Sam Brusco, Sean Fenske, and Michael Barbella08.14.19
ODT’s Top 10 list chronicles the orthopedic technology companies that have amassed the highest annual revenue. They aren’t the sole source for interesting and innovative technologies, however—many novel orthopedic solutions emerge from smaller firms flying under the radar. These organizations are integral to improving orthopedic care and have piqued the ODT editors’ interests.
CyMedica Orthopedics & Kaia Health
Sam Brusco • Associate Editor
Orthopedics has traditionally been a data-driven discipline. The American Academy of Orthopaedic Surgeons (AAOS), for example, founded the American Joint Replacement Registry (AJRR) in 2009 as a national center to collect hip and knee replacement data. According to AAOS, the AJRR’s database contains over 1.5 million procedures. By collecting and reporting procedural data, AJRR offers actionable information to guide physician and patient decision making, empowering healthcare organizations to enhance the patient experience and reduce complications and revision rates. AJRR also helps device manufacturers strengthen postmarket surveillance.
A digital health strategy can work wonders for the data-driven orthopedic field. Digital health technologies engage information and communication technologies to improve healthcare delivery’s efficiency, which is integral in transitioning to value-based care. Hardware and software both play a role in a digital health device, leveraging telemedicine, web-based analysis, e-mail, smartphones and apps, text messages, wearable devices, and clinic or remote monitoring sensors. New computing capabilities and learning systems emerge all the time, enabling much greater capacity to use information to generate new insights, implement predictive models, and create adaptive solutions for musculoskeletal care.
Knee-Jerk Reaction
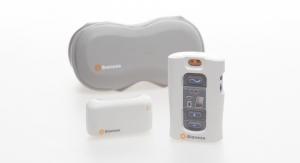
Scottsdale, Ariz.-based CyMedica Orthopedics begins its description with “We are here to support value-based care.” Founded in 2013 by a prominent orthopedic surgeon and several medical device entrepreneurs, the company targets knee surgery rehabilitation. CyMedica asserts patient engagement outside the office is key to successful value-based care implementation—specifically by making rehab more personalized, accessible, and easier for patients.
e-vive, CyMedica’s app-controlled, data-driven muscle activation therapy and patient engagement solution, won FDA clearance in January 2017. The tele-rehab solution for ACL and total knee replacement surgery patients consists of three components. A sensor-laden conductive garment captures and transmits range of motion data and steps while holding electrodes in place to deliver effective quadriceps activation via neuromuscular electrical stimulation (NMES). A controller monitors and controls the muscle stimulation. The e-vive app, operable on any smartphone, collects data points and sends them to the cloud for physicians or physical therapists to view and analyze. Treatment is then adjusted (or kept the same) based on clinician review.
The company presented successful trial results for e-vive at this year’s AAOS meeting. According to Cleveland Clinic researchers, patients receiving NMES therapy with e-vive during the trial returned to function earlier and reported increased quadriceps strength overall as compared to those who had received the standard of care. Those patients also reported a 12.9 percent rise in quadriceps strength at three weeks post-op—a period where patients typically experience a significant loss of quadriceps strength.
CyMedica launched a 120-patient trial evaluating e-vive for knee osteoarthritis (OA) at Northwell Health in June. e-vive data will be assessed in combination with activity and health data collected by commercially available smartwatches to determine how the technology can improve OA symptoms.
“It comes at a time when an overwhelming number of knee arthritis patients are in urgent need of non-narcotic, non-addictive, and home-based wearable therapies to help the millions of Americans who suffer from this painful chronic condition,” commented Rob Morocco, CyMedica’s president and CEO.
Mind-Body App-roach to Back Pain
Lower back pain is one of the chief causes of disability and can be expensive to treat. According to a Journal of Bone and Joint Surgery study, U.S. low back pain treatment costs top $100 billion annually. Back pain is also one of the most common reasons for missed work, accounting for over 264 million lost work days each year. Chronic back pain patients also risk a dangerous dependence on opioid painkillers.
Kaia Health, a digital therapeutics startup formed in 2015, developed an app-based approach to chronic pain treatment. The app leverages mobile technology to deliver multimodal, “mind-body therapy” for musculoskeletal disorders. The therapy includes guided physical exercises, psychological techniques, and on-demand medical education.
