more complex function, but also want the design to be simple and low cost. Single-use products are getting more attention. The top priority, however, is always high quality, durability, and intuitive ease of use for surgeons or clinicians.
“For example, we design cases and trays so that brackets cannot be installed backwards, installed in the wrong locations, or missed altogether in the assembly operation,” said Ron Estes, vice president of operations for Estes Design and Manufacturing, an Indianapolis, Ind.-based sheet metal fabricator that provides design, engineering, prototyping, and manufacturing services to the orthopedic industry. “Field rejects are very costly for OEMs, so having simple, reliable designs is very important to them. Latches need to be intuitive and user-friendly, yet robust enough to survive drops in the field and reliable enough to survive the autoclave cycles. Cases and trays need to be serviceable through numerous cleaning and autoclave cycles, yet still function like new.”
Following the same theme, more OEMs are designing surgical instruments that are easier to clean. This means they must be easy to disassemble and reassemble. “More clever mechanisms are being used, instead of locking devices such as ball plungers and screws,” said Dave Neal, president and founder of STAT Design, a Morris Plains, N.J.-based designer and developer of orthopedic implants and instruments. “Locking mechanisms are being created by wire with electrical discharge machining [EDM] to create intricate spring connections in metal instruments. Also, encapsulated magnets within instruments are being used to connect one instrument to another, enhancing cleanability and reducing sterilization concerns.”
Medical device manufacturers are also getting better at understanding the value of integrating human factors engineering and industrial design into their products, which makes them more intuitive and easier to use for the end user. This is a simple and lower-cost way to add value, enhance functionality, and build brand reputation.
“While most orthopedic companies are engineer-driven, an increasing number of companies are embracing the need to build designs from a user perspective,” said Bryce Rutter, founder and CEO of Metaphase Design Group, a St. Louis, Mo.-based consulting firm that specializes human factors engineering, ergonomic design, and hand-intensive product and packaging design. “To a large extent, this trend has been influenced by the FDA's requirement to conduct usability research—both formative and summative usability studies—on all types of users who interact with a product’s design.”
A lower-cost approach for many OEMs is simply redesigning their existing products to add value and reduce cost. Some OEMs are reluctant to invest heavily to come up with a next-generation solution when instead they can boost revenues and reduce risk by value engineering existing products—maintaining or improving performance while stripping out costs. For example, some companies are considering how to take their legacy implants or instrument sets and reduce manufacturing costs by making them with 3D printing or other additive manufacturing techniques.
“More OEMs are asking how they can take what they have right now and streamline it to make it more attractive to hospitals so that they can either get a new hospital account, or have better margins and costs,” said Chris Scifert, director of engineering for Orchid Design, a division of Orchid Orthopedic Solutions offering design engineering and prototyping services for the orthopedic market.
The other driver behind lower cost is speed. OEMs seek designs that speed up product development by using approved materials and methods, reducing part complexity, making assembly easier, and streamlining regulatory approvals.
“Innovation is a given, but speed is absolutely critical,” said Daniel Gerbec, founder and president of Zigg Design, a Nibley, Utah-based consulting firm that provides design and manufacturing services to the orthopedic industry. “Every month of delay results in a tangible loss of revenue.”
Defining the OEM/Designer Relationship
How much design is outsourced varies by client and project. Some OEMs are hands-off, giving the CM complete freedom to design the entire system so that meets all the OEM-supplied design criteria. Others are very hands-on, providing plenty of input throughout the project. Gerbec notes that Zigg Design often works closely with an OEM’s development team, sometimes as high as executive corporate management. “We also spend a lot of time with an OEM’s consulting surgeon teams, making sure we understand their needs for safe and effective products,” he said.
If suppliers are allowed to design, it is best to work closely with the OEM team, noted Estes. “For example, the OEM team would need to supply Estes with models of the instruments/trials, along with the groupings of the instruments into each tray,” he said. “Sometimes the OEM even supplies the sequence of instrument use in the operation, just to make sure instruments are arranged logically. Once conceptual designs are submitted to the OEM, any number of iterations of design modifications can take place, prior to design lock.”
