Sam Brusco, Associate Editor03.13.20
University of Connecticut’s Laboratory for Innovative Microtechnologies & Biomechanics (LIMB) aims to develop tools and technologies to solve engineering and medical challenges to improve quality of life. To meet this end, LIMB combines advancements in engineering, biology, biomaterials, and medicine. The lab’s research is multidisciplinary, focused on developing micro/nanotechnologies, biomechanics, and advanced manufacturing strategies to engineer smart systems and wearable platforms for health monitoring and chronic wound treatment.
One project, for example, aims to tackle non-healing wounds brought on by diabetes mellitus (Type 2), which, left unchecked, can trigger increased healthcare costs, decreased quality of life, infections, amputations, and death. They’re developing an active wearable or implantable device to monitor and regulate the injury site to facilitate healing.
Another goal for LIMB is to develop advanced bioprinting and biotextile processes to fabricate tissue-like constructs from biological materials. The lab also seeks to combine nanotechnological and bioprinting advances to build biomanufacturing platforms. LIMB recognized one application is treatment of musculoskeletal disorders.
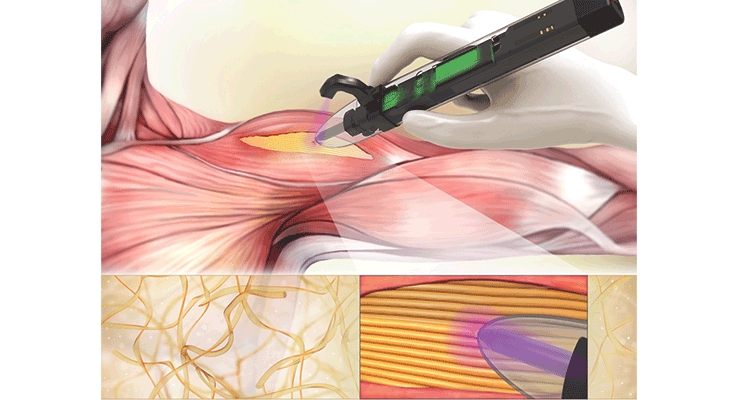
Dr. Ali Tamayol, an associate professor at UConn’s Department of Biomedical Engineering and LIMB’s principal investigator, recently developed a handheld bioprinter pen that could significantly change the way musculoskeletal procedures are performed. The bioprinter enables surgeons to deposit materials to help support cell and tissue growth (scaffolds) directly into defect sites associated with weakened skeletal muscles. This project’s research was published at the end of January in ACS Applied Bio Materials.
“The printer is robust and allows proper filling of the cavity with fibrillar scaffolds in which fibers resemble the architecture of the native tissue,” Dr. Tamayol told the press.
The scaffolds essentially remove the need for suturing because they adhere precisely to the injury’s surrounding tissues and mimic the existing tissue’s properties. According to LIMB, the methods to treat volumetric muscle loss (VML) are inadequate. The geometry of skeletal muscle defects in VML injuries varies with each case. 3D printing emerged as one strategy to fabricate scaffolds matching the geometry of each defect site.
Dr. Indranil Sanha, plastic surgeon at Harvard’s Brigham and Women’s Hospital, who joined Dr. Tamayol in this study, has expertise in treating muscle injuries. Dr. Sinha said a “good solution currently does not exist for patients who suffer volumetric muscle loss. A customizable, printed gel establishes the foundation for a new treatment paradigm can improve the care of our trauma patients.”
The muscle-printing pen extrudes a material called gelatin methacrylol (GelMA), a collagen-like biomaterial designed to mimic skeletal tissue and act as a bioadhesive. Then, a UV light attached to the pen melds the strips of GelMA together, sort of like soldering.
Of course, existing 3D bioprinting technology has its share of issues. Successfully implanting hydrogel-based scaffolds requires a very specific biomaterial to be printed that will adhere to the defect site. 3D bioprinted scaffolds that mimic skeletal muscles have previously been created in vitro, but they haven’t yet been successfully implanted in an actual subject. Further, the time and facilities needed to image the defect site, process to render computer models, and print a suitable scaffold currently prevent immediate reconstructions following traumatic injuries.
