Victoria Trafka, BSME, MSME, President and Lead Engineer, Engineering & Quality Solutions Inc. 12.02.19
After many years in the orthopedic industry, I’ve run across the same product development professionals many times at different companies or in different roles, and thought, “What a small world.” At orthopedic conferences and ASTM meetings, as well as on LinkedIn, I see people who have moved to a different company or transitioned within their current company to a new position. What’s unusual is the product development people tend to stay within the orthopedic and spine industry and not cross over into other medical device technology sectors, even though there are countless other types of products to be designed and improved. In contrast, I’ve known numerous individuals who worked on other medical products, such as catheters, fluid management systems, dialysis machines, wound care devices, etc., and they seem to move to different industry segments easily. This got me thinking about why the orthopedic sector is different. I concluded orthopedic devices are vastly different from most medical technologies, and, therefore, the task of developing the products is unique. An orthopedic product’s function is complex, strength and reliability are critical, and the user is demanding. Further, the people up to the task of developing these products are similarly unique. Following are five distinct ways in which orthopedic product development is different from virtually all other medical device development.
1. Permanent Implants
Many orthopedic products are implanted into the human body—quite a few for the remainder of the patient’s lifetime—while most other healthcare technologies are not. The majority of medical devices are used on the patient or in the patient only temporarily (e.g., surgical instruments, IVs, diagnostic machines, gloves, crutches, etc.). While these products interact with patients and medical professionals, they remain outside the body in ambient conditions where they can be observed, manipulated, and controlled. In contrast, most orthopedic and spine devices are implanted during a surgical procedure and then left to perform their intended function within the body. Therefore, the implants need to meet strict requirements for biocompatibility, reliability, and safety, and the product’s expected lifetime can be much longer than the average medical device. They may be expected to function for dozens of years and endure countless of cycles of use, while existing in a somewhat corrosive environment. The devices, their condition, and their function generally can’t be observed without X-rays, imaging scans, or additional surgery. These factors make the task of designing these products much more challenging and specialized, and a higher risk compared to many other medical devices.
2. Unparalleled Strength Within a Minimal Size
Orthopedic and spine products are used to repair and support the musculoskeletal system, which is unlike any other medical device. They need to support the weight of the human body and enable muscles and tendons to function properly, while still maintaining as many degrees of natural motion as possible. As such, the products must offer enhanced strength, longevity, and function. The average medical device doesn’t need to perform this much heavy lifting (literally). In order to verify these requirements can be met, orthopedic and spine products have to be rigorously tested for mechanical strength and function; comprehending this testing is a specialty in itself. Product development engineers absolutely need to understand the testing and device indications in order to design a product that will ultimately pass those tests as well as function as required in the body. Further, a very common product criteria is for a smaller, lightweight device that will be more comfortable for the patient. Compare these challenges to the previously mentioned devices—the frame of the X-ray machine can be solid and stout; the gloves only need to be used for an hour and are then discarded; the fluid pump and canister can be placed conveniently out of the way. When reviewing the differences in these technologies, it becomes obvious orthopedic products present a unique challenge for designers.
3. Unique Device Application and Treatment
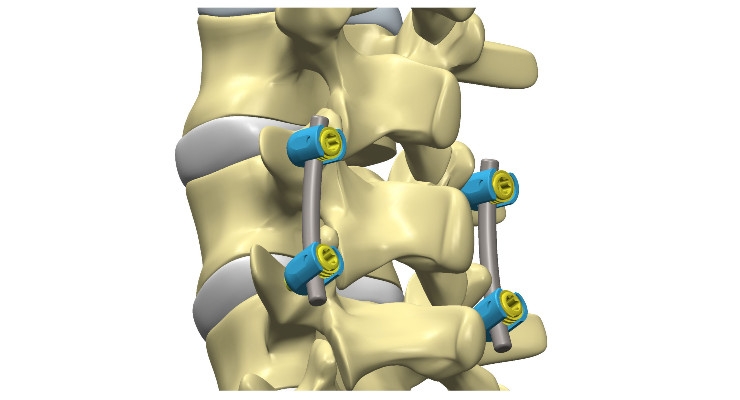
No two patients’ bones or treatment needs are the same, and the skeletal system has very complex geometry. As a result, orthopedic and spine procedures need to be specifically tailored to each patient and the implants must accommodate an incredible range of anatomy and surgical scenarios. This results in product lines requiring dozens of size increments, shape options, adjustments, and ranges of function. Even the product materials may need to be different for a given patient. Additionally, many implants need to be customized during surgery, such as adjusting pedicle screw head angles and bending spinal rods to fit the patient and enable the desired spinal correction. The first time I saw the test set-up for a variable angle pedicle screw construct and thought about that assembly surviving 5 million loading cycles, I was amazed! That’s because the screws have variable head angles to accommodate the patient’s treatment needs, but they also must lock into position and not move for the rest of the patient’s life. To achieve this performance, product development engineers must create designs that are reliable and strong, but also flexible to accommodate the patient. This requires creativity and resourcefulness, a knowledge of the surgical procedures, and an understanding of the product’s intended function within the body. Most other medical devices have a single function, a few sizes, and are not adjustable, making their designs much more straightforward.