How much an OEM protects its intellectual property (IP) can also determine the level of involvement for outside designers. “One thing that’s attractive about how Orchid Design works, compared to other contract manufacturers, is that we don’t require the customer to assign us any IP,” said Scifert. “We know that the customer’s IP is important to them, so we don’t require them to share or license any IP that we develop in conjunction with their project.”
It is rare that an OEM has internal human factors engineering and industrial design resources. This is primarily due to the few number of new products they bring to market on an annual basis, which in turn would not cover the cost of these high-salary personnel. Instead, OEMs typically outsource these needs to companies like Metaphase Design Group.
“Our working relationships with OEMs start in the early stages of a new product development program,” said Rutter. “We remain deeply entrenched in these working teams through commercialization by providing continuous human factors engineering and industrial design services, including the formative and summative usability testing required by the FDA. Our scope of work often goes beyond the product to include the entire ecosystem surrounding that product, including instructions for use, design of mobile apps, and all forms of informational and instructional materials.”
OEMs outsource design to reduce costs and share risk. They expect their CMs to be at the top of their game in all aspects of design, including new tools and technologies, advantages and disadvantages of new materials, and masterful knowledge of regulatory compliance. Increasingly, OEMs simply want to turn their projects over to a trusted CM and await the solution. Therefore, medical device designers must stay on top of the latest advances in manufacturing technologies, materials, and changes in medical protocols. New materials, in particular, are challenging because they bring a new set of parameters that must receive regulatory clearance. This can push the product into a new classification category or make the product difficult to get cleared—risks that make OEMs uneasy.
“OEMs aren’t usually the ones trying to get new materials approved; that usually comes from material suppliers,” noted Arley Perez, senior product development engineer for Orchid Design. “Once approval is received, they are pretty heavy in pushing them. Even with new manufacturing processes for materials such as titanium, you’re at high volume so you can get a good impact. You want to stay on top of it because there are probably things that have been requested in the past that could be a good match for a new material. You don’t want customers asking you about it—instead you want to be informing them.”
“Designers must also understand the risks associated with the introduction of new materials,” added Gerbec. “Aside from the obvious risks surrounding biocompatibility, they must understand the changes in potential failure modes as they change material properties.”
For example, material behaviors can change depending upon the manufacturing process. “A Ti-6-4 implant may behave very differently if made via an additive process versus more traditional machining,” said Gerbec. “One of the reasons that we are able to make reliable Ti-6-4 implant via traditional machining is that the microstructure of the material is very well understood and controlled. This is the result of considerable work done in the aerospace industry during the 1950s-1960s. I feel that we still have far to go before we can use the additive process to make high-performance, safety-critical parts.”
Designers stay on top of technology and material trends in multiple ways, including attending conferences and reading technology magazines and Internet search reviews. “Missing out on a new technology or material could mean the difference in the company not achieving its minimum project rate of return and thereby cancelling the project,” said Neal. “Not only would the company miss out on potential sales, in the end it is the patient who suffers the most.”
New Design Technologies
An explosion of new software programs over the last four to five years has enabled a heightened level of detail and control that greatly expands design options. For example, nTopology Element software allows designers to create porous structures in CAD for AM techniques by utilizing user-defined input, imported data, or finite element analysis (FEA) results. Another program, Anybodytech.com, a musculoskeletal simulation product, provides a repository of body models with sample applications that allow designers to determine muscle and joint forces and other key biomechanical impacts on orthopedic products.