The company’s personal trainer app uses computer vision technology to become a personal exercise coach. It visually monitors the user via the smartphone’s camera, keeping track of repetitions and offering vocal feedback to correct posture and motion. It provides a more accessible, less expensive alternative to one-on-one, in-person physiotherapy—someone suffering chronic musculoskeletal pain might need weeks of guided treatment at a dedicated pain center. Kaia aims to democratize access to proven multimodal treatments and decrease pharmaceutical reliance.
“Most treatments against chronic back pain are just crazy expensive and crazy ineffective. Which is a weird combination,” Kaia Health co-founder and CEO Konstantin Mehl told TechCrunch in January. “There [are] a lot of people out there who don’t know how to cope with their pain.”
Results from the company’s first randomized controlled trial—published in NPJ Digital Medicine in May—showed patients using the Kaia app for back pain reported significantly lower pain levels than those treated with physiotherapy and online education. “This study can help add to the growing body of evidence that generally supports the use of multimodal treatments for chronic conditions,” said Thomas R. Tölle, M.D., Ph.D., head of Pain Medicine, Technical University Munich, Germany.
Kaia’s primary focus so far has been back pain, but the company also offers an app for multimodal pulmonary rehab for chronic obstructive pulmonary disorder (COPD) patients.
Reflexion Health & IlluminOss Medical
Sean Fenske • Editor-in-Chief
The orthopedic industry is typically viewed as mature and well-established. The use of metal implants to treat problems with bones has remained relatively unchanged for decades. In more recent years, however, more and more companies are introducing products that don’t resemble those that came before. These technologies are disrupting “tried and true” care protocols and offering what could be more effective alternatives. In some cases, they feature innovations from other industries that are successfully incorporated into the orthopedic offering to enhance the level of care provided.
Digital Motivation
One such example of an orthopedic technology solution provider enabling a better level of care for patients using “outside” innovations would be Reflexion Health. The firm offers VERA, which stands for virtual exercise rehabilitation assistant. Leveraging 3D motion-capture technology, telehealth, and a game-like interface with an avatar, users receive coaching and encouragement as they progress through exercises and education both pre- and post-procedure. The experience could be compared to that of a child engaging with the Nintendo Wii gaming system where actions on-screen are the result of the player’s bodily movements (although the actual VERA system leverages technology from Microsoft, not Nintendo).
“Incorporating game mechanics in our rehabilitation applications presents our patients with an experience that they will crave sufficiently to justify the investment of the patient in the application,” Sudipto Sur, chief technology officer at Reflexion Health, explained in an article he authored for Medical Product Outsourcing. “When it is known that there is a finite term to the engagement, we must question what the means available are to drive the patient to feel that the investment has a payoff. Successful games often target addictiveness, but in our scenario, a successful experience should result in faster recovery and therefore a shorter term of engagement.”
Reflexion Health offers its technology in different formats, based on the intended user. The VERAHome, which is the technology used by the patient, provides the convenience of not requiring travel, which could be problematic for someone recovering from a surgical procedure. It records the exercises performed, measuring up to 26 joints and limbs in motion as much as 30 times per second. Further, it provides audio and visual feedback on the movements and also enables communication with physicians via telehealth.
The VERAClinic is the physician-side component of the system, keeping the clinician informed on their patient’s progress. Through the solution, a doctor can monitor a patient’s adherence to their physical therapy regimen, create personalized therapy plans, administer functional tests, and generate reports.
The company gained its 510(k) clearance back in late 2015 for the rehabilitation of the lower extremities (i.e., knee and hip replacement surgery), but just recently obtained another 510(k) for the system to be used for “rehabilitating the trunk and upper extremities.”
Further adding to the credibility of the solution, representatives from both Duke Clinical Research Institute and Yale New Haven Health presented their study findings during the 2019 American Academy of Orthopedic Surgeons conference. Dr. Janet Prvu Bettger from Duke found VERA to be “as safe and effective as traditional physical therapy, while saving $2,745 per patient in a bundled-payment setting.” Dr. Mary O’Connor shared the findings of Yale New Haven Hospital’s Center for Musculoskeletal Care experience; she concluded the VERA platform provided “world class” patient satisfaction.
Inside-Out Repair
Trauma that results in a bone fracture is typically the outcome of an unfortunate experience for the victim. Regrettably, the procedures and devices utilized to help restore the bone after the break can be, in some cases, even more burdensome and only prolong the discomfort one feels. If bone screws, plates, or a rod is used, the recovery period may be increased or affect the patient for years.
