Orthopedic Design & Technology Magazine
November/December 2020
-

Making the Grade: Orthopedic Materials Spotlight
Advancements with materials are enabling the use of fabrication processes not previously available for certain applications.11.18.20
-

Mending Mishaps: A Trauma Technology Overview
An examination of an orthopedic market not fueled by elective surgeries.11.18.20
-

Life, Interrupted: The 2020 Year in Review
Lessons learned from the global coronavirus pandemic.11.18.20
-
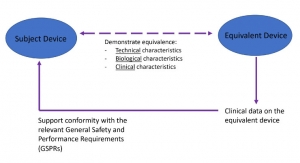 Regulatory PerspectivesMedical Device Equivalence Requirements Strengthened under the EU MDRThis column focuses on the directive under EU 2017/745 on medical devices (MDR), highlighting some of the changes in claiming and demonstrating equivalence.11.18.20
Regulatory PerspectivesMedical Device Equivalence Requirements Strengthened under the EU MDRThis column focuses on the directive under EU 2017/745 on medical devices (MDR), highlighting some of the changes in claiming and demonstrating equivalence.11.18.20
-
 Orthopedic InsightsSolid Footing During Shaky Times: Current Compliance ConsiderationsThe changes and challenges wrought by COVID-19 have significantly impacted the medtech industry.11.18.20
Orthopedic InsightsSolid Footing During Shaky Times: Current Compliance ConsiderationsThe changes and challenges wrought by COVID-19 have significantly impacted the medtech industry.11.18.20
-
 Best PracticesVirtually the Same? The Challenges of Online ConferencesLike everyone else on the planet, I’m turning increasingly to virtual solutions to our physical challenges in the midst of a pandemic.11.18.20
Best PracticesVirtually the Same? The Challenges of Online ConferencesLike everyone else on the planet, I’m turning increasingly to virtual solutions to our physical challenges in the midst of a pandemic.11.18.20
-
 Market SnapshotObesity and the Impact on Hip and Knee ImplantsObesity causes multiple health issues, and the COVID-19 pandemic is only adding to the problem.11.18.20
Market SnapshotObesity and the Impact on Hip and Knee ImplantsObesity causes multiple health issues, and the COVID-19 pandemic is only adding to the problem.11.18.20
-
 EditorialThe Lingering Impact of COVID-19There are lessons to be learned from this experience and there could even be elements that cause a lasting change regarding the orthopedic industry.11.18.20
EditorialThe Lingering Impact of COVID-19There are lessons to be learned from this experience and there could even be elements that cause a lasting change regarding the orthopedic industry.11.18.20
-
 The Last WordStryker Strikes Back After Pandemic SlumpLate last year, Stryker announced it would snap up Wright Medical to fortify its position in the trauma and extremities markets.11.18.20
The Last WordStryker Strikes Back After Pandemic SlumpLate last year, Stryker announced it would snap up Wright Medical to fortify its position in the trauma and extremities markets.11.18.20
-
 Columns13 Mistakes to Avoid in a 510k SubmissionThe FDA 510(k) review process can broadly be divided into two steps—acceptance review and substantive review.11.18.20
Columns13 Mistakes to Avoid in a 510k SubmissionThe FDA 510(k) review process can broadly be divided into two steps—acceptance review and substantive review.11.18.20
-
 ColumnsA Supply Chain Strategy for Pandemic SurvivalMedical device supply chain managers will likely conclude that 2020 is a year best forgotten.11.18.20
ColumnsA Supply Chain Strategy for Pandemic SurvivalMedical device supply chain managers will likely conclude that 2020 is a year best forgotten.11.18.20
