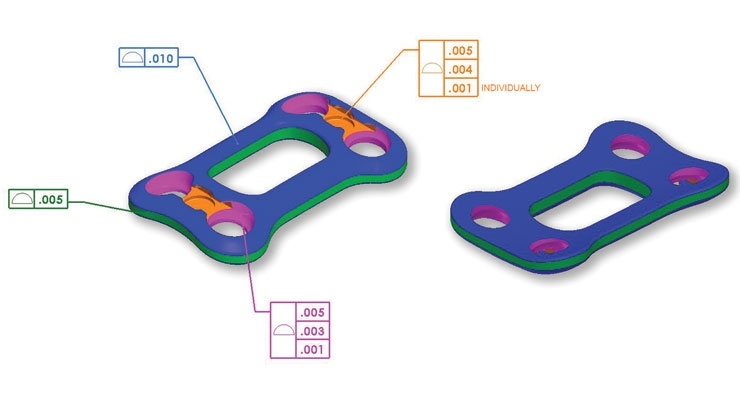
Lowell has been working with customers on broader use of geometric dimensioning and tolerancing (GD&T) and the ASME Y14.5 2009 standard. “Design, manufacturing, and inspection must all have a shared understanding, so we developed a process to manufacture components and assemblies that meet our customer’s requirements and design intent,” said Allen. “GD&T is a common language we use to reduce confusion and the possibility of error.”
The ASME Y14.5 2009 standard is especially helpful in three areas: datum mobility that allows for greater freedom in design functionality; coordinate measuring machine (CMM) algorithm selection to improve feature verification; and improvements to 3D CAD modelling for model-based definitions. “We see both GD&T and ASME Y14.5 2009 as essential to improving the design process in the future,” added Allen.
Additive manufacturing continues to advance rapidly, with a growing number of materials that can be printed, including metals and delicate bioresorbables (for example, vascular scaffolds). “With 3D printing, we can detail the design of an implant down to the exact porous coating, so we know exactly what sort of bone in-growth we will get,” said Justin Conway, lead additive manufacturing engineer at Orchid’s Memphis facility. “On the manufacturing side, software can show the complete trajectory of the melting beam we use to form a part, the energy consumed, and all the design inputs. A massive amount of input and detail can be put into a part before it is 3D-printed. We can do things now with design that weren’t possible just a few years ago.”
There is, however, a significant gap between AM and design. “This is a great place for divisions like Orchid Design to step in and work with customers on advising design for AM,” added Conway. “When we get involved on the front end of the design process, we can identify which products are good candidates for AM and which ones are not.”
Forward-thinking companies in other manufacturing industries have established additive manufacturing labs to teach their engineers how to design products specifically for additive manufacturing technologies. For example, Caterpillar has established its in-house “AM Factory” in Dunlap, Ill., which is outfitted with the latest AM and 3D printing equipment. Caterpillar engineers bring their projects into this space to invent products that can only be manufactured using AM.
Eventually the orthopedic market must embrace the Internet of Things (IoT) and Industry 4.0. This means integrating the same technologies that are starting to transform other industries, such as automation, robotics, sensors, data collection and analysis, simulation, virtual reality/augmented reality, and additive manufacturing.
“Mechanical engineers and designers, for example, are working with computer scientists to create software that is integrated into smart tools that have built-in sensors for proximity, that are taken from the game design industry,” said Neal. “These instruments are used to more precisely locate 3D position in space, improving the alignment and positioning of implants.”
“There is opportunity to reduce the cost of care while simultaneously improving patient safety through the collective intelligence capable with the IoT,” said Sean Hägen, principal and director of research and synthesis for BlackHägen Design, a Dunedin, Fla.-based consulting firm specializing in usability design and development. The initial challenges, he notes, are not technical: the willingness of clinicians to be monitored and how to protect the anonymity of the data for medilegal and privacy reasons.
In 2016 the FDA, in association with medical robotics companies, academia, and surgeons, held a Delphi Consensus Summit to outline what information future robotics systems should collect for a registry. The registry the FDA is proposing could collect some basic data that would enable the human factors engineers across the medical robotics industry to better understand use error (root cause). Hägen was one of the facilitators. “We covered a lot of interesting ideas, including inputs regarding use error and training opportunities,” he said. “The findings are just now starting to form really useful insights, such as developing decision-support algorithms that will be supported by IoT.”
