Maria Shepherd, President and Founder, Medi-Vantage03.29.19
Ten years ago—2009—was a controversial time for spine surgeons. This was the year the outcomes of two randomized controlled trials were published in The New England Journal of Medicine (NEJM).1,2 The results? The outcomes for vertebral compression fractures (VCF) randomized to vertebroplasty or a sham procedure showed comparable pain management for patients in the trials. Both studies concluded that vertebroplasty provided no clinical benefit. Additionally, the authors stated any benefits shown in prior studies were most likely due to a “placebo effect.” The backlash was immediate. Some physicians disputed the study design in the two trials. Other highly skilled surgeons with good outcomes in vertebroplasty questioned how this controversy was generated.
The medical device business impact of the studies was rapid and damaging. But what was the impact of the studies over the long term? One report3 published in 2010 stated vertebroplasty, kyphoplasty, and vertebral augmentation procedure rates declined by 10 percent after the NEJM publications in August 2009. There was much concern among surgeons and referring physicians that the procedure rate drop was due to a substantial decrease in patient referrals.
Why This Is Important
In 2009, worldwide estimates placed the number of patients suffering from VCF at approximately 1.4 million.4,5 Also in 2009, in the U.S., it was reported that out of the 750,000 cases of VCF diagnosed annually, approximately one third (250,000 patients) received treatment.5,6 Most vertebral compression fractures resolve over time with conservative treatment (e.g., bed rest, braces, pain medications). However, the significant segment of patients with VCF that do not resolve and improve continue to report chronic pain. These are the patients that are referred for a surgical procedure.
In 2017, the worldwide vertebral compression fracture medical device market was estimated at $2.381 billion and is forecasted to grow to $4.876 billion by 20267. This is a compound annual growth rate (CAGR) of 8.25 percent (Table 1).
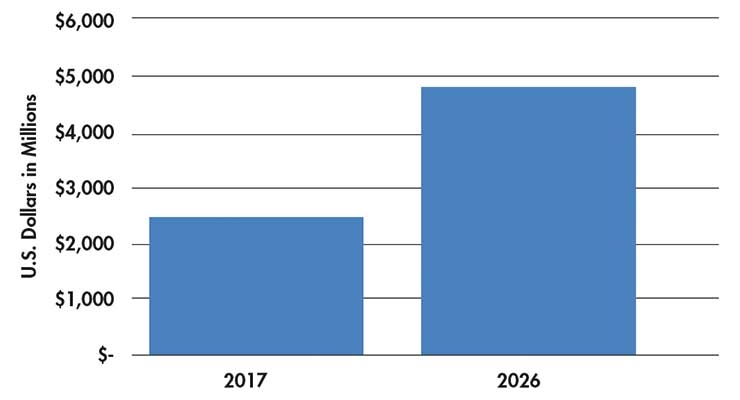
Table 1: Estimated global revenues from VCF surgical procedures7
Key market drivers are an aging population, reimbursement rates for minimally invasive surgical procedures for VCF repair, unhealthy lifestyles (e.g., obesity or smoking), and an increase in the popularity of kyphoplasty for the treatment of VCF.
Vertebroplasty vs. Kyphoplasty—Which Procedure Will Win?
In 2007, it was estimated there were more than 140,000 percutaneous VCF procedures and predictions had this number rising to over 180,000 by 20098—a CAGR of 13 percent. Then the publication of the outcomes of the two randomized controlled trials led to an editorial in the Journal9 reviewing the concerns of insurers and questioned whether they should limit or completely withdraw coverage for vertebroplasty procedures. Since then, Blue Cross Blue Shield announced that vertebroplasty and kyphoplasty did not meet the conditions of its efficacy criteria.10
The consequences of the NEJM studies focused high scrutiny on vertebroplasty and kyphoplasty procedures. The article also referenced VCF surgical complication rates (Table 2).11
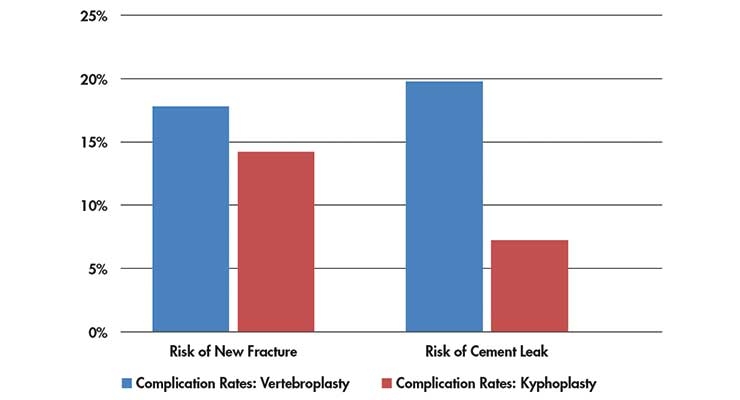
Table 2: Complication rates of VCF procedures11
The metanalysis contained 168 studies that met the study inclusion criteria. There was a statistically significant difference in improvement with vertebroplasty vs. kyphoplasty (p<0.001). New fracture risk was 17.9 percent with vertebroplasty vs. 14.1 percent with kyphoplasty (p<0.01). Cement leak risk was 19.7 percent with vertebroplasty versus 7 percent with kyphoplasty (p<0.001).11
That Was 2009—Where Are We 10 Years Later?