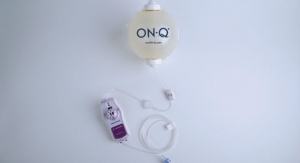
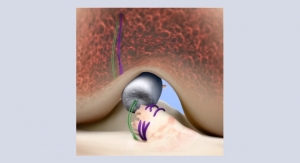
Perhaps recognizing the opportunity to disrupt this paradigm, the founders behind IlluminOss Medical developed a proprietary system that offered an alternative to the traditional method of orthopedic fracture repair. In basic terms, the solution involves the combination of a balloon angioplasty catheter and a light-cured liquid monomer (similar to dental cement). The catheter is inserted into the intramedullary canal of the fractured bone in a minimally invasive procedure. The monomer is then transferred into the balloon, which expands to fill the cavity and conform to the walls of the canal. Once the process is complete, the light source is employed, curing the monomer-infused balloon. The cured implant provides longitudinal strength and rotational stability as the bone heals.
“The IlluminOss System was developed with an aim to provide improved patient experiences and outcomes when treating pathologic fractures. There is a critical need to make less invasive orthopedic fracture repair options available to an aging and underserved market segment,” Robert Rabiner, chief technology officer of IlluminOss, said in a statement following the company’s announcement that it had obtained the first de novo clearance by the FDA’s orthopedic branch.
The device actually has a pair of clearances for its use in the United States. The first was the aforementioned de novo, which was obtained in late 2017 for “treatment of impending and actual pathological fractures of the humerus, radius and ulna from metastatic bone disease.”
The additional clearance was granted in August 2018 for use in “skeletally mature patients in the treatment of traumatic, fragility, pathological, and impending pathological fractures of the humerus, radius and ulna.” The product also has a CE mark for use in Europe, and internationally it’s been in use since 2010.
Emovi & MIACH Orthopaedics Inc.
Michael Barbella • Managing Editor
Despite all the advances made in modern medicine, there’s still a good bit of mystery surrounding the human knee.
Consider, for example, the kneecap. Also known as the patella, this bone plays a vital role in joint flexing and other motions requiring leg movement. Yet scientists are still baffled by its evolutionary history. “We know almost nothing about what the kneecap did when it first evolved, when both the tendons that held the bone and the bone itself were thinner and not as well-developed,” paleontologist Brian Switek wrote in his recent book, “Skeleton Keys.”
Scientists are equally bewildered by the fabella—a bean-shaped bone that is becoming more common in homo sapiens. A 2019 Imperial College London study found that nearly four in 10 people—almost four times the rate a century ago—now have the extra bone, tucked into the knee’s tendons. The study results, which spanned more than 21,000 knee analyses over 150 years, showed the rate of fabellae occurrence in humans more than tripled between 1918 and 2018.
“We don’t know what the fabella’s function is—nobody has ever looked into it!” Michael Berthaume, study lead author, said in a statement. “The fabella may behave like other sesamoid bones to help reduce friction within tendons, redirecting muscle forces, or, as in the case of the kneecap, increasing the mechanical force of that muscle. Or it could be doing nothing at all.”
Neither the kneecap nor the fabella, however, are perhaps as much of an enigma as the anterior cruciate ligament (ACL), the only knee tissue that lacks regenerative and healing powers. Researchers have tried for years to induce knee cartilage regrowth, to no avail. Some experts believe cartilage deterioration prevention measures are the better way to go.
“Once you have damage to the cartilage,” notes cartilage biologist Richard Loeser of UNC School of Medicine-Chapel Hill, “it’s not going to repair itself.”
Probably not. Which explains researchers’ steadfast hunt for the right repair tool.
Treating the Cause, Not the Symptoms
Add knee pain to the roster of the joint’s many mysteries. The precise cause of discomfort in the body’s largest joint has historically been difficult to determine due to the nature of assessments.
Clinicians typically diagnose knee pain based on observation, manual testing, imaging (X-ray, MRIs), and patient history. These assessments routinely are conducted while the joint is in a static standing or lying down position rather than in motion (when the pain is greatest).
Montreal-based Emovi is attempting to improve knee pain diagnostics with a 3D assessment tool that specifically examines joint function. The innovation was developed by research teams from three Canadian universities.