Orchid is implementing its own internal IoT strategy. The first step is connecting all its assets via the Internet and seeing how these systems interact and perform. This is especially helpful for predictive maintenance and reducing downtime. “We then want take these IoT capabilities and use them to manufacture smart implants,” said Gopal. “We are in good position for developing technology and manufacturing for smart devices, but that is a longer-term goal. The first three-five years are focused on integrating IoT to create a smart factory. Phase 2 would then be doubling up on core competencies in order to make smart devices.”
Balancing Design and Cost
Trading off between design and cost is a constant challenge for medical devices designers. Without a constant eye on the economics of the manufacturing/packaging route associated with the device, costs can very easily get out of hand. Designers also need to be aware of what hospitals/insurance companies will pay for a particular medical product, which helps establish limits for how intricate or complex a new product can be. The goal, of course, is to increase functionality and performance, while keeping the design simple (lower costs, affordability).
“In our business, the trade-off is mostly related to the complexity of the assembly,” said Estes. “The simpler you can make the design to assemble, the more favorable the trade-off. Life cycle costs are important, too. A product that can achieve its targeted life span in years or cycles with no failures, even though it may be more expensive initially, will have lower cost of ownership for the customer over the entire life cycle.”
To control costs and simplify assembly, OEMs often work with their CMs to narrow the list of critical features in a design. This helps keep a project on track, on time, and reduce costs. “Critical feature confirmation, or what we call CFC, is a method to figure out which device features are critical and which ones are not,” said Allen. “It uses designed experiments to test product and design requirements against key design inputs. The test results indicate which features are critical. It alleviates pain points in getting a product to market in three important ways: accelerated design reviews, simplified quality control/inspection, and improving regulatory review.”
Although controlling cost is important, determining human performance in medical device design should not be undervalued. “All the technology in the world is meaningless if the design is unintuitive, or it creates use errors,” said Rutter. “The cost of being wrong downstream orders up a magnitude of more cost and dramatically increases legal risk, rather than the investment in human factors engineering to design out all potential confusion and to mitigate use errors.”
Additive manufacturing has the potential to create seemingly impossible designs at affordable costs. Companies such as Markforged are bringing down the cost of metal additive manufacturing equipment to make this technology more affordable for start-ups. Other breakthrough technologies in orthopedic design are virtual reality (VR) and augmented reality (AR). These help designers and manufacturers visualize and interact with their designs, without the need for fabricating expensive functional prototypes. Using AR and VR early in the design process reduces errors, keeps costs down, and increases speed to market.
Internet of Things technologies—additive manufacturing, sensor technologies, automation, robotics, VR and AR, and data analytics—are starting to take hold in the orthopedic design space and offer fantastic design and manufacturing potential. However, the pace is agonizingly slow for designers, who are eager use these tools to make next-generation products that transform the patient experience.
“We are already seeing the infusion of sensing technologies that convert ‘dumb’ instruments to smart instruments, the accelerated race in surgical robotics to personalized pharmacological prescriptions, wireless devices that live-stream our health status to our clinicians, and diagnostic devices that dramatically shorten test turnaround times from days to hours or minutes,” said Rutter. “Although it may initially be more expensive, companies that choose to be at the forefront of disruptive technologies will have a competitive edge in the marketplace.”
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.
Although many OEMs are reluctant to push forward with new products, they still want to have a share of this tempting market. To stay involved, they are increasingly outsourcing product design to contract manufacturers (CMs) and design engineers. Smaller start-up companies are also outsourcing design because they don’t have the capital or expertise to do it on their own (including navigating regulatory pathways).
Ideally, OEMs want to partner with a trusted CM—from design through production, if possible. CMs are investing in more services, equipment, and expertise to meet these needs. To stand out from the crowd, CMs must know how to use the latest technologies and materials to design products—for example, additive manufacturing and how it can be used to customize devices and instruments. Therefore, an increasing number of small and large OEMs are partnering with CMs to bring advanced designs and products to the marketplace—without having to make heavy internal investments or create/maintain R&D departments.
