Marketwired04.25.17
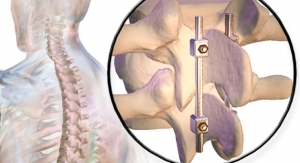
Amedica Corporation, a developer and commercializer of silicon nitride, has been granted marketing clearance for its Valeo interbody fusion devices in Australia.
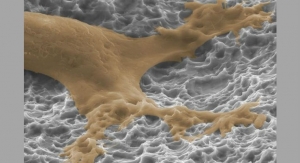
The Valeo product line is made entirely of Amedica’s proprietary medical grade silicon nitride ceramic—an ideal material for fusion because of its nanostructured surface, osteoconductivity, osteoinductivity, anti-microbial properties, and ease of radiographic imaging.
“The Australian clearance is a tremendous opportunity for Amedica. Interestingly, during 1986-1988, Australian surgeons and engineers were the first to synthesize reaction-bonded silicon nitride, and implant the material in lumbar spine fusion patients. Favorable outcomes in terms of pain relief and successful fusion were reported at the 15-year follow-up time interval. Thus, the pioneering clinical use of silicon nitride in spine fusion originated in Australia, and we are excited to see the material return to its place of birth,” said Dr. Sonny Bal, Amedica CEO. “The successful outcomes from Australia are now validated by 45 scientific papers published by Amedica in journals, and clinical data that will be published throughout this year. We look forward to working with Australian surgeons, and providing them with improved options for spinal fusion surgery.”
Amedica is the only manufacturer of medical grade silicon nitride—a biomaterial technology ideal for use in spinal fusion and across a variety of medical applications. The company’s products are manufactured in its ISO 13485 certified manufacturing facility located in Salt Lake City, Utah. Amedica’s U.S. Food and Drug Administration-cleared and CE-marked spine products are currently marketed in the United States and select markets in Europe and South America through its distributor network.
The Valeo product line is made entirely of Amedica’s proprietary medical grade silicon nitride ceramic—an ideal material for fusion because of its nanostructured surface, osteoconductivity, osteoinductivity, anti-microbial properties, and ease of radiographic imaging.
“The Australian clearance is a tremendous opportunity for Amedica. Interestingly, during 1986-1988, Australian surgeons and engineers were the first to synthesize reaction-bonded silicon nitride, and implant the material in lumbar spine fusion patients. Favorable outcomes in terms of pain relief and successful fusion were reported at the 15-year follow-up time interval. Thus, the pioneering clinical use of silicon nitride in spine fusion originated in Australia, and we are excited to see the material return to its place of birth,” said Dr. Sonny Bal, Amedica CEO. “The successful outcomes from Australia are now validated by 45 scientific papers published by Amedica in journals, and clinical data that will be published throughout this year. We look forward to working with Australian surgeons, and providing them with improved options for spinal fusion surgery.”
Amedica is the only manufacturer of medical grade silicon nitride—a biomaterial technology ideal for use in spinal fusion and across a variety of medical applications. The company’s products are manufactured in its ISO 13485 certified manufacturing facility located in Salt Lake City, Utah. Amedica’s U.S. Food and Drug Administration-cleared and CE-marked spine products are currently marketed in the United States and select markets in Europe and South America through its distributor network.