PR Newswire05.27.16
Interventional Spine Inc. has received U.S. Food and Drug Administration (FDA) clearance for its 80 Lumbar Lordotic Opticage Expandable Intervertebral Body Fusion Device. The product joins the Interventional Spine family of Opticage Expandable Interbody Fusion Devices cleared by the FDA late last year, and also used with the company’s PerX360 System.
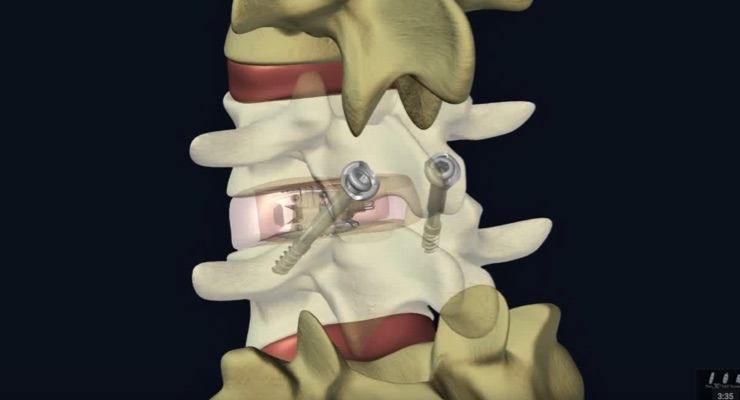
Watch the video below to learn more about Interventional Spine's PerX360 System:
The Lordotic 80 Opticage is a lumbar intervertebral body fusion device and is indicated for use with autogenous bone graft in patients with degenerative disc disease (DDD) at one or two contiguous levels from L2-S1.
Lumbar Lordosis is a condition in which the spine in the lower back tends to curve more than normal. As a result of the abnormal curvature of the lower back, increased pressure is put on the lower spine resulting in pain for the patient. The Lordotic 80 Opticage is an option that surgeons may use to help correct the pressure on the lower spine potentially alleviating the pain that patients experience as a result of lordosis. The new Opticage provides options to be implanted via posterior, transforaminal or lateral approach. In addition, the Lordotic 80 Opticage provides for optimum graft injection through its patented delivery system.
Interventional Spine is currently in the process of assessing other sizes and configurations for its line of Lordotic Expandable Opticages. “Based on existing market trends, Interventional Spine expects the new Lordotic Opticages to be a significant source of revenue for the company,” said Joseph Darling, Interventional Spine’s chief operating officer. “More and more surgeons have been requesting a Lumbar Lordotic expandable cage from us. We are very excited with the latest addition of the Lordotic 80 Opticage, as it provides a pathway for Interventional Spine’s vision of becoming a leader in the expandable cage market, and addresses the needs of surgeons and patients worldwide.’
Interventional Spine Inc. is a privately held company based in Irvine, Calif., that designs, develops, and markets patented implantable spinal devices that can be deployed through both traditional and minimally invasive techniques.
Watch the video below to learn more about Interventional Spine's PerX360 System:
The Lordotic 80 Opticage is a lumbar intervertebral body fusion device and is indicated for use with autogenous bone graft in patients with degenerative disc disease (DDD) at one or two contiguous levels from L2-S1.
Lumbar Lordosis is a condition in which the spine in the lower back tends to curve more than normal. As a result of the abnormal curvature of the lower back, increased pressure is put on the lower spine resulting in pain for the patient. The Lordotic 80 Opticage is an option that surgeons may use to help correct the pressure on the lower spine potentially alleviating the pain that patients experience as a result of lordosis. The new Opticage provides options to be implanted via posterior, transforaminal or lateral approach. In addition, the Lordotic 80 Opticage provides for optimum graft injection through its patented delivery system.
Interventional Spine is currently in the process of assessing other sizes and configurations for its line of Lordotic Expandable Opticages. “Based on existing market trends, Interventional Spine expects the new Lordotic Opticages to be a significant source of revenue for the company,” said Joseph Darling, Interventional Spine’s chief operating officer. “More and more surgeons have been requesting a Lumbar Lordotic expandable cage from us. We are very excited with the latest addition of the Lordotic 80 Opticage, as it provides a pathway for Interventional Spine’s vision of becoming a leader in the expandable cage market, and addresses the needs of surgeons and patients worldwide.’
Interventional Spine Inc. is a privately held company based in Irvine, Calif., that designs, develops, and markets patented implantable spinal devices that can be deployed through both traditional and minimally invasive techniques.