“OEMs do want to be able to come up with new contributions like minimally invasive therapies, which reduce time in the OR and lead to quicker recoveries,” said Brett Gopal, senior director of innovation and network strategy with Orchid Orthopedic Solutions, a Holt, Mich.-based provider of contract manufacturing and design services for the orthopedic market. “At the end of the day it is about benefitting the patient—but OEMs would still like to make advanced products that cost less, wherever possible.”
“I attended a seminar recently where a major OEM stated that innovation and collaboration are alive and well,” said Philip Allen, director of sales and marketing for Lowell, a Minneapolis, Minn.-based contract manufacturer of complex implants for the orthopedic and cardiovascular markets. “Another said that they have more ideas than they can get to. This means design and development teams are working overtime on new projects. OEMs are using tuck-in acquisitions for new products, but are also engaged in breakthrough projects for major disease states. This creates many opportunities for us to assist in the design process.”
What OEMs Want
OEMs, of course, want everything, and at a lower cost. They want devices to be smaller, thinner, and stronger. They are interested in new materials and the latest advanced manufacturing techniques on the market, but also want their CMs to be the experts and assume the risks. OEMs seek device designs that are more anatomically correct. They want to add more complex function, but also want the design to be simple and low cost. Single-use products are getting more attention. The top priority, however, is always high quality, durability, and intuitive ease of use for surgeons or clinicians.
“For example, we design cases and trays so that brackets cannot be installed backwards, installed in the wrong locations, or missed altogether in the assembly operation,” said Ron Estes, vice president of operations for Estes Design and Manufacturing, an Indianapolis, Ind.-based sheet metal fabricator that provides design, engineering, prototyping, and manufacturing services to the orthopedic industry. “Field rejects are very costly for OEMs, so having simple, reliable designs is very important to them. Latches need to be intuitive and user-friendly, yet robust enough to survive drops in the field and reliable enough to survive the autoclave cycles. Cases and trays need to be serviceable through numerous cleaning and autoclave cycles, yet still function like new.”
Following the same theme, more OEMs are designing surgical instruments that are easier to clean. This means they must be easy to disassemble and reassemble. “More clever mechanisms are being used, instead of locking devices such as ball plungers and screws,” said Dave Neal, president and founder of STAT Design, a Morris Plains, N.J.-based designer and developer of orthopedic implants and instruments. “Locking mechanisms are being created by wire with electrical discharge machining [EDM] to create intricate spring connections in metal instruments. Also, encapsulated magnets within instruments are being used to connect one instrument to another, enhancing cleanability and reducing sterilization concerns.”
Medical device manufacturers are also getting better at understanding the value of integrating human factors engineering and industrial design into their products, which makes them more intuitive and easier to use for the end user. This is a simple and lower-cost way to add value, enhance functionality, and build brand reputation.
“While most orthopedic companies are engineer-driven, an increasing number of companies are embracing the need to build designs from a user perspective,” said Bryce Rutter, founder and CEO of Metaphase Design Group, a St. Louis, Mo.-based consulting firm that specializes human factors engineering, ergonomic design, and hand-intensive product and packaging design. “To a large extent, this trend has been influenced by the FDA's requirement to conduct usability research—both formative and summative usability studies—on all types of users who interact with a product’s design.”
A lower-cost approach for many OEMs is simply redesigning their existing products to add value and reduce cost. Some OEMs are reluctant to invest heavily to come up with a next-generation solution when instead they can boost revenues and reduce risk by value engineering existing products—maintaining or improving performance while stripping out costs. For example, some companies are considering how to take their legacy implants or instrument sets and reduce manufacturing costs by making them with 3D printing or other additive manufacturing techniques.
