Until now, it has been virtually impossible to manufacture implants from polymer compounds with bioactive particles by injection molding, because a thin polymer layer usually forms on the surface that overlays the particles, preventing the implant from growing in. “Implant manufacturers were missing a PEEK material that fuses with bone cells and thus accelerates healing, for example after spinal surgery,” says Marc Knebel, Head of the Medical Systems Market Segment at Evonik.
The functional specialty additive is biphasic calcium phosphate (BCP). Calcium phosphates are a natural component of bone. If osteoblasts find body-like substances at the implant, they can dock there more easily. This positively influences osteointegration, the interfacial fusion between bone and implant.
“We tested VESTAKEEP® Fusion on injection molding machines in the clean room and found that it was very easy to process,” confirms Samaplast CEO Stefan Okle. “Thanks to the excellent exchange with Evonik, we were able to produce a prototype within ten days.” Previously, tool development alone had taken eight to ten weeks. With rapid prototyping, this process can be shortened considerably.
For manufacturers of implants, VESTAKEEP® Fusion offers the added value of fully preserving the osteoconductive function during injection molding. With this unique combination of material and processing properties, osteoconductive plastic implants can now be manufactured in just one production step. There is no need to additionally coat them with titanium or other expensive processes. This saves costs and improves product quality.
The ability to process VESTAKEEP® Fusion by injection molding expands the range of applications for osteoconductive implants, particularly in spinal surgery. Until now, spinal implants made of PEEK have mainly been milled. There are a variety of designs and sizes on the market, often produced in small quantities. And this is precisely where there is great potential for injection molding osteoconductive PEEK implants with VESTAKEEP® Fusion: different modules can be produced with one basic mold.

Spinal cage prototype demonstrates design versatility.
Thanks to the design freedom in injection molding of VESTAKEEP® Fusion, specific cage variants can be produced—tailored to the anatomical conditions and different surgical techniques such as PLIF, TLIF, and ALIF. The cage models show the design diversity in the interlocks and in the cavities for ossification. In addition, the design facilitates instrument guidance during surgery and also enables minimally invasive, patient-friendly surgical techniques.
In vitro studies document over 30 percent increased cell attachment and cell proliferation. In preclinical tests, histological examination reveals a dense network of newly formed osteoblasts at the bone-implant interface. Bone histomorphometry shows an approximately twofold increase in bone apposition. And pull-out tests demonstrate a more than twofold increase in implant fixation.
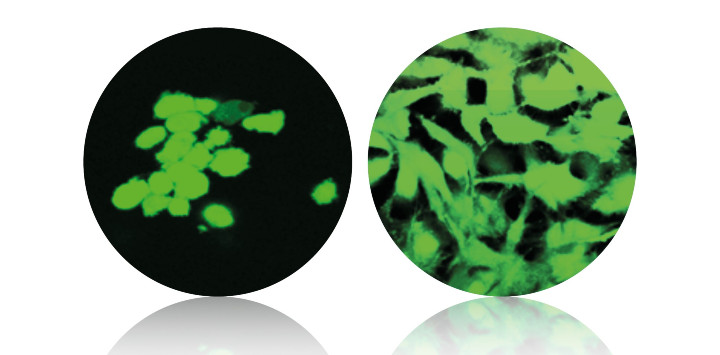
In vitro studies document over 30 percent increased cell attachment and cell proliferation.
Click here to find out more about Evonik >>>>>
Biphasic Calcium Phosphate Accelerates Healing
The polymer designers from Evonik have succeeded in giving PEEK osteoconductive properties with the help of an additive, i.e., modifying the compound so that bone cells adhere to the implant. The development team at Evonik explored more than 50 different formulations to match the morphology and size of the particles to achieve optimal mechanical properties so that the functional additives are available on the surface of the injection molded part. “We are closing this gap in the market with VESTAKEEP® Fusion and offering an alternative to titanium, stainless steel or cobalt-chromium implants right away,” Knebel adds.The functional specialty additive is biphasic calcium phosphate (BCP). Calcium phosphates are a natural component of bone. If osteoblasts find body-like substances at the implant, they can dock there more easily. This positively influences osteointegration, the interfacial fusion between bone and implant.
Osteoconductive PEEK Biomaterial for Injection Molding
VESTAKEEP® Fusion was developed with a goal to process in different manufacturing technologies and is initially offered as granules and semi-finished products. It can be milled, compression molded, extruded, and even injection molded. For the latter, Evonik has optimized the compound in close cooperation with Samaplast AG.“We tested VESTAKEEP® Fusion on injection molding machines in the clean room and found that it was very easy to process,” confirms Samaplast CEO Stefan Okle. “Thanks to the excellent exchange with Evonik, we were able to produce a prototype within ten days.” Previously, tool development alone had taken eight to ten weeks. With rapid prototyping, this process can be shortened considerably.
For manufacturers of implants, VESTAKEEP® Fusion offers the added value of fully preserving the osteoconductive function during injection molding. With this unique combination of material and processing properties, osteoconductive plastic implants can now be manufactured in just one production step. There is no need to additionally coat them with titanium or other expensive processes. This saves costs and improves product quality.
The ability to process VESTAKEEP® Fusion by injection molding expands the range of applications for osteoconductive implants, particularly in spinal surgery. Until now, spinal implants made of PEEK have mainly been milled. There are a variety of designs and sizes on the market, often produced in small quantities. And this is precisely where there is great potential for injection molding osteoconductive PEEK implants with VESTAKEEP® Fusion: different modules can be produced with one basic mold.
Cage Prototype Demonstrates the Design Diversity
Evonik and Samaplast are demonstrating what is possible with the first prototypes of intervertebral implants, known as cages or spacers, which are used to fill the disc space in the spine following its removal due to disc degeneration. “We have produced a cage that contains all the details, such as serrations, threads, and apertures, that are relevant for spinal implants,” explains Okle.
Spinal cage prototype demonstrates design versatility.
Thanks to the design freedom in injection molding of VESTAKEEP® Fusion, specific cage variants can be produced—tailored to the anatomical conditions and different surgical techniques such as PLIF, TLIF, and ALIF. The cage models show the design diversity in the interlocks and in the cavities for ossification. In addition, the design facilitates instrument guidance during surgery and also enables minimally invasive, patient-friendly surgical techniques.
Over 30 Percent Higher Cell Proliferation
With VESTAKEEP® Fusion, Evonik is introducing a new product line of next-generation PEEK-based implant materials. The osteoconductive properties of the new PEEK material were achieved by using a special functional additive—biphasic calcium phosphate—and allow bone cells to adhere to implants more quickly, thus positively influencing fusion, so called osteointegration, at the boundary between the bone and the implant. This, in turn, will accelerate bone fusion and convalescence.In vitro studies document over 30 percent increased cell attachment and cell proliferation. In preclinical tests, histological examination reveals a dense network of newly formed osteoblasts at the bone-implant interface. Bone histomorphometry shows an approximately twofold increase in bone apposition. And pull-out tests demonstrate a more than twofold increase in implant fixation.

In vitro studies document over 30 percent increased cell attachment and cell proliferation.
OSTEO PEEK Filament for 3D Printed Medical Implants
In the meantime, Evonik has launched its osteoconductive PEEK filament for 3D printed implants. The high-performance polymer can be processed in common extrusion-based 3D printing technologies such as fused filament fabrication (FFF).Click here to find out more about Evonik >>>>>