In a market research report, it was stated the 2016 North American vertebroplasty market (i.e., Canada, Mexico, U.S.) will drop from a forecasted $72 million to approximately $29.4 million by 2023.12
This report states that vertebroplasty, developed as a minimally invasive procedure to be performed in an outpatient setting for the treatment of VCF, has highly variable outcomes and definitive evidence demonstrating procedural efficacy does not exist. It predicted the North America market will see declining rates of adoption in favor of kyphoplasty.
The price tags of vertebroplasty and kyphoplasty are different enough to warrant increased adoption in developing and price sensitive markets. The report stated vertebroplasty revenues in the Middle East and Africa, (i.e., Egypt, Israel, Saudi Arabia, South Africa, and UAE) will increase from $4.1 million in 2016 to approximately $6.5 million by 2023 (Table 3).12
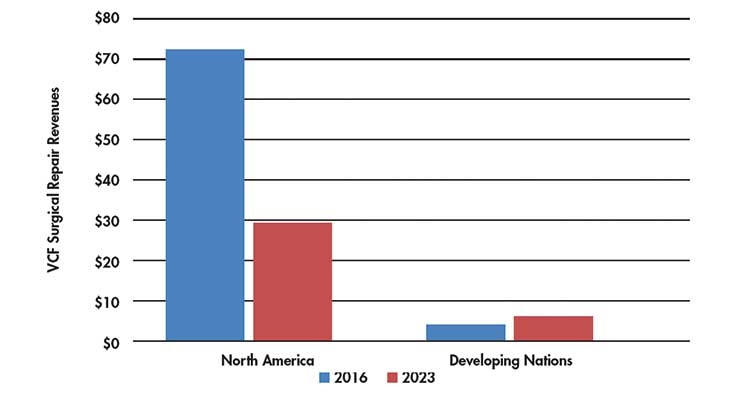
Table 3: Estimated regional revenues of VCF procedures12
In the 2010 article in ODT—”Update on the Controversy in Vertebroplasty”13—I predicted the effort to restore M.D. confidence in vertebroplasty would require a significant investment in R&D, clinical studies, and sales and marketing at companies that provide devices for VCF. Reports stated the commercial impact of the two NEJM studies continued throughout 2009 and U.S. VCF device sales generated only $75 million in 1Q10.14
The Medi-Vantage Perspective
The VCF surgical repair market is still one with a vast base of underserved patients. Even with the setbacks from the NEJM studies, there is still high opportunity for innovators in orthopedic surgical repair products. In our surgeon interviews, we hear surgical skill has a significant impact on VCF surgical outcomes. This means the opportunity to improve devices, based on surgeon feedback, is readily available.
References
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.
The medical device business impact of the studies was rapid and damaging. But what was the impact of the studies over the long term? One report3 published in 2010 stated vertebroplasty, kyphoplasty, and vertebral augmentation procedure rates declined by 10 percent after the NEJM publications in August 2009. There was much concern among surgeons and referring physicians that the procedure rate drop was due to a substantial decrease in patient referrals.
Why This Is Important
In 2009, worldwide estimates placed the number of patients suffering from VCF at approximately 1.4 million.4,5 Also in 2009, in the U.S., it was reported that out of the 750,000 cases of VCF diagnosed annually, approximately one third (250,000 patients) received treatment.5,6 Most vertebral compression fractures resolve over time with conservative treatment (e.g., bed rest, braces, pain medications). However, the significant segment of patients with VCF that do not resolve and improve continue to report chronic pain. These are the patients that are referred for a surgical procedure.
In 2017, the worldwide vertebral compression fracture medical device market was estimated at $2.381 billion and is forecasted to grow to $4.876 billion by 20267. This is a compound annual growth rate (CAGR) of 8.25 percent (Table 1).

Table 1: Estimated global revenues from VCF surgical procedures7
Key market drivers are an aging population, reimbursement rates for minimally invasive surgical procedures for VCF repair, unhealthy lifestyles (e.g., obesity or smoking), and an increase in the popularity of kyphoplasty for the treatment of VCF.