“More OEMs are asking how they can take what they have right now and streamline it to make it more attractive to hospitals so that they can either get a new hospital account, or have better margins and costs,” said Chris Scifert, director of engineering for Orchid Design, a division of Orchid Orthopedic Solutions offering design engineering and prototyping services for the orthopedic market.
The other driver behind lower cost is speed. OEMs seek designs that speed up product development by using approved materials and methods, reducing part complexity, making assembly easier, and streamlining regulatory approvals.
“Innovation is a given, but speed is absolutely critical,” said Daniel Gerbec, founder and president of Zigg Design, a Nibley, Utah-based consulting firm that provides design and manufacturing services to the orthopedic industry. “Every month of delay results in a tangible loss of revenue.”
Defining the OEM/Designer Relationship
How much design is outsourced varies by client and project. Some OEMs are hands-off, giving the CM complete freedom to design the entire system so that meets all the OEM-supplied design criteria. Others are very hands-on, providing plenty of input throughout the project. Gerbec notes that Zigg Design often works closely with an OEM’s development team, sometimes as high as executive corporate management. “We also spend a lot of time with an OEM’s consulting surgeon teams, making sure we understand their needs for safe and effective products,” he said.
If suppliers are allowed to design, it is best to work closely with the OEM team, noted Estes. “For example, the OEM team would need to supply Estes with models of the instruments/trials, along with the groupings of the instruments into each tray,” he said. “Sometimes the OEM even supplies the sequence of instrument use in the operation, just to make sure instruments are arranged logically. Once conceptual designs are submitted to the OEM, any number of iterations of design modifications can take place, prior to design lock.”
How much an OEM protects its intellectual property (IP) can also determine the level of involvement for outside designers. “One thing that’s attractive about how Orchid Design works, compared to other contract manufacturers, is that we don’t require the customer to assign us any IP,” said Scifert. “We know that the customer’s IP is important to them, so we don’t require them to share or license any IP that we develop in conjunction with their project.”
It is rare that an OEM has internal human factors engineering and industrial design resources. This is primarily due to the few number of new products they bring to market on an annual basis, which in turn would not cover the cost of these high-salary personnel. Instead, OEMs typically outsource these needs to companies like Metaphase Design Group.
“Our working relationships with OEMs start in the early stages of a new product development program,” said Rutter. “We remain deeply entrenched in these working teams through commercialization by providing continuous human factors engineering and industrial design services, including the formative and summative usability testing required by the FDA. Our scope of work often goes beyond the product to include the entire ecosystem surrounding that product, including instructions for use, design of mobile apps, and all forms of informational and instructional materials.”
OEMs outsource design to reduce costs and share risk. They expect their CMs to be at the top of their game in all aspects of design, including new tools and technologies, advantages and disadvantages of new materials, and masterful knowledge of regulatory compliance. Increasingly, OEMs simply want to turn their projects over to a trusted CM and await the solution. Therefore, medical device designers must stay on top of the latest advances in manufacturing technologies, materials, and changes in medical protocols. New materials, in particular, are challenging because they bring a new set of parameters that must receive regulatory clearance. This can push the product into a new classification category or make the product difficult to get cleared—risks that make OEMs uneasy.
“OEMs aren’t usually the ones trying to get new materials approved; that usually comes from material suppliers,” noted Arley Perez, senior product development engineer for Orchid Design. “Once approval is received, they are pretty heavy in pushing them. Even with new manufacturing processes for materials such as titanium, you’re at high volume so you can get a good impact. You want to stay on top of it because there are probably things that have been requested in the past that could be a good match for a new material. You don’t want customers asking you about it—instead you want to be informing them.”
“Designers must also understand the risks associated with the introduction of new materials,” added Gerbec. “Aside from the obvious risks surrounding biocompatibility, they must understand the changes in potential failure modes as they change material properties.”
