Business Wire07.28.16
Orthofix International N.V., a diversified, global medical device company, today announced regulatory approval from the Japanese Ministry of Health, Labor and Welfare (MHLW) of the PHOENIX Minimally Invasive Spinal (MIS) Fixation System. The introduction of PHOENIX will set the stage for Orthofix to bring more of its innovative spine technologies to this important market in the near future.
“The Japanese approval and full market launch of the PHOENIX MIS fixation system represents an important step towards broadening the availability of our spine fixation products globally by entering the second largest spine market in the world,” said Ray Fujikawa, President of Orthofix Spine Fixation. “We are committed to continuing to work towards securing additional product approvals in order to provide surgeons in Japan with a full range of our spine fixation solutions.”
Estimates by healthcare market research firm GlobalData indicate that by 2020 the spinal fusion market will be $684 million in Japan.
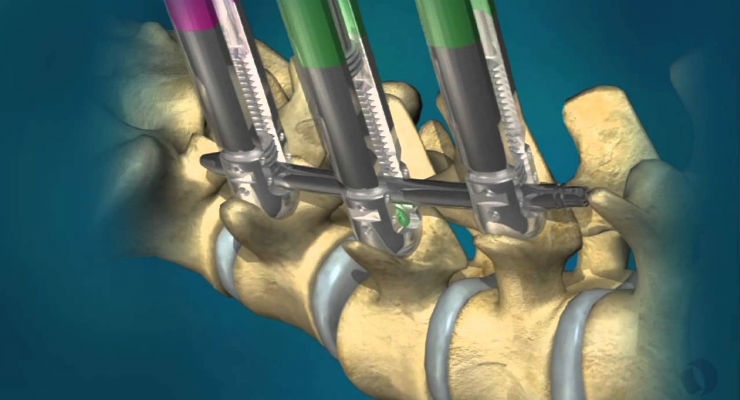
The PHOENIX MIS system enables the percutaneous placement of pedicle screws designed to provide stabilization and correction to assist fusion in patients suffering from degenerative disc disease, spinal stenosis and trauma conditions in the middle and lower back. Featuring a unique implant design for percutaneous and mini open surgical approaches, the PHOENIX MIS system permits a smaller working diameter, thereby reducing the surgical incision.
“The Japanese approval and full market launch of the PHOENIX MIS fixation system represents an important step towards broadening the availability of our spine fixation products globally by entering the second largest spine market in the world,” said Ray Fujikawa, President of Orthofix Spine Fixation. “We are committed to continuing to work towards securing additional product approvals in order to provide surgeons in Japan with a full range of our spine fixation solutions.”
Estimates by healthcare market research firm GlobalData indicate that by 2020 the spinal fusion market will be $684 million in Japan.
The PHOENIX MIS system enables the percutaneous placement of pedicle screws designed to provide stabilization and correction to assist fusion in patients suffering from degenerative disc disease, spinal stenosis and trauma conditions in the middle and lower back. Featuring a unique implant design for percutaneous and mini open surgical approaches, the PHOENIX MIS system permits a smaller working diameter, thereby reducing the surgical incision.