Dr. Tamayol’s solutions solve this problem—his bioprinter prints gelatin-based hydrogels, or “bioink.” Based on LIMB’s research, this bioink proved effective in adhering to defect sites of mice with VML injury. The bioink hydrogel was cross-linked in situ, and its adhesiveness to skeletal muscles was assessed ex vivo. The mice demonstrated significant increase in muscle hypertrophy following Dr. Tamayol’s therapy. After 28 days of implantation, the LIMB team did not observe any inflammation or damage to surrounding tissue where the bioink scaffolding had adhered. Through testing, it was also found the scaffold had similar tear and shear strength to natural muscles.
“This is a new generation of 3D printers than enables clinicians to directly print the scaffold within the patient’s body,” said Dr. Tamayol. “Best of all, this system does not require the presence of sophisticated imaging and printing systems.”
While the study was only done on mice and can’t be translated to human subjects, Dr. Tamayol and Dr. Sinha have filed a patent on this technology to treat musculoskeletal injuries. Hopefully, they can move forward with developing future human trials.
One project, for example, aims to tackle non-healing wounds brought on by diabetes mellitus (Type 2), which, left unchecked, can trigger increased healthcare costs, decreased quality of life, infections, amputations, and death. They’re developing an active wearable or implantable device to monitor and regulate the injury site to facilitate healing.
Another goal for LIMB is to develop advanced bioprinting and biotextile processes to fabricate tissue-like constructs from biological materials. The lab also seeks to combine nanotechnological and bioprinting advances to build biomanufacturing platforms. LIMB recognized one application is treatment of musculoskeletal disorders.
Dr. Ali Tamayol, an associate professor at UConn’s Department of Biomedical Engineering and LIMB’s principal investigator, recently developed a handheld bioprinter pen that could significantly change the way musculoskeletal procedures are performed. The bioprinter enables surgeons to deposit materials to help support cell and tissue growth (scaffolds) directly into defect sites associated with weakened skeletal muscles. This project’s research was published at the end of January in ACS Applied Bio Materials.
“The printer is robust and allows proper filling of the cavity with fibrillar scaffolds in which fibers resemble the architecture of the native tissue,” Dr. Tamayol told the press.
The scaffolds essentially remove the need for suturing because they adhere precisely to the injury’s surrounding tissues and mimic the existing tissue’s properties. According to LIMB, the methods to treat volumetric muscle loss (VML) are inadequate. The geometry of skeletal muscle defects in VML injuries varies with each case. 3D printing emerged as one strategy to fabricate scaffolds matching the geometry of each defect site.
Dr. Indranil Sanha, plastic surgeon at Harvard’s Brigham and Women’s Hospital, who joined Dr. Tamayol in this study, has expertise in treating muscle injuries. Dr. Sinha said a “good solution currently does not exist for patients who suffer volumetric muscle loss. A customizable, printed gel establishes the foundation for a new treatment paradigm can improve the care of our trauma patients.”
The muscle-printing pen extrudes a material called gelatin methacrylol (GelMA), a collagen-like biomaterial designed to mimic skeletal tissue and act as a bioadhesive. Then, a UV light attached to the pen melds the strips of GelMA together, sort of like soldering.
Of course, existing 3D bioprinting technology has its share of issues. Successfully implanting hydrogel-based scaffolds requires a very specific biomaterial to be printed that will adhere to the defect site. 3D bioprinted scaffolds that mimic skeletal muscles have previously been created in vitro, but they haven’t yet been successfully implanted in an actual subject. Further, the time and facilities needed to image the defect site, process to render computer models, and print a suitable scaffold currently prevent immediate reconstructions following traumatic injuries.
Dr. Tamayol’s solutions solve this problem—his bioprinter prints gelatin-based hydrogels, or “bioink.” Based on LIMB’s research, this bioink proved effective in adhering to defect sites of mice with VML injury. The bioink hydrogel was cross-linked in situ, and its adhesiveness to skeletal muscles was assessed ex vivo. The mice demonstrated significant increase in muscle hypertrophy following Dr. Tamayol’s therapy. After 28 days of implantation, the LIMB team did not observe any inflammation or damage to surrounding tissue where the bioink scaffolding had adhered. Through testing, it was also found the scaffold had similar tear and shear strength to natural muscles.
“This is a new generation of 3D printers than enables clinicians to directly print the scaffold within the patient’s body,” said Dr. Tamayol. “Best of all, this system does not require the presence of sophisticated imaging and printing systems.”
While the study was only done on mice and can’t be translated to human subjects, Dr. Tamayol and Dr. Sinha have filed a patent on this technology to treat musculoskeletal injuries. Hopefully, they can move forward with developing future human trials.