For example, material behaviors can change depending upon the manufacturing process. “A Ti-6-4 implant may behave very differently if made via an additive process versus more traditional machining,” said Gerbec. “One of the reasons that we are able to make reliable Ti-6-4 implant via traditional machining is that the microstructure of the material is very well understood and controlled. This is the result of considerable work done in the aerospace industry during the 1950s-1960s. I feel that we still have far to go before we can use the additive process to make high-performance, safety-critical parts.”
Designers stay on top of technology and material trends in multiple ways, including attending conferences and reading technology magazines and Internet search reviews. “Missing out on a new technology or material could mean the difference in the company not achieving its minimum project rate of return and thereby cancelling the project,” said Neal. “Not only would the company miss out on potential sales, in the end it is the patient who suffers the most.”
New Design Technologies
An explosion of new software programs over the last four to five years has enabled a heightened level of detail and control that greatly expands design options. For example, nTopology Element software allows designers to create porous structures in CAD for AM techniques by utilizing user-defined input, imported data, or finite element analysis (FEA) results. Another program, Anybodytech.com, a musculoskeletal simulation product, provides a repository of body models with sample applications that allow designers to determine muscle and joint forces and other key biomechanical impacts on orthopedic products.
Lowell has been working with customers on broader use of geometric dimensioning and tolerancing (GD&T) and the ASME Y14.5 2009 standard. “Design, manufacturing, and inspection must all have a shared understanding, so we developed a process to manufacture components and assemblies that meet our customer’s requirements and design intent,” said Allen. “GD&T is a common language we use to reduce confusion and the possibility of error.”
The ASME Y14.5 2009 standard is especially helpful in three areas: datum mobility that allows for greater freedom in design functionality; coordinate measuring machine (CMM) algorithm selection to improve feature verification; and improvements to 3D CAD modelling for model-based definitions. “We see both GD&T and ASME Y14.5 2009 as essential to improving the design process in the future,” added Allen.
Additive manufacturing continues to advance rapidly, with a growing number of materials that can be printed, including metals and delicate bioresorbables (for example, vascular scaffolds). “With 3D printing, we can detail the design of an implant down to the exact porous coating, so we know exactly what sort of bone in-growth we will get,” said Justin Conway, lead additive manufacturing engineer at Orchid’s Memphis facility. “On the manufacturing side, software can show the complete trajectory of the melting beam we use to form a part, the energy consumed, and all the design inputs. A massive amount of input and detail can be put into a part before it is 3D-printed. We can do things now with design that weren’t possible just a few years ago.”
There is, however, a significant gap between AM and design. “This is a great place for divisions like Orchid Design to step in and work with customers on advising design for AM,” added Conway. “When we get involved on the front end of the design process, we can identify which products are good candidates for AM and which ones are not.”
Forward-thinking companies in other manufacturing industries have established additive manufacturing labs to teach their engineers how to design products specifically for additive manufacturing technologies. For example, Caterpillar has established its in-house “AM Factory” in Dunlap, Ill., which is outfitted with the latest AM and 3D printing equipment. Caterpillar engineers bring their projects into this space to invent products that can only be manufactured using AM.
Eventually the orthopedic market must embrace the Internet of Things (IoT) and Industry 4.0. This means integrating the same technologies that are starting to transform other industries, such as automation, robotics, sensors, data collection and analysis, simulation, virtual reality/augmented reality, and additive manufacturing.
“Mechanical engineers and designers, for example, are working with computer scientists to create software that is integrated into smart tools that have built-in sensors for proximity, that are taken from the game design industry,” said Neal. “These instruments are used to more precisely locate 3D position in space, improving the alignment and positioning of implants.”
“There is opportunity to reduce the cost of care while simultaneously improving patient safety through the collective intelligence capable with the IoT,” said Sean Hägen, principal and director of research and synthesis for BlackHägen Design, a Dunedin, Fla.-based consulting firm specializing in usability design and development. The initial challenges, he notes, are not technical: the willingness of clinicians to be monitored and how to protect the anonymity of the data for medilegal and privacy reasons.