The company’s KneeKG uses mechanical markers and functional deficiencies to give clinicians a glimpse of the knee’s inner workings. CEO Michelle Laflamme likens the device to an electrocardiogram, saying it allows medical professionals to “understand the cause of symptoms patients are experiencing. That insight can be used to personalize therapy. Clinicians can know exactly what can be done.”
The 20-minute test is performed in a sports medicine clinic, hospital physical therapy department, or a physical therapy clinic. Cleared by the U.S. Food and Drug Administration and commercially available in the United States, the KneeKG is suitable for people suffering from general knee pain, knee osteoarthritis, ligament injuries, and anterior knee pain/patellofemoral pain syndrome.
Emovi received a $20 million capital infusion earlier this year that will allow the firm to expand the KneeKG to clinics and hospitals throughout North America. The device also is available in Australia, Canada, China, and the United Kingdom.
A BEAR-able ACL Fix
No clotting.
No kidding—the key to natural ACL repair could really be that simple.
Credit for the discovery goes to orthopedic surgeon Martha Murray, M.D., founder and chief medical officer of Westborough, Mass.-based MIACH Orthopaedics Inc., developer of a bioengineered sponge that simulates healing of torn ACLs.
“We wanted to find a way to encourage the ACL to heal itself,” Murray said earlier this year in touting promising study results for the company’s implant. “We hoped we could find a better way than removing the torn ACL and replacing it with perfectly normal tissue from somewhere else, so we started working on a protein-based scaffold.”
Before the scaffold could be developed, however, researchers had to determine the cause of the ACL’s curative defect. Thus, researchers compared the ACL to self-healing tissue like the medial collateral ligament; they found that in MCL tears, a blood clot forms between the torn tissue ends and acts as a healing “bridge” for the damaged ligament. But no such clot forms in ACL injuries due to the ligament’s joint fluid. Consequently, there is no natural structure for healing.
MIACH Orthopaedic’s Bridge-Enhanced ACL Repair (BEAR) is surgically placed between the ACL’s torn ends and then fed some of the patient’s own blood to facilitate clotting (and eventually form a healing bridge). The method has been tested successfully in nearly 100 patients at Boston Children’s Hospital, and studies have shown the treatment is comparatively as effective as traditional ACL repair surgeries 24 months post-procedure. In addition, the BEAR implant did not spawn any infections or severe inflammatory reactions. Preclinical studies also suggest that BEAR can significantly reduce the rate of arthritis compared to reconstructive surgery.
Last fall, the 3-year-old company closed a $22.5 million round of Series A financing co-led by Amzak Health Investors LLC and DSM Venturing, with participation from the NFL Players Association and additional supporters. The financing went toward funding ongoing clinical trials at Boston Children’s Hospital and Rhode Island Hospital.
CyMedica Orthopedics & Kaia Health
Sam Brusco • Associate Editor
Orthopedics has traditionally been a data-driven discipline. The American Academy of Orthopaedic Surgeons (AAOS), for example, founded the American Joint Replacement Registry (AJRR) in 2009 as a national center to collect hip and knee replacement data. According to AAOS, the AJRR’s database contains over 1.5 million procedures. By collecting and reporting procedural data, AJRR offers actionable information to guide physician and patient decision making, empowering healthcare organizations to enhance the patient experience and reduce complications and revision rates. AJRR also helps device manufacturers strengthen postmarket surveillance.
A digital health strategy can work wonders for the data-driven orthopedic field. Digital health technologies engage information and communication technologies to improve healthcare delivery’s efficiency, which is integral in transitioning to value-based care. Hardware and software both play a role in a digital health device, leveraging telemedicine, web-based analysis, e-mail, smartphones and apps, text messages, wearable devices, and clinic or remote monitoring sensors. New computing capabilities and learning systems emerge all the time, enabling much greater capacity to use information to generate new insights, implement predictive models, and create adaptive solutions for musculoskeletal care.
Knee-Jerk Reaction
Scottsdale, Ariz.-based CyMedica Orthopedics begins its description with “We are here to support value-based care.” Founded in 2013 by a prominent orthopedic surgeon and several medical device entrepreneurs, the company targets knee surgery rehabilitation. CyMedica asserts patient engagement outside the office is key to successful value-based care implementation—specifically by making rehab more personalized, accessible, and easier for patients.
e-vive, CyMedica’s app-controlled, data-driven muscle activation therapy and patient engagement solution, won FDA clearance in January 2017. The tele-rehab solution for ACL and total knee replacement surgery patients consists of three components. A sensor-laden conductive garment captures and transmits range of motion data and steps while holding electrodes in place to deliver effective quadriceps activation via neuromuscular electrical stimulation (NMES). A controller monitors and controls the muscle stimulation. The e-vive app, operable on any smartphone, collects data points and sends them to the cloud for physicians or physical therapists to view and analyze. Treatment is then adjusted (or kept the same) based on clinician review.