4. Specialized Technology
Orthopedics is a specialty industry with unique technology and processes used to produce the products. The materials, coatings, and manufacturing techniques used for orthopedic devices are not often used for most other medical devices. Orthopedic products are commonly made of various implantable materials, such as titanium, vanadium, custom stainless steel, cobalt chrome, nitinol, and molybdenum, and what seems to be an endless array of different alloys. In contrast, other medical devices are frequently made of plastic, stainless steel, and/or aluminum—material and technology experience that can transfer from a product like a syringe to a fluid pump, but not really to orthopedic products. Furthermore, the surface finishes and markings used in orthopedic devices have been custom developed for the last 30 years so they can be manufactured consistently, will last the lifetime of the device, and will minimize patient reactions. The industry has made great advances due to unfortunate problems—such as implants prematurely wearing or fracturing—and no one wants to see a repeat of those issues. Therefore, development engineers must thoroughly understand the applicable materials and processes, which are unlike those used for other medical devices, in order to design products that are manufacturable, functional, and safe.
5. Surgeons and Patients Demand Perfection
Orthopedic and spine surgeons are often perfectionists and can be viewed as high maintenance. They should be. Since every patient and procedure is unique, a successful surgery depends on a combination of superb products and surgical skills. A great surgeon will have a challenging time achieving a great outcome with the wrong sized implant or with an incomplete instrument set. These surgeons have to be both excellent doctors and skilled artists, and they’re also increasingly being measured for efficiency by the hospitals. This combination creates a customer who is not easy to please, which means development teams must be able to listen to surgeons and develop products that meet all of their needs by functioning flawlessly every time. Sometimes, this translates to adding more features or sizes, while other times, it means simplifying function.
Furthermore, orthopedic patients today are equally demanding and expect to return to full physical activity after a procedure. They’re not satisfied with an improvement in their condition; rather, they want to resume skiing, tennis, rock climbing, and all other activities they enjoyed previously. It’s not easy to identify and meet the needs of the surgeon and patient; not many product development engineers have the right mix of skills to accomplish that task. While all medical devices need to consistently work flawlessly, the customer is generally not as demanding as orthopedic device end users.
Given the five major differences of orthopedic design provided, one may wonder why anyone works in the orthopedic or spine product development space rather than working on another medical device. The answer, however, is simple—those who work in the industry “get” orthopedics and love the products they produce. It’s a very satisfying career for someone who has the right set of skills—problem solving, understanding of mechanical engineering and solid mechanics, ability to listen and learn from surgeons, a talent for coming up with elegant solutions to a challenge, and a desire to make products that truly help people. Product development professionals who thrive in the orthopedic industry continue to deliver great products because they embrace the five challenges outlined in this article. As a result, they fervently choose to stay in the industry. As such, we’ll probably continue to think, “What a small world,” whenever starting a new project, attending a conference, or seeing an update on LinkedIn.
Victoria Trafka, BSME, MSME, is president and lead engineer at Engineering & Quality Solutions Inc. Her passion is improving people’s mobility and quality of life with innovative medical devices. Trafka has spent the majority of her career working within the orthopedic trauma and spine industry for a wide variety of companies, from startups to global market leaders. While her current focus is product development and quality systems, she has also held positions in manufacturing, quality, and project management. This varied experience helps her understand every step in the process, from device idea to launch and long-term compliance, which results in a more robust product in less time. Trafka’s skill and contacts throughout the medical device industry help companies proceed on the right path to market.
1. Permanent Implants
Many orthopedic products are implanted into the human body—quite a few for the remainder of the patient’s lifetime—while most other healthcare technologies are not. The majority of medical devices are used on the patient or in the patient only temporarily (e.g., surgical instruments, IVs, diagnostic machines, gloves, crutches, etc.). While these products interact with patients and medical professionals, they remain outside the body in ambient conditions where they can be observed, manipulated, and controlled. In contrast, most orthopedic and spine devices are implanted during a surgical procedure and then left to perform their intended function within the body. Therefore, the implants need to meet strict requirements for biocompatibility, reliability, and safety, and the product’s expected lifetime can be much longer than the average medical device. They may be expected to function for dozens of years and endure countless of cycles of use, while existing in a somewhat corrosive environment. The devices, their condition, and their function generally can’t be observed without X-rays, imaging scans, or additional surgery. These factors make the task of designing these products much more challenging and specialized, and a higher risk compared to many other medical devices.
2. Unparalleled Strength Within a Minimal Size
Orthopedic and spine products are used to repair and support the musculoskeletal system, which is unlike any other medical device. They need to support the weight of the human body and enable muscles and tendons to function properly, while still maintaining as many degrees of natural motion as possible. As such, the products must offer enhanced strength, longevity, and function. The average medical device doesn’t need to perform this much heavy lifting (literally). In order to verify these requirements can be met, orthopedic and spine products have to be rigorously tested for mechanical strength and function; comprehending this testing is a specialty in itself. Product development engineers absolutely need to understand the testing and device indications in order to design a product that will ultimately pass those tests as well as function as required in the body. Further, a very common product criteria is for a smaller, lightweight device that will be more comfortable for the patient. Compare these challenges to the previously mentioned devices—the frame of the X-ray machine can be solid and stout; the gloves only need to be used for an hour and are then discarded; the fluid pump and canister can be placed conveniently out of the way. When reviewing the differences in these technologies, it becomes obvious orthopedic products present a unique challenge for designers.