In 2016 the FDA, in association with medical robotics companies, academia, and surgeons, held a Delphi Consensus Summit to outline what information future robotics systems should collect for a registry. The registry the FDA is proposing could collect some basic data that would enable the human factors engineers across the medical robotics industry to better understand use error (root cause). Hägen was one of the facilitators. “We covered a lot of interesting ideas, including inputs regarding use error and training opportunities,” he said. “The findings are just now starting to form really useful insights, such as developing decision-support algorithms that will be supported by IoT.”
Orchid is implementing its own internal IoT strategy. The first step is connecting all its assets via the Internet and seeing how these systems interact and perform. This is especially helpful for predictive maintenance and reducing downtime. “We then want take these IoT capabilities and use them to manufacture smart implants,” said Gopal. “We are in good position for developing technology and manufacturing for smart devices, but that is a longer-term goal. The first three-five years are focused on integrating IoT to create a smart factory. Phase 2 would then be doubling up on core competencies in order to make smart devices.”
Balancing Design and Cost
Trading off between design and cost is a constant challenge for medical devices designers. Without a constant eye on the economics of the manufacturing/packaging route associated with the device, costs can very easily get out of hand. Designers also need to be aware of what hospitals/insurance companies will pay for a particular medical product, which helps establish limits for how intricate or complex a new product can be. The goal, of course, is to increase functionality and performance, while keeping the design simple (lower costs, affordability).
“In our business, the trade-off is mostly related to the complexity of the assembly,” said Estes. “The simpler you can make the design to assemble, the more favorable the trade-off. Life cycle costs are important, too. A product that can achieve its targeted life span in years or cycles with no failures, even though it may be more expensive initially, will have lower cost of ownership for the customer over the entire life cycle.”
To control costs and simplify assembly, OEMs often work with their CMs to narrow the list of critical features in a design. This helps keep a project on track, on time, and reduce costs. “Critical feature confirmation, or what we call CFC, is a method to figure out which device features are critical and which ones are not,” said Allen. “It uses designed experiments to test product and design requirements against key design inputs. The test results indicate which features are critical. It alleviates pain points in getting a product to market in three important ways: accelerated design reviews, simplified quality control/inspection, and improving regulatory review.”
Although controlling cost is important, determining human performance in medical device design should not be undervalued. “All the technology in the world is meaningless if the design is unintuitive, or it creates use errors,” said Rutter. “The cost of being wrong downstream orders up a magnitude of more cost and dramatically increases legal risk, rather than the investment in human factors engineering to design out all potential confusion and to mitigate use errors.”
Additive manufacturing has the potential to create seemingly impossible designs at affordable costs. Companies such as Markforged are bringing down the cost of metal additive manufacturing equipment to make this technology more affordable for start-ups. Other breakthrough technologies in orthopedic design are virtual reality (VR) and augmented reality (AR). These help designers and manufacturers visualize and interact with their designs, without the need for fabricating expensive functional prototypes. Using AR and VR early in the design process reduces errors, keeps costs down, and increases speed to market.
Internet of Things technologies—additive manufacturing, sensor technologies, automation, robotics, VR and AR, and data analytics—are starting to take hold in the orthopedic design space and offer fantastic design and manufacturing potential. However, the pace is agonizingly slow for designers, who are eager use these tools to make next-generation products that transform the patient experience.
“We are already seeing the infusion of sensing technologies that convert ‘dumb’ instruments to smart instruments, the accelerated race in surgical robotics to personalized pharmacological prescriptions, wireless devices that live-stream our health status to our clinicians, and diagnostic devices that dramatically shorten test turnaround times from days to hours or minutes,” said Rutter. “Although it may initially be more expensive, companies that choose to be at the forefront of disruptive technologies will have a competitive edge in the marketplace.”
Mark Crawford is a full-time freelance business and marketing/communications writer based in Madison, Wis. His clients range from startups to global manufacturing leaders. He also writes a variety of feature articles for regional and national publications and is the author of five books.