The company presented successful trial results for e-vive at this year’s AAOS meeting. According to Cleveland Clinic researchers, patients receiving NMES therapy with e-vive during the trial returned to function earlier and reported increased quadriceps strength overall as compared to those who had received the standard of care. Those patients also reported a 12.9 percent rise in quadriceps strength at three weeks post-op—a period where patients typically experience a significant loss of quadriceps strength.
CyMedica launched a 120-patient trial evaluating e-vive for knee osteoarthritis (OA) at Northwell Health in June. e-vive data will be assessed in combination with activity and health data collected by commercially available smartwatches to determine how the technology can improve OA symptoms.
“It comes at a time when an overwhelming number of knee arthritis patients are in urgent need of non-narcotic, non-addictive, and home-based wearable therapies to help the millions of Americans who suffer from this painful chronic condition,” commented Rob Morocco, CyMedica’s president and CEO.
Mind-Body App-roach to Back Pain
Lower back pain is one of the chief causes of disability and can be expensive to treat. According to a Journal of Bone and Joint Surgery study, U.S. low back pain treatment costs top $100 billion annually. Back pain is also one of the most common reasons for missed work, accounting for over 264 million lost work days each year. Chronic back pain patients also risk a dangerous dependence on opioid painkillers.
Kaia Health, a digital therapeutics startup formed in 2015, developed an app-based approach to chronic pain treatment. The app leverages mobile technology to deliver multimodal, “mind-body therapy” for musculoskeletal disorders. The therapy includes guided physical exercises, psychological techniques, and on-demand medical education.
The company’s personal trainer app uses computer vision technology to become a personal exercise coach. It visually monitors the user via the smartphone’s camera, keeping track of repetitions and offering vocal feedback to correct posture and motion. It provides a more accessible, less expensive alternative to one-on-one, in-person physiotherapy—someone suffering chronic musculoskeletal pain might need weeks of guided treatment at a dedicated pain center. Kaia aims to democratize access to proven multimodal treatments and decrease pharmaceutical reliance.
“Most treatments against chronic back pain are just crazy expensive and crazy ineffective. Which is a weird combination,” Kaia Health co-founder and CEO Konstantin Mehl told TechCrunch in January. “There [are] a lot of people out there who don’t know how to cope with their pain.”
Results from the company’s first randomized controlled trial—published in NPJ Digital Medicine in May—showed patients using the Kaia app for back pain reported significantly lower pain levels than those treated with physiotherapy and online education. “This study can help add to the growing body of evidence that generally supports the use of multimodal treatments for chronic conditions,” said Thomas R. Tölle, M.D., Ph.D., head of Pain Medicine, Technical University Munich, Germany.
Kaia’s primary focus so far has been back pain, but the company also offers an app for multimodal pulmonary rehab for chronic obstructive pulmonary disorder (COPD) patients.
Reflexion Health & IlluminOss Medical
Sean Fenske • Editor-in-Chief
The orthopedic industry is typically viewed as mature and well-established. The use of metal implants to treat problems with bones has remained relatively unchanged for decades. In more recent years, however, more and more companies are introducing products that don’t resemble those that came before. These technologies are disrupting “tried and true” care protocols and offering what could be more effective alternatives. In some cases, they feature innovations from other industries that are successfully incorporated into the orthopedic offering to enhance the level of care provided.
Digital Motivation
One such example of an orthopedic technology solution provider enabling a better level of care for patients using “outside” innovations would be Reflexion Health. The firm offers VERA, which stands for virtual exercise rehabilitation assistant. Leveraging 3D motion-capture technology, telehealth, and a game-like interface with an avatar, users receive coaching and encouragement as they progress through exercises and education both pre- and post-procedure. The experience could be compared to that of a child engaging with the Nintendo Wii gaming system where actions on-screen are the result of the player’s bodily movements (although the actual VERA system leverages technology from Microsoft, not Nintendo).
