Orthopedic Design & Technology Magazine
November/December 2021
-

Material Innovations Provide Solutions for New Orthopedic Products
Material advancements enable new opportunities for orthopedic device makers.11.17.21
-
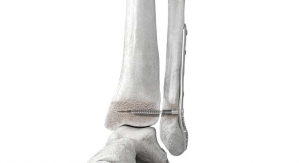
Extremities Devices Create Challenges and Opportunities for Manufacturers
An examination of the fragmented, high-growth extremity orthopedics market.11.17.21
-

Pandemic Creates Conditions for Buying Bonanza—2021 Year in Review
2021 offered a strong rebound in M&A activity in medtech, fueled by a record high of nearly $500 billion in financial firepower.11.17.21
-

CMS Creates Reimbursement Quagmire for ASCs—2021 Year in Review
CMS proposed reversing a three-year phaseout of its inpatient-only (IPO) service list.11.17.21
-

Supply Chain Struggles Plague Orthopedic Manufacturers—2021 Year in Review
Rising infections in Asia this past spring exacerbated shipping delays at southern China ports.11.17.21
-

MDR Finally Arrives, but Challenges Follow—2021 Year in Review
Resolving these challenges is critical to ensuring the MDR ship remains aloft and capable of fostering product innovation.11.17.21
-
 Regulatory PerspectivesFDA Regulatory Challenges for Antimicrobial Orthopedic DevicesPreventing and managing orthopedic-associated infections is one of the major challenges in orthopedic surgery.11.17.21
Regulatory PerspectivesFDA Regulatory Challenges for Antimicrobial Orthopedic DevicesPreventing and managing orthopedic-associated infections is one of the major challenges in orthopedic surgery.11.17.21
-
 Orthopedic InsightsEU MDR Interpretation and Understanding—Devil in the DetailsWhile the date to conform to all transitional requirements has passed, a new wave of scrutiny will arrive as organizations seek certification under the new MDR.11.17.21
Orthopedic InsightsEU MDR Interpretation and Understanding—Devil in the DetailsWhile the date to conform to all transitional requirements has passed, a new wave of scrutiny will arrive as organizations seek certification under the new MDR.11.17.21
-
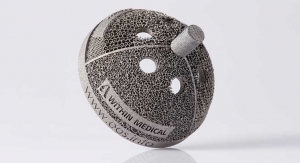 Technology ProfileDesigning Orthopedic Devices for Additive ManufacturingThe opportunities to leverage AM to improve current products or production processes in the here and now is enormous.11.17.21
Technology ProfileDesigning Orthopedic Devices for Additive ManufacturingThe opportunities to leverage AM to improve current products or production processes in the here and now is enormous.11.17.21
-
 Best PracticesCOVID and Life Have Me Rethinking My ConnectionsEarly in my career, I learned the value of following up on handshakes and intros, even if there was no obvious short-term benefit.11.17.21
Best PracticesCOVID and Life Have Me Rethinking My ConnectionsEarly in my career, I learned the value of following up on handshakes and intros, even if there was no obvious short-term benefit.11.17.21
-
 EditorialOrthopedics Device Industry: 2021 Review and 2022 ExpectationsThe end of a year marks a time to take a moment to reflect on what’s happened and what’s potentially coming.11.17.21
EditorialOrthopedics Device Industry: 2021 Review and 2022 ExpectationsThe end of a year marks a time to take a moment to reflect on what’s happened and what’s potentially coming.11.17.21
-
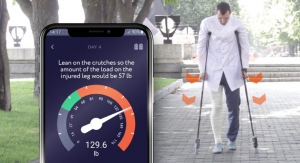 The Last Word‘Smart Crutches’ Help Boost Leg Injury RecoveryBased on a doctor’s weight-bearing parameters, an alert is sent if the leg is loaded incorrectly with too much or too little weight.11.17.21
The Last Word‘Smart Crutches’ Help Boost Leg Injury RecoveryBased on a doctor’s weight-bearing parameters, an alert is sent if the leg is loaded incorrectly with too much or too little weight.11.17.21
-
 ColumnsOvercoming Packaging Challenges with Innovative MaterialsAt times, packaging orthopedic devices such as implants can prove to be challenging because of their size and sterility requirements.11.17.21
ColumnsOvercoming Packaging Challenges with Innovative MaterialsAt times, packaging orthopedic devices such as implants can prove to be challenging because of their size and sterility requirements.11.17.21
-
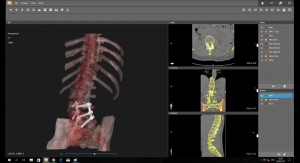 ColumnsMedical Device Manufacturing at the Point of CareAdditive manufacturing is a tool that can be reasonably deployed at the point of care.11.17.21
ColumnsMedical Device Manufacturing at the Point of CareAdditive manufacturing is a tool that can be reasonably deployed at the point of care.11.17.21
















