Business Wire05.03.17
European regulators have granted CE Marking to IMPLANET's new JAZZ Standalone implant.
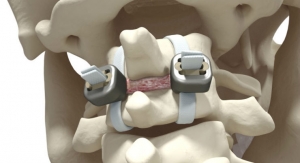
JAZZ Standalone is an implant that further strengthens the device’s freestanding, rodless fixation line, launched in 2016 with the JAZZ Lock implant. It is used in the treatment of adult degenerative bone disorders, the largest spine surgery market segment, representing $246 million in Europe, according to the company.
“The result of our work with thought-leading surgeons, the JAZZ Standalone is a new variant of our JAZZ technology devoted to treating degenerative pathologies," said Régis Le Couedic, Implanet’s product development and manufacturing director. "This implant utilizes the JAZZ Band platform and enables vertebral segments to be stabilized without the need for pedicle screws and rods. This is a fast, simple and safe surgical procedure that only requires limited surgical accessibility.”
The JAZZ Standalone offers fast and simple freestanding posterior fixation to replace traditional fixation systems. Degenerative spine surgery is experiencing strong growth in Europe and the United States, driven by an aging demographic that demands functional restoration in a less invasive manner.
The JAZZ Standalone will be incorporated in the JAZZ product range without new investments in equipment or training for surgical teams.
“We are continuing to rigorously pursue our execution plan by regularly marketing new products and line extensions. The marketing approval of a major new component of the JAZZ Band platform in Europe is another key breakthrough in terms of expanding the range," said Ludovic Lastennet, Implanet CEO. "Indeed, the JAZZ Standalone meets a major and persistent demand from surgeons regarding their need to continually simplify surgical procedures. It should rapidly be adopted by our partners, whether they be pediatric surgeons or surgeons specializing in adult degenerative disorders.”
Founded in 2007, IMPLANET is an orthopedic implant manufacturer. Its flagship product, the JAZZ latest-generation implant, aims to treat spinal pathologies requiring vertebral fusion surgery. Protected by four families of international patents, JAZZ has obtained 510(k) regulatory clearance from the U.S. Food and Drug Administration and the CE mark. IMPLANET employs 48 staff and recorded 2016 sales of 7.8 million euros. Based near Bordeaux in France, IMPLANET established a U.S. subsidiary in Boston, Mass., in 2013.
JAZZ Standalone is an implant that further strengthens the device’s freestanding, rodless fixation line, launched in 2016 with the JAZZ Lock implant. It is used in the treatment of adult degenerative bone disorders, the largest spine surgery market segment, representing $246 million in Europe, according to the company.
“The result of our work with thought-leading surgeons, the JAZZ Standalone is a new variant of our JAZZ technology devoted to treating degenerative pathologies," said Régis Le Couedic, Implanet’s product development and manufacturing director. "This implant utilizes the JAZZ Band platform and enables vertebral segments to be stabilized without the need for pedicle screws and rods. This is a fast, simple and safe surgical procedure that only requires limited surgical accessibility.”
The JAZZ Standalone offers fast and simple freestanding posterior fixation to replace traditional fixation systems. Degenerative spine surgery is experiencing strong growth in Europe and the United States, driven by an aging demographic that demands functional restoration in a less invasive manner.
The JAZZ Standalone will be incorporated in the JAZZ product range without new investments in equipment or training for surgical teams.
“We are continuing to rigorously pursue our execution plan by regularly marketing new products and line extensions. The marketing approval of a major new component of the JAZZ Band platform in Europe is another key breakthrough in terms of expanding the range," said Ludovic Lastennet, Implanet CEO. "Indeed, the JAZZ Standalone meets a major and persistent demand from surgeons regarding their need to continually simplify surgical procedures. It should rapidly be adopted by our partners, whether they be pediatric surgeons or surgeons specializing in adult degenerative disorders.”
Founded in 2007, IMPLANET is an orthopedic implant manufacturer. Its flagship product, the JAZZ latest-generation implant, aims to treat spinal pathologies requiring vertebral fusion surgery. Protected by four families of international patents, JAZZ has obtained 510(k) regulatory clearance from the U.S. Food and Drug Administration and the CE mark. IMPLANET employs 48 staff and recorded 2016 sales of 7.8 million euros. Based near Bordeaux in France, IMPLANET established a U.S. subsidiary in Boston, Mass., in 2013.