“Incorporating game mechanics in our rehabilitation applications presents our patients with an experience that they will crave sufficiently to justify the investment of the patient in the application,” Sudipto Sur, chief technology officer at Reflexion Health, explained in an article he authored for Medical Product Outsourcing. “When it is known that there is a finite term to the engagement, we must question what the means available are to drive the patient to feel that the investment has a payoff. Successful games often target addictiveness, but in our scenario, a successful experience should result in faster recovery and therefore a shorter term of engagement.”
Reflexion Health offers its technology in different formats, based on the intended user. The VERAHome, which is the technology used by the patient, provides the convenience of not requiring travel, which could be problematic for someone recovering from a surgical procedure. It records the exercises performed, measuring up to 26 joints and limbs in motion as much as 30 times per second. Further, it provides audio and visual feedback on the movements and also enables communication with physicians via telehealth.
The VERAClinic is the physician-side component of the system, keeping the clinician informed on their patient’s progress. Through the solution, a doctor can monitor a patient’s adherence to their physical therapy regimen, create personalized therapy plans, administer functional tests, and generate reports.
The company gained its 510(k) clearance back in late 2015 for the rehabilitation of the lower extremities (i.e., knee and hip replacement surgery), but just recently obtained another 510(k) for the system to be used for “rehabilitating the trunk and upper extremities.”
Further adding to the credibility of the solution, representatives from both Duke Clinical Research Institute and Yale New Haven Health presented their study findings during the 2019 American Academy of Orthopedic Surgeons conference. Dr. Janet Prvu Bettger from Duke found VERA to be “as safe and effective as traditional physical therapy, while saving $2,745 per patient in a bundled-payment setting.” Dr. Mary O’Connor shared the findings of Yale New Haven Hospital’s Center for Musculoskeletal Care experience; she concluded the VERA platform provided “world class” patient satisfaction.
Inside-Out Repair
Trauma that results in a bone fracture is typically the outcome of an unfortunate experience for the victim. Regrettably, the procedures and devices utilized to help restore the bone after the break can be, in some cases, even more burdensome and only prolong the discomfort one feels. If bone screws, plates, or a rod is used, the recovery period may be increased or affect the patient for years.
Perhaps recognizing the opportunity to disrupt this paradigm, the founders behind IlluminOss Medical developed a proprietary system that offered an alternative to the traditional method of orthopedic fracture repair. In basic terms, the solution involves the combination of a balloon angioplasty catheter and a light-cured liquid monomer (similar to dental cement). The catheter is inserted into the intramedullary canal of the fractured bone in a minimally invasive procedure. The monomer is then transferred into the balloon, which expands to fill the cavity and conform to the walls of the canal. Once the process is complete, the light source is employed, curing the monomer-infused balloon. The cured implant provides longitudinal strength and rotational stability as the bone heals.
“The IlluminOss System was developed with an aim to provide improved patient experiences and outcomes when treating pathologic fractures. There is a critical need to make less invasive orthopedic fracture repair options available to an aging and underserved market segment,” Robert Rabiner, chief technology officer of IlluminOss, said in a statement following the company’s announcement that it had obtained the first de novo clearance by the FDA’s orthopedic branch.
The device actually has a pair of clearances for its use in the United States. The first was the aforementioned de novo, which was obtained in late 2017 for “treatment of impending and actual pathological fractures of the humerus, radius and ulna from metastatic bone disease.”
The additional clearance was granted in August 2018 for use in “skeletally mature patients in the treatment of traumatic, fragility, pathological, and impending pathological fractures of the humerus, radius and ulna.” The product also has a CE mark for use in Europe, and internationally it’s been in use since 2010.
Emovi & MIACH Orthopaedics Inc.
Michael Barbella • Managing Editor
Despite all the advances made in modern medicine, there’s still a good bit of mystery surrounding the human knee.
Consider, for example, the kneecap. Also known as the patella, this bone plays a vital role in joint flexing and other motions requiring leg movement. Yet scientists are still baffled by its evolutionary history. “We know almost nothing about what the kneecap did when it first evolved, when both the tendons that held the bone and the bone itself were thinner and not as well-developed,” paleontologist Brian Switek wrote in his recent book, “Skeleton Keys.”
Scientists are equally bewildered by the fabella—a bean-shaped bone that is becoming more common in homo sapiens. A 2019 Imperial College London study found that nearly four in 10 people—almost four times the rate a century ago—now have the extra bone, tucked into the knee’s tendons. The study results, which spanned more than 21,000 knee analyses over 150 years, showed the rate of fabellae occurrence in humans more than tripled between 1918 and 2018.