3. Unique Device Application and Treatment
No two patients’ bones or treatment needs are the same, and the skeletal system has very complex geometry. As a result, orthopedic and spine procedures need to be specifically tailored to each patient and the implants must accommodate an incredible range of anatomy and surgical scenarios. This results in product lines requiring dozens of size increments, shape options, adjustments, and ranges of function. Even the product materials may need to be different for a given patient. Additionally, many implants need to be customized during surgery, such as adjusting pedicle screw head angles and bending spinal rods to fit the patient and enable the desired spinal correction. The first time I saw the test set-up for a variable angle pedicle screw construct and thought about that assembly surviving 5 million loading cycles, I was amazed! That’s because the screws have variable head angles to accommodate the patient’s treatment needs, but they also must lock into position and not move for the rest of the patient’s life. To achieve this performance, product development engineers must create designs that are reliable and strong, but also flexible to accommodate the patient. This requires creativity and resourcefulness, a knowledge of the surgical procedures, and an understanding of the product’s intended function within the body. Most other medical devices have a single function, a few sizes, and are not adjustable, making their designs much more straightforward.
4. Specialized Technology
Orthopedics is a specialty industry with unique technology and processes used to produce the products. The materials, coatings, and manufacturing techniques used for orthopedic devices are not often used for most other medical devices. Orthopedic products are commonly made of various implantable materials, such as titanium, vanadium, custom stainless steel, cobalt chrome, nitinol, and molybdenum, and what seems to be an endless array of different alloys. In contrast, other medical devices are frequently made of plastic, stainless steel, and/or aluminum—material and technology experience that can transfer from a product like a syringe to a fluid pump, but not really to orthopedic products. Furthermore, the surface finishes and markings used in orthopedic devices have been custom developed for the last 30 years so they can be manufactured consistently, will last the lifetime of the device, and will minimize patient reactions. The industry has made great advances due to unfortunate problems—such as implants prematurely wearing or fracturing—and no one wants to see a repeat of those issues. Therefore, development engineers must thoroughly understand the applicable materials and processes, which are unlike those used for other medical devices, in order to design products that are manufacturable, functional, and safe.
5. Surgeons and Patients Demand Perfection
Orthopedic and spine surgeons are often perfectionists and can be viewed as high maintenance. They should be. Since every patient and procedure is unique, a successful surgery depends on a combination of superb products and surgical skills. A great surgeon will have a challenging time achieving a great outcome with the wrong sized implant or with an incomplete instrument set. These surgeons have to be both excellent doctors and skilled artists, and they’re also increasingly being measured for efficiency by the hospitals. This combination creates a customer who is not easy to please, which means development teams must be able to listen to surgeons and develop products that meet all of their needs by functioning flawlessly every time. Sometimes, this translates to adding more features or sizes, while other times, it means simplifying function.
Furthermore, orthopedic patients today are equally demanding and expect to return to full physical activity after a procedure. They’re not satisfied with an improvement in their condition; rather, they want to resume skiing, tennis, rock climbing, and all other activities they enjoyed previously. It’s not easy to identify and meet the needs of the surgeon and patient; not many product development engineers have the right mix of skills to accomplish that task. While all medical devices need to consistently work flawlessly, the customer is generally not as demanding as orthopedic device end users.
Given the five major differences of orthopedic design provided, one may wonder why anyone works in the orthopedic or spine product development space rather than working on another medical device. The answer, however, is simple—those who work in the industry “get” orthopedics and love the products they produce. It’s a very satisfying career for someone who has the right set of skills—problem solving, understanding of mechanical engineering and solid mechanics, ability to listen and learn from surgeons, a talent for coming up with elegant solutions to a challenge, and a desire to make products that truly help people. Product development professionals who thrive in the orthopedic industry continue to deliver great products because they embrace the five challenges outlined in this article. As a result, they fervently choose to stay in the industry. As such, we’ll probably continue to think, “What a small world,” whenever starting a new project, attending a conference, or seeing an update on LinkedIn.
Victoria Trafka, BSME, MSME, is president and lead engineer at Engineering & Quality Solutions Inc. Her passion is improving people’s mobility and quality of life with innovative medical devices. Trafka has spent the majority of her career working within the orthopedic trauma and spine industry for a wide variety of companies, from startups to global market leaders. While her current focus is product development and quality systems, she has also held positions in manufacturing, quality, and project management. This varied experience helps her understand every step in the process, from device idea to launch and long-term compliance, which results in a more robust product in less time. Trafka’s skill and contacts throughout the medical device industry help companies proceed on the right path to market.