Vertebroplasty vs. Kyphoplasty—Which Procedure Will Win?
In 2007, it was estimated there were more than 140,000 percutaneous VCF procedures and predictions had this number rising to over 180,000 by 20098—a CAGR of 13 percent. Then the publication of the outcomes of the two randomized controlled trials led to an editorial in the Journal9 reviewing the concerns of insurers and questioned whether they should limit or completely withdraw coverage for vertebroplasty procedures. Since then, Blue Cross Blue Shield announced that vertebroplasty and kyphoplasty did not meet the conditions of its efficacy criteria.10
The consequences of the NEJM studies focused high scrutiny on vertebroplasty and kyphoplasty procedures. The article also referenced VCF surgical complication rates (Table 2).11

Table 2: Complication rates of VCF procedures11
The metanalysis contained 168 studies that met the study inclusion criteria. There was a statistically significant difference in improvement with vertebroplasty vs. kyphoplasty (p<0.001). New fracture risk was 17.9 percent with vertebroplasty vs. 14.1 percent with kyphoplasty (p<0.01). Cement leak risk was 19.7 percent with vertebroplasty versus 7 percent with kyphoplasty (p<0.001).11
That Was 2009—Where Are We 10 Years Later?
In a market research report, it was stated the 2016 North American vertebroplasty market (i.e., Canada, Mexico, U.S.) will drop from a forecasted $72 million to approximately $29.4 million by 2023.12
This report states that vertebroplasty, developed as a minimally invasive procedure to be performed in an outpatient setting for the treatment of VCF, has highly variable outcomes and definitive evidence demonstrating procedural efficacy does not exist. It predicted the North America market will see declining rates of adoption in favor of kyphoplasty.
The price tags of vertebroplasty and kyphoplasty are different enough to warrant increased adoption in developing and price sensitive markets. The report stated vertebroplasty revenues in the Middle East and Africa, (i.e., Egypt, Israel, Saudi Arabia, South Africa, and UAE) will increase from $4.1 million in 2016 to approximately $6.5 million by 2023 (Table 3).12

Table 3: Estimated regional revenues of VCF procedures12
In the 2010 article in ODT—”Update on the Controversy in Vertebroplasty”13—I predicted the effort to restore M.D. confidence in vertebroplasty would require a significant investment in R&D, clinical studies, and sales and marketing at companies that provide devices for VCF. Reports stated the commercial impact of the two NEJM studies continued throughout 2009 and U.S. VCF device sales generated only $75 million in 1Q10.14
The Medi-Vantage Perspective
The VCF surgical repair market is still one with a vast base of underserved patients. Even with the setbacks from the NEJM studies, there is still high opportunity for innovators in orthopedic surgical repair products. In our surgeon interviews, we hear surgical skill has a significant impact on VCF surgical outcomes. This means the opportunity to improve devices, based on surgeon feedback, is readily available.
References
- Kalmes, et.al, A randomized trial of Vertebroplasty for osteoporotic spinal fractures, NEJM, 2009; 361:569-579.
- Buchbinder, et. al, A randomized trial of Vertebroplasty for painful osteoporotic vertebral fractures, NEJM, 2009; 557-568.
- http://bit.ly/odt190301
- Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporosis Int 2006;17:1726-33.
- Barnard, Karen, et al. “Recent Clinical Trials in Osteoporosis: A Firm Foundation or Falling Short?” PLoS One, vol. 11, no. 5, Public Library of Science, May 2016, p. e0156068.
- Carmona RH, Office of the Surgeon General. Bone health and osteoporosis: a report of the Surgeon General. Rockville, MD: Department of Health and Human Services, 2004.
- http://bit.ly/odt190302
- http://bit.ly/odt190303
- Perspective, Elshug, et. al, N ENGL J MED 364;15.
- http://bit.ly/odt190304
- http://bit.ly/odt190305
- http://bit.ly/odt190306
- http://bit.ly/odt190307
- MRG, Press Release May, 6, 2010.
Maria Shepherd has more than 20 years of leadership experience in medical device/life science marketing in small startups and top-tier companies. After her industry career, including her role as vice president of marketing for Oridion Medical, where she boosted the company valuation prior to its acquisition by Covidien/Medtronic, director of marketing for Philips Medical, and senior management roles at Boston Scientific Corp., she founded Medi-Vantage. Medi-Vantage provides marketing, business strategy, and innovation research for the medical device, diagnostic, and digital health industries. The firm quantitatively and qualitatively sizes and segments opportunities, evaluates new technologies, provides marketing services, and assesses prospective acquisitions. Shepherd has taught marketing and product development courses and is a member of the Aligo Medtech Investment Committee (www.aligo.com). She can be reached at 855-343-3100. Visit her website at www.medi-vantage.com.