“We don’t know what the fabella’s function is—nobody has ever looked into it!” Michael Berthaume, study lead author, said in a statement. “The fabella may behave like other sesamoid bones to help reduce friction within tendons, redirecting muscle forces, or, as in the case of the kneecap, increasing the mechanical force of that muscle. Or it could be doing nothing at all.”
Neither the kneecap nor the fabella, however, are perhaps as much of an enigma as the anterior cruciate ligament (ACL), the only knee tissue that lacks regenerative and healing powers. Researchers have tried for years to induce knee cartilage regrowth, to no avail. Some experts believe cartilage deterioration prevention measures are the better way to go.
“Once you have damage to the cartilage,” notes cartilage biologist Richard Loeser of UNC School of Medicine-Chapel Hill, “it’s not going to repair itself.”
Probably not. Which explains researchers’ steadfast hunt for the right repair tool.
Treating the Cause, Not the Symptoms
Add knee pain to the roster of the joint’s many mysteries. The precise cause of discomfort in the body’s largest joint has historically been difficult to determine due to the nature of assessments.
Clinicians typically diagnose knee pain based on observation, manual testing, imaging (X-ray, MRIs), and patient history. These assessments routinely are conducted while the joint is in a static standing or lying down position rather than in motion (when the pain is greatest).
Montreal-based Emovi is attempting to improve knee pain diagnostics with a 3D assessment tool that specifically examines joint function. The innovation was developed by research teams from three Canadian universities.
The company’s KneeKG uses mechanical markers and functional deficiencies to give clinicians a glimpse of the knee’s inner workings. CEO Michelle Laflamme likens the device to an electrocardiogram, saying it allows medical professionals to “understand the cause of symptoms patients are experiencing. That insight can be used to personalize therapy. Clinicians can know exactly what can be done.”
The 20-minute test is performed in a sports medicine clinic, hospital physical therapy department, or a physical therapy clinic. Cleared by the U.S. Food and Drug Administration and commercially available in the United States, the KneeKG is suitable for people suffering from general knee pain, knee osteoarthritis, ligament injuries, and anterior knee pain/patellofemoral pain syndrome.
Emovi received a $20 million capital infusion earlier this year that will allow the firm to expand the KneeKG to clinics and hospitals throughout North America. The device also is available in Australia, Canada, China, and the United Kingdom.
A BEAR-able ACL Fix
No clotting.
No kidding—the key to natural ACL repair could really be that simple.
Credit for the discovery goes to orthopedic surgeon Martha Murray, M.D., founder and chief medical officer of Westborough, Mass.-based MIACH Orthopaedics Inc., developer of a bioengineered sponge that simulates healing of torn ACLs.
“We wanted to find a way to encourage the ACL to heal itself,” Murray said earlier this year in touting promising study results for the company’s implant. “We hoped we could find a better way than removing the torn ACL and replacing it with perfectly normal tissue from somewhere else, so we started working on a protein-based scaffold.”
Before the scaffold could be developed, however, researchers had to determine the cause of the ACL’s curative defect. Thus, researchers compared the ACL to self-healing tissue like the medial collateral ligament; they found that in MCL tears, a blood clot forms between the torn tissue ends and acts as a healing “bridge” for the damaged ligament. But no such clot forms in ACL injuries due to the ligament’s joint fluid. Consequently, there is no natural structure for healing.
MIACH Orthopaedic’s Bridge-Enhanced ACL Repair (BEAR) is surgically placed between the ACL’s torn ends and then fed some of the patient’s own blood to facilitate clotting (and eventually form a healing bridge). The method has been tested successfully in nearly 100 patients at Boston Children’s Hospital, and studies have shown the treatment is comparatively as effective as traditional ACL repair surgeries 24 months post-procedure. In addition, the BEAR implant did not spawn any infections or severe inflammatory reactions. Preclinical studies also suggest that BEAR can significantly reduce the rate of arthritis compared to reconstructive surgery.
Last fall, the 3-year-old company closed a $22.5 million round of Series A financing co-led by Amzak Health Investors LLC and DSM Venturing, with participation from the NFL Players Association and additional supporters. The financing went toward funding ongoing clinical trials at Boston Children’s Hospital and Rhode